Introduction
Ovarian cancer is a frequently detected cancer in women, and about 90% of ovarian cancers are epithelial ovarian cancer (EOC). Epithelial ovarian cancer is a very malignant cancer in the female reproductive system and is associated with very high morbidity and mortality rates [1]. Most of these cancers are diagnosed at very advanced stages, and treatment at these stages becomes almost impossible. The 5-year survival rate is only 30% for EOC. The main treatment options for EOC include surgery followed by chemotherapy [2]. However, early metastasis, late diagnosis, limited treatment options, drug resistance and the side effects associated with the available drugs are some of the major concerns in the treatment of EOCs. Consistent with these issues, there is a pressing need to explore more efficient, effective and viable treatment options for EOC.
Natural products have long been considered to be important sources of anticancer drugs [3]. Epidemiological studies have revealed that high dietary intake of naturally occurring plant secondary metabolites, such as flavonoids, through vegetables, fruits and beverages, are associated with lower risks of cancer. This is further justified by several in vitro and in vivo studies [4]. Nonetheless, it has been reported that out of the 155 small-molecule anticancer drugs, the number of drugs that are natural products and/or synthesized from natural products accounts for 72.9% [5]. Among natural sources, plants represent an important source of anticancer drugs; and several plant-derived compounds are currently used as anticancer drugs or are being assessed in clinical trials [6–8].
Sugiol is a diterpene and has been isolated from several species of the genus Salvia [9, 10]. Previously it has been reported that sugiol exhibits anticancer activity against a range of cancer types such as colon, breast and lung cancer [11]. Therefore, in the present study we determined the anticancer activity of sugiol against breast cancer cells. The mechanism for the anticancer activity of sugiol has not been explicitly explored and RAF/mitogen activated protein (MAP)/extracellular signal-regulated kinase (ERK) (MEK)/ERK (RAF/MEK/ERK) signalling pathways have been reported to play important roles in the tumourigenesis and progression of several cancers including ovarian cancer [12]. Therefore, we also investigated the effect of sugiol on the RAF/MEK/ERK signalling pathway.
Material and methods
Chemicals, reagents and cell cultures
Sugiol and other chemicals were of reagent grade and purchased from Sigma Chemical Co. (St. Louis, Missouri, USA) unless otherwise mentioned. The normal human ovarian epithelial cell line (Moody) and the human ovarian tumour cell line (SKOV3) were obtained from Type Culture Collection of Chinese Academy of Sciences, Shanghai, China. The cells were cultured in RPMI-1640 medium containing 10% fetal bovine serum, 100 U/ml penicillin and 100 μg/ml streptomycin and maintained in a humidified atmosphere containing 5% CO2.
CCK8 assay for assessment of cell viability
The cell viability of the normal ovarian cells (Moody) and the ovarian cancer cells (SKOV3) was determined by CCK8 assy. In brief, 5 × 103 cells were seeded on a 96-well plate and then kept at 37°C for incubation in a humidified, 5% CO2 atmosphere. After overnight incubation the cells were treated with different concentrations of sugiol (0–200 μM) for 24 h. Then, 10 μl of CCK8 was added to each well and again incubated at 37°C for 1 h. The optical density at OD450 nm was determined by a microplate spectrophotometer (BioRad, Segrate, Italy). The cell viability was presented as percentage of the control.
AO/EB, DAPI, annexin V/PI staining
SKOV3 cells were seeded at the density of 2 × 105 cells/well in 6-well plates and administered with different concentrations of Sugiol for 24 h. The cells were then stained with a mixture of AO and EB. Stained cells were examined under the fluorescent microscope. DAPI staining was carried by incubating the cells in 6-well plates for 20 min in the dark and treated with different concentrations of sugiol. The cells were then washed with PBS, fixed in formaldehyde (10%) and then again washed with PBS. The DAPI stained cells were then examined by fluorescence microscope. For annexin V/PI a similar procedure as that of DAPI was followed except the cells were stained with annexin V/PI and investigated by flow cytometry.
Determination of mitochondrial membrane potential (MMP)
SKOV3 cells were seeded at a density of 2 × 105 cells/well in a 6-well plate and incubated for 24 h and treated with 0, 12.5, 25 and 50 µM sugiol for 48 h at 37°C in 5% CO2 and 95% air. Then cells from all samples were collected, washed twice with PBS and re-suspended in 500 μl of DiOC6 (1 μmol/l) for MMP at 37°C in a dark room for 30 min. The samples were then examined instantly using a flow cytometer as described previously in the literature.
Cell cycle analysis
The cell cycle analysis was carried out by flow cytometry after staining with propidium iodide. Briefly, SKOV3 cells were plated at the density of 2 × 105 and administered with different concentrations of sugiol (0, 12.5, 25 and 50 μM) and the cells were harvested and kept in ethanol (70%) at 4°C. After overnight incubation the cells were collected again by centrifugation at 1000 rpm for 10 min and washed with PBS and finally suspended in 250 ml of PBS and treated with RNase for 20 min. Afterwards the cells were stained with propidium iodide and the cell cycle analysis was carried out by flow cytometry.
Cell migration assay
Cell migration of SKOV3 cells was determined by wound healing assay. Briefly, cells were cultured until confluence. Then the SKOV3 wells were treated with 0, 12.5, 25 and 50 μM (concentrations lower than IC50 because doses higher than IC50 exhibit considerable cytotoxic effects) of sugiol and the cells were scratched with a sterile pipette tip and then incubated for 24 h. The wound healing capacity of sugiol-treated cells was determined by comparing the wound length with that of the untreated control cells.
Protein expression by western blotting
Total protein from untreated and sugiol-treated cells was isolated in RIPA lysis buffer. Equal protein extracts from each group were run on SDS PAGE and then transferred to a polyvinylidene fluoride membrane. This was followed by blocking with 5% non-fat milk and incubation at room temperature for 1 h. Then the membranes were incubated with a specific primary antibody at 4°C overnight. This was followed by washing in washing buffer and incubation for 1 h with a suitable secondary antibody. The protein bands of interest were visualised by an ECL Advanced Western Blot Detection Kit (GE Healthcare, Uppsala, Sweden).
Statistical analysis
Data were expressed as mean of the replicates ± SD. Statistical significance and IC50 values were analyzed by GraphPad Prism Demo, Version 5 (GraphPad Software, San Diego, California, USA). *P < 0.05, **p < 0.01, ***p < 0.001 were considered as indicating a significant difference.
Results
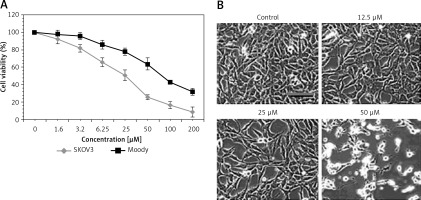
Sugiol exerts antiproliferative effects on SKOV3 cells
The antiproliferative effect of sugiol against human ovarian SKOV3 cancer cells and human normal Moody ovarian cells was evaluated by CCK-8 assay. The results indicated that sugiol exerted dose-dependent antiproliferative effects on SKOV3. However, sugiol was found to be less cytotoxic against the normal cells (Figure 1 A). The IC50 of sugiol against SKOV3 cells was found to be 25 μM as compared to the IC50 of 62.5 μM against the normal ovarian cells. Moreover, sugiol also influenced the cell morphology of SKOV3 cells (Figure 1 B). As the concentration of sugiol was increased in the SKOV3 cancer cells they turned round, contracted and separated from the substratum (Figure 1 B), which are the chief morphological alternations associated with apoptosis.
Sugiol triggers mitochondrial apoptosis
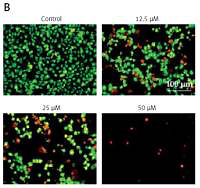
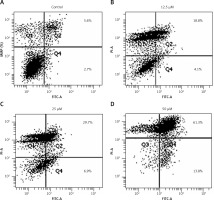
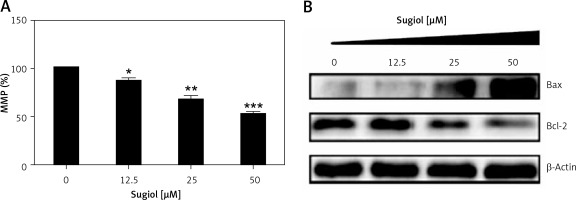
Since sugiol induced morphological changes in SKOV3 cells that are characteristic of apoptosis, we carried out DAPI and AO/EB staining. The results indicated that sugiol induced apoptosis in SKOV3 cells, as evident from the increasing number of white nuclei in the case of DAPI staining (Figure 2 A) and orange fluorescence in the case AO/EB staining (Figure 2 B). To estimate the apoptotic cell populations, we carried out annexin V/IP staining, and it was observed that apoptotic cells increased with increase in the concentration of sugiol (Figure 3). The apoptotic cell populations were found to be 5.6, 18.8, 29.7 and 61.3% at the concentrations of 0, 12.5, 25 and 50 μM of sugiol. Next, we determined the effect of different concentrations of sugiol on MMP. The results indicated that sugiol treatment decreased the MMP of SKOV3 cells in a dose-dependent manner (Figure 4 A). The inhibition of cell migration was also associated with the concomitant upregulation of Bax and downregulation of Bcl-2 expression (Figure 4 B).
Figure 2
Sugiol induces apoptosis in SKOV3 cells as depicted by DAPI staining (A), AO/EB staining (B). The experiments were carried out in triplicate
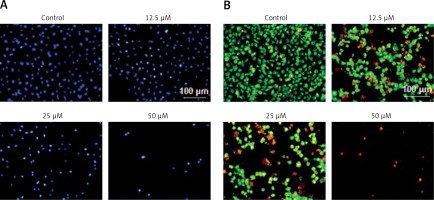
Figure 3
Effect of indicated concentrations of sugiol on (A) mitochondrial membrane potential (MMP) as determined by flow cytometry (n = 9), (B) expression of Bax and Bcl-2 by western blotting
All experiments were carried out three times in triplicate (n = 9) and expressed as mean ± SD. The results were considered significant at *p < 0.01, **p < 0.001 and ***p < 0.0001.
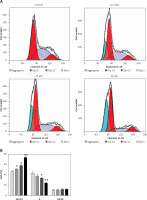
Sugiol induces G0/G1 cell cycle arrest
Cell cycle arrest is an important mechanism by which anticancer agents exert their inhibitory effects. Therefore we also determined the effect of sugiol on cell cycle phase distribution of sugiol-treated SKOV3 cells (Figures 5 A, B). Our results indicated that the percentage of SKOV3 cells was considerably increased in G0 at the concentrations of 0 to 50 µM of sugiol, causing G0/G1 cell cycle phase arrest. Additionally the populations of SKOV3 cells in G0 phase were marginally increased at a dose of 12.5 µM, reasonably increased at 25 µM, and dramatically increased at 50 µM, indicating a dose-dependent effect of sugiol.
Sugiol inhibits cell migration
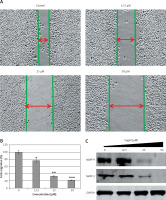
We also examined whether sugiol could inhibit the migration of SKOV3 cancer cells at different concentrations by wound healing assay. The results of wound healing assay showed that sugiol reduced the migratory capability of SKOV3 cells in a dose-dependent manner. While as in control the cells showed fairly good capacity to migrate, in treatment, the cells showed less potential to migrate, as depicted in Figures 6 A and B. Additionally, we observed that sugiol caused a reduction in the expression of MMP-9 and MMP-2 in a dose-dependent manner (Figure 6 B).
Figure 6
Effect of indicated concentrations of sugiol on cell migrations (A) wound healing assay, (B) estimation of % cell migration, (C) expression of MMP-9 and MMP-2
The experiments were carried out three times in triplicate (n = 9). The results were considered significant at *p < 0.01, **p < 0.001 and ***p < 0.0001.
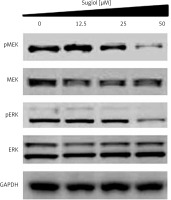
Sugiol blocks RAF/MEK/ERK signalling pathway
The RAF/MEK/ERK signalling pathway has been reported to play important roles in the tumourigenesis and progression of several cancers including ovarian cancer. Therefore, we evaluated the effect of varied concentrations of sugiol on RAF/MEK/ERK cascade. The results indicated that sugiol inhibited the phosphorylation of MEK and ERK in a dose-dependent manner (Figure 7).
Discussion
Ovarian cancer is one of the lethal cancers detected in women. It is often referred to as the silent killer as it remains symptomless and undetected until the advanced stage, at which it becomes almost impossible to treat [13]. Besides late diagnosis, poor availability of suitable drugs, rapid metastasis, development of drug resistance and toxicities associated with the chemotherapy constitute the main hurdles in the treatment of ovarian cancers [14]. Therefore there is an imperative need to explore new and viable drug options. Sugiol is a diterpene and has been reported to possess tremendous pharmacological potential [11]. In the present study we evaluated the anticancer effects of sugiol against ovarian cancer cells. The results indicated that sugiol significantly inhibited the growth of SKOV3 cells (IC50; 25 μM). However, sugiol showed less cytotoxicity towards normal ovarian cells (IC50; 62.5 μM), indicating that sugiol targets cancer cells more potently. Our results are in agreement with previous studies in which sugiol was reported to inhibit the growth of several types of cancers including prostate cancer [11, 15–17]. Moreover, sugiol treatment triggered several morphological changes in SKOV3 cells that are characteristic of apoptosis. Taking cue from this, we carried out DAPI and AO/EB staining to examine whether sugiol induces apoptosis in SKOV3 cells. Interestingly, we observed that sugiol increased for the DNA fragmentation as observed by DAPI and led to an increase in the orange fluorescence as observed by AO/EB staining indicative of apoptosis. These results were further substantiated by the results of annexin V/IP staining which showed that sugiol caused a significant increase in the apoptotic cell populations of SKOV3 cells. Our results are further supported by previous studies in which sugiol was reported to induce apoptosis in cancer cells [18]. To determine whether the apoptosis follows the mitochondrial route we determined the effect of sugiol on MMP, and it was observed that sugiol significantly decreased the mitochondrial membrane potential, which was associated with an increase in the Bax/Bcl-2 ratio. It has been reported that drugs with apoptosis-inducing properties exhibit the potential to minimize potential drug resistance [19], and our results clearly indicated that sugiol has apoptosis-inducting properties, suggesting that sugiol may prove an important lead molecule for the treatment of ovarian cancer. Besides apoptosis, cell cycle arrest is another mechanism by which anticancer agents exert their effects. Our results revealed that sugiol caused G0/G1 cell cycle arrest. Apoptotic cell death is triggered when explicit checkpoints are arrested during cell division [20]. Consistent with this, several anticancer agents cause cell cycle arrest and have been found to be clinically effective for cancer treatment [20]. Cell migration is the key feature of cancer progression and metastasis [21, 22] and suppression of cell migration may prove essential in inhibition of metastasis in vivo. This may ensure longer survival of patients. In our study we observed that sugiol inhibited the migration of SKOV3 cells, which was associated with a concomitant decrease in the expression of MM-2/9. Mitogen-activated protein kinase (MAPK) cascades are key signalling pathways involved in the regulation of normal cell proliferation, survival and differentiation. Aberrant regulation of MAPK cascades contributes to cancer and other human diseases [23]. Therefore, we determined the effect of sugiol on the RAF/MEK/ERK pathway, and it was observed that sugiol could inhibit the phosphorylation of MEK and ERK, indicating that sugiol may prove a potential candidate to target this pathway. These results indicate that sugiol has potent anticancer activity. However, evaluation of sugiol against more cell lines and under in vivo conditions will enable better understanding of sugiol-induced anti-proliferative effects on ovarian cancer.
In conclusion, sugiol exerts potent anticancer effects on SKOV3 cells. These anticancer effects are attributed to the cell cycle arrest, apoptosis and inhibition of the RAF/MEK/ERK pathway. Moreover, this study opens the way for further evaluation of sugiol under in vivo conditions.