Introduction
The mammalian target of rapamycin (mTOR) inhibitors sirolimus and everolimus (EVR) are immunosuppressive drugs commonly used in renal transplantation. mTOR inhibitors have antiangiogenic and antineoplastic properties, and therefore are associated with a lower risk of malignancy than other immunosuppressive agents. Reduced incidence of cytomegalovirus and BK virus (BKV) infections in kidney transplant (KTx) recipients receiving EVR were also reported [1, 2]. Pulmonary adverse effects are relatively common in KTx recipients treated with mTOR inhibitors with a reported frequency of 2-11% and the onset of symptoms between 1 and 51 months after the initiation of therapy [3]. Several distinct types of pulmonary damage have been recognized, including lymphocytic interstitial pneumonitis, lymphocytic alveolitis, bronchiolitis obliterans with organizing pneumonia, focal pulmonary fibrosis, and even diffuse alveolar hemorrhage [3-5]. At the onset of this complication, patients with mTOR inhibitor-associated pneumonitis usually present with a cough and/or dyspnea and/or respiratory insufficiency, and sometimes with systemic symptoms such as fever and fatigue. Occasionally, only changes on high-resolution computed tomography (HRCT) scans are observed, with no accompanying clinical symptoms. The most common on HRCT scans are ground-glass opacities, inter-/intra-lobular septal linear thickening, or multifocal areas of parenchymal lung consolidation with a basilar predominance [3-5]. Also rare cases of opportunistic pulmonary infections, Pneumocystis jiroveci pneumonia and invasive pulmonary aspergillosis, mimicking typical noninfectious mTOR inhibitor-induced pulmonary adverse events, were reported in both EVR and temsirolimus treated patients [6-8].
Even though both the clinical symptoms and radiological signs have been well described, the etiopathogenic mechanism of mTOR-inhibitor-associated pulmonary toxicity is still unclear.
Case report
A 68-year-old man with end-stage renal disease of unknown etiology underwent a KTx from a 32-year old deceased female donor, who died of head trauma (cytomegalovirus D–/R+, Epstein-Barr virus D+/R+). He had a past history of type 2 diabetes, hypertension, coronary artery disease and squamous cell carcinoma of the tibial area resected 8 years before the KTx. His past medical history also included pulmonary tuberculosis. He had been maintained on haemodialysis for the preceding seven years. At the time of KTx he was in a good general condition. HLA mismatching was 0 : 1 : 0 (A, B, DR), respectively. Anesthesia and surgery were uneventful, with good initial kidney perfusion. He underwent basiliximab induction therapy and commenced EVR, cyclosporine (CsA) and prednisone maintenance therapy. EVR was introduced because of the history of squamous cell carcinoma. CsA was chosen over tacrolimus in order to prevent significant deterioration of glycemic control in a patient with type 2 diabetes. The doses of EVR and cyclosporine were adjusted to maintain standard target trough levels. The chest X-ray performed on the day of KTx showed no obvious abnormality. Three months later the patient presented with fever, progressive dyspnea, and a productive cough for two weeks. On general examination, the patient was conscious and well-oriented. He had a pulse rate of 108/min, lood pressure of 100/60 mmHg, respiratory rate of 28 cycles/min and there was evident cyanosis. Room air oxygen saturation was around 90%. Other systemic examinations were normal. Admission chest radiography showed patchy infiltrates suggestive of bacterial pneumonia in the left inferior segments corresponding to crackles, rhonchi and wheezes on auscultation. His laboratory testing revealed a white blood cell (WBC) count of 15.8 × 109/l, hemoglobin 11 g/dl, platelet count 216 × 109/l, absolute neutrophil count 14.9 × 109/l, absolute lymphocyte count 0.32 × 109/l, and C-reactive protein (CRP) 281 mg/l. Kidney function was stable with serum creatinine of 0.92 mg/dl and MDRD eGFR on admission > 60 ml/min/1.73 m2. The EVR level was 7.1 ng/ml and cyclosporine level 150 ng/ml (Table 1).
Table 1
Trough levels of cyclosporine and everolimus after kidney transplantation
He initially received amoxicillin/clavulanic acid. Bacterial cultures of blood and urine were negative. The symptoms improved, and fever resolved, with a decrease in both WBC and CRP levels over the next few days with empiric antibiotic treatment. There was clinical improvement, but without a radiological resolution. An HRCT scan (Fig. 1A) revealed a disseminated nodular pattern in both lungs, accompanied by peribronchial consolidations and lung parenchyma retraction dominating in the lower lobes. No pleural effusion was seen. Bronchoscopy was unremarkable. A transbronchial biopsy revealed mild nonspecific inflammation with areas of fibrosis in the bronchial walls. Bronchoalveolar lavage was sent for cell count, bacterial and viral culture, and fungal and acid-fast bacillus analysis. Both sputum and bronchoalveolar lavage cultures were positive for Achromobacter denitrificans. After a complete diagnostic workup including repeated bacterial and mycobacterial cultures, QuantiFERON1-TB Gold test, blood CMV-PCR, blood Aspergillus galactomannan and Candida mannan, immunofluorescent staining of bronchoalveolar fluid for Pneumocystis jiroveci, other pulmonary opportunistic infections were ruled out, and a diagnosis of A. denitrificans pneumonia and EVR pulmonary toxicity was made. A 17-day course of piperacillin/tazobactam chosen according to the susceptibility profile (Table 2), and a reduction of the EVR dose to the target trough levels of 2.8-3.2 ng/ml resulted only in partial resolution of radiological abnormalities confirmed by HRCT (Fig. 1B). After three weeks of antibiotic treatment EVR was withheld. EVR discontinuation with no additional antibiotic treatment resulted in complete recovery and a complete resolution of pulmonary infiltrates in an HRCT performed at a further 10-week follow-up (Fig. 1C). The immunosuppressive regimen on discharge was restricted to cyclosporine and glucocorticosteroids.
Fig. 1
A) Initial computed tomography (CT) of the lungs shows poorly defined bilateral patchy consolidations without pleural effusion, B) chest CT after 3-week antibiotic treatment and everolimus (EVR) dose reduction shows only partial resolution of bilateral patchy consolidations, C) CT performed 10 weeks after EVR discontinuation shows complete resolution of symptoms
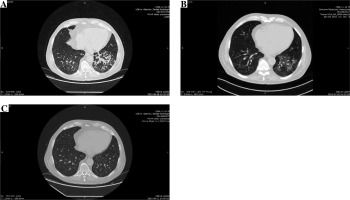
Table 2
Antimicrobial susceptibility testing for Achromobacter denitrificans strains isolated from both sputum and broncho-alveolar lavage
We performed a functional analysis of peripheral blood neutrophils and monocytes using Phagotest and Phagoburst tests (both Glycotope Biotechnology, Germany).
Phagotest measures the ability to perform phagocytosis, or specifically the ability of the phagocytes to take up the bacteria, by assessing the level of fluorescence emitted by phagocytes – both monocytes and neutrophils. The fluorescence comes from fluorescently stained bacteria engulfed by the phagocytes. Phagotest was performed in accordance with the manufacturer’s protocol and with modifications. The original protocol involves the use of Escherichia coli stained with FITC (fluorescein) only (included in kit). Phagotest was also used for assessing the phagocytosis of A. denitrificans stained with CFDA (carboxyfluorescein diacetate succinimidyl ester). This method was established in our laboratory and described elsewhere [9]. The fluorescence emitted by phagocytes reflects the intensity of the engulfment of bacteria. Apart from using different bacteria, all stages of the manufacturer’s manual were performed in the same manner for E. coli and A. denitrificans phagocytosis assessment. Phagoburst measures the ability of phagocytes to effectively kill the bacteria by the oxidative burst. The intensity of oxidative burst is estimated by assessing the conversion of non-fluorescent DHR123 (dihydrorhodamine 123) into green fluorescence-emitting R123 (rhodamine 123) in the presence of reactive oxidants. The higher the fluorescence is, the more intense is the oxidative burst. As in case of Phagotest, the assessment with E. coli (already in the manufacturer’s kit) and A. denitrificans isolated from the patient’s sputum and bronchoalveolar lavage was performed according to the manufacturer’s protocol. The fluorescence intensity was assessed using a FACScan cytometer (BD, USA).
Flow cytometry techniques have been extensively used to evaluate phagocyte function. Both Phagotest and Phagoburst have been shown to be useful tests for the evaluation of phagocytosis and oxidative burst activity in different research contexts, providing repeatable and credible results [10-13].
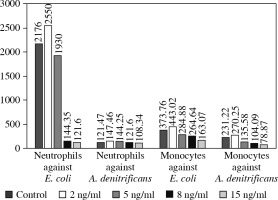
The ex vivo phagocytic activity (Fig. 2) and intensity of oxidative burst (Fig. 3) measured by the mean fluorescent intensity (MFI) of neutrophils and monocytes against E. coli and A. denitrificans were assessed using patients’ cells collected on EVR (2.8 ng/ml) and using patient cells collected 3 weeks after EVR discontinuation, and using cells from a healthy control.
Fig. 2
Ex vivo phagocytic activity of neutrophils and monocytes against Escherichia coli and achromobacter denitrificans measured by mean fluorescent intensity (MFI)
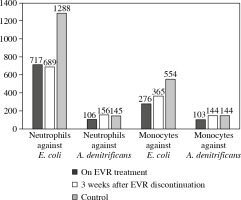
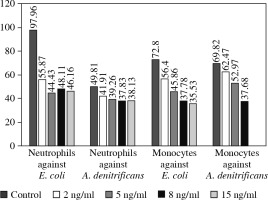
Fig. 3
Ex vivo intensity of oxidative burst of neutrophils and monocytes against Escherichia coli and achromobacter denitrificans measured by mean fluorescent intensity (MFI)
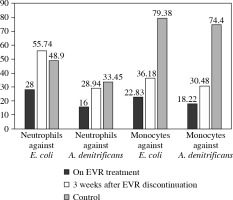
The phagocytic activity against A. denitrificans and E. coli of patient neutrophils and monocytes collected during EVR treatment was notably lower compared to neutrophils and monocytes from a healthy control, especially in the case of E. coli. The phagocytic activity of patient neutrophils and monocytes after 3 weeks of EVR absence was higher than patient cell activity in the presence of EVR. The phagocytic activity of patient cells off EVR against A. denitrificans was similar to the activity of healthy controls; however, the activity of patient cells off EVR against E. coli was still below the activity of control cells (Fig. 2).
The level of oxidative burst from neutrophils and monocytes, even more so, obtained from the patient on EVR treatment was significantly lower than the oxidative burst from the healthy control cells. After 3 weeks of EVR absence, the oxidative burst of patient neutrophils engulfing E. coli and A. denitrificans was restored to a level similar to the healthy control, even though the patient continued to receive prednisone and CsA. The level of oxidative burst carried out by patient monocytes was also improved with EVR absence, but still lower by half compared to monocytes of a healthy control (Fig. 3).
To confirm the aforementioned EVR properties we performed a similar assay in vitro using peripheral blood neutrophils and monocytes from a healthy donor. These cells were exposed to four different concentrations of EVR, ranging from 2 to 15 ng/ml (2 ng/ml, 5 ng/ml, 8 ng/ml and 15 ng/ml). The processes of phagocytosis and oxidative burst generation against both A. denitrificans and E. coli were again significantly affected by increasing doses of EVR in a dose-dependent manner (Figs. 4 and 5).
Discussion and conclusions
Few animal or in vitro studies have evaluated the pathogenesis of mTOR inhibitor-induced lung toxicities, and the underlying mechanisms remain to be determined. An immunological dose-independent origin is suggested, with three main mechanisms involved. First, mTOR inhibitors might expose cryptic antigens and induce an autoimmune response [14]. Second, a delayed-type hypersensitivity reaction is possible with an mTOR inhibitor as a hapten [15]. Third, pulmonary inflammation could be the consequence of cytokine production alterations caused by mTOR inhibitors. In vitro studies confirm that mTOR inhibitors stimulate the release of a number of proinflammatory cytokines such as interleukin (IL)-12, IL-23, tumor necrosis factor (TNF) and IL-1β while inhibiting the secretion of the anti-inflammatory cytokine IL-10 [15-17]. Additionally, a dose-related effect – and therefore directly toxic mTOR effect – has been suggested [14, 18].
Washino et al. found in an animal model that treatment with another mTOR inhibitor, temsirolimus, depleted alveolar macrophages in vivo, which, as the authors hypothesized, might lead to an accumulation of surfactant lipids, a condition seen in many respiratory pathologies. Temsirolimus also inhibited macrophage proliferation at lower drug concentrations, whereas it induced cell death in primary cultured alveolar macrophages at higher concentrations in vitro [19]. In another study by Wislez et al. [20] temsirolimus induced apoptosis of intraepithelial macrophages in alveolar epithelial neoplasia in K-rasLA1 mice. Several clinical case reports of EVR-induced lung injury also present macrophage depletion in BAL cell analyses [21, 22]. Observations on the mTOR inhibitor influence on neutrophils and monocytes/macrophages are consistent with our results showing dose-dependent impairment of neutrophil/monocyte phagocytic activity and oxidative burst generation. Fernandez-Botran et al. reported that in patients suffering from community-acquired pneumonia, blood neutrophil functional responses (phagocytosis and phagocytosis-stimulated respiratory burst activity) were elevated as compared to healthy controls [23]. We observed that in our patient with pneumonia blood neutrophil and monocytes functional responses were significantly reduced after exposure to EVR, as demonstrated by ex vivo assays.
It is known that alveolar macrophages are major effector cells in initiating and orchestrating the respiratory immune response to infectious agents. Alveolar macrophage depletion and inadequate alveolar macrophage function may result in an increase in pulmonary surfactants, but also affect the resolution of tissue inflammation, leading to collateral damage. Macrophage-depleted mice showed both higher concentrations of intrapulmonary cytokines, with greater numbers of activated polymorphonuclear cells, and accumulation of apoptotic and secondary necrotic neutrophils when compared to control mice [24].
Achromobacter denitrificans is a gram-negative, mobile, strictly aerobic, ubiquitous, nonglucosefermenting , oxidase- and catalase-positive bacterium present in soil and water [25]. This bacterium only rarely causes human infections. Most of the infections by Achromobacter are asymptomatic. The symptomatic infections usually occur in immunodeficient patients and include cases of bacteriemia, natural valve or prosthetic valve endocarditis, meningitis, pneumonia, peritonitis, conjunctivitis, osteomyelitis, intra-abdominal abscess, and prosthesis infections [26].
Therefore, the observed A. denitrificans pneumonia was probably a superimposed phenomenon resulting from the same, as in the case of mTOR-related pneumonitis and impaired function of neutrophils and monocytes/macrophages. Given the presented clinical scenario it is difficult to clearly distinguish between the relative contributions of infection and possible EVR toxicity. Other cases of opportunistic pulmonary infections, Pneumocystis jiroveci pneumonia and invasive pulmonary aspergillosis, mimicking typical pulmonary adverse noninfectious mTOR inhibitor-induced pneumonitis and cryptogenic organizing pneumonia, have been reported previously in both EVR and temsirolimus treated patients [6-8]. In all three cases the identification of opportunistic pathogens in both bronchoalveolar lavage and biopsy specimens was an unexpected finding. Pneumocystis jiroveci infection induced only mild chronic inflammation assessed by bronchoalveolar lavage fluid analysis, and the authors concluded that the paucity of inflammatory reaction could result from immunosuppression due to cancer recurrence or glucocorticosteroid treatment [6]. This could also reflect the impaired function of neutrophils and monocytes/macrophages.
In a large phase III clinical trial program of EVR the incidence of interstitial lung disease was 0.4%. In three of the four heart Tx recipients the discontinuation of EVR resulted in interstitial lung disease improvement or resolution, while there was no improvement in a patient who continued to receive EVR. The outcome was fatal in the KTx recipient, in whom EVR therapy was continued and in the liver Tx recipient despite EVR discontinuation, probably due to a superimposed infection. After having analyzed the available literature, the authors concluded that prompt discontinuation of mTOR inhibitor therapy as soon as interstitial lung disease is diagnosed is crucial to ensure a favorable outcome [27].
Withdrawal of immunosuppression is a key component of treating infections in immunocompromised patients. However, the neutrophil and monocyte/macrophage function clearly improved after withdrawing EVR, even though the patient was still maintained on prednisolone and cyclosporine. The in vitro dose-dependent impairment of a healthy volunteer’s neutrophil/monocyte functional responses also would suggest direct EVR toxicity. This is in agreement with a study by Solazzo et al., which reported a relationship of interstitial lung disease to EVR trough concentration > 9.03 ng/ml in a group of 500 KTx recipients on an EVR and CsA combined protocol [28].
In conclusion, the present report is the first to suggest dose-dependent impairment of neutrophil/monocyte functional responses such as phagocytic activity and oxidative burst generation, as possible mechanisms of mTOR associated pulmonary toxicity. The phenomenon described provides one possible explanation; however, more rigorous investigation is needed to define the exact biological machinery associated with this clinical condition.