Introduction
Cardiovascular diseases (CVD) constitute the leading cause of death in patients with chronic kidney disease (CKD) [1]. Kidney transplantation (KTx) is associated with improvement in mortality due to CVD, but the risk is still high [2]. Pathological changes in vascular structure and function in CKD patients are the risk factors of CVD [3]. Endothelial dysfunction (ED) has been shown to independently predict a worse long-term outcome in many study groups [4]. The main functions of endothelium are the control of vascular tone, coagulation, fibrinolysis and immune response. Inflammation, oxidative stress, dyslipidemia, hyperglycemia, hypertension and uremic toxins are the potential causes of its dysfunction in patients with CKD [5]. There is evidence to show that the endothelial function improves following KTx [6, 7], but the risk of ED still exists due to the effect of immunosuppressive drugs, metabolic complications and partly because of suboptimal graft function [8].
A measurement of endothelium dependent dilation (flow-mediated dilation – FMD) is the most commonly used test for assessing endothelial function [9]. The dilation response with increased blood flow following transient ischemia is mainly mediated by nitric oxide (NO) released from endothelial cells and impaired FMD is widely used as a clinical marker of ED. An exogenous NO donor such as nitroglycerin (NTG) can be used to determine endothelium-independent vasodilation (NTG-mediated dilation NMD) and to serve as a measure of maximum obtainable vasodilation response, reflecting vascular smooth muscle function [10].
The aim of this study was to evaluate the endothelial-dependent and -independent arterial vessels’ reactivity in patients in the long term after KTx and compare it with age-related healthy people.
Material and methods
Two groups of subjects were examined: 94 healthy individuals (51 males, 43 females) and 36 consecutive non-smoking, Caucasian KTx recipients from a deceased donor (15 females; 21 males) with stable graft function, transplanted between 2010 and 2013 and followed up in our transplant outpatient clinic. The immunosuppressive regimen consisted of steroids with a mean dose of prednisone 5.0 ±2.5 mg/24 h, mycophenolate mofetil or sodium and tacrolimus (TAC). All patients were treated with antihypertensive drugs and with statins due to hyperlipidemia. Patients with atherosclerotic disease, congestive heart failure, uncontrolled hypertension, diabetes mellitus, with active inflammation and treated with nitrates or renin-angiotensin-aldosterone inhibitors were excluded from the analysis.
The information concerning patients’ medical history and medications, including immunosuppressive therapy, was obtained from medical charts. Biochemical parameters of the study group (KTx patients) such as serum creatinine, glucose, cholesterol, triglycerides and high-density lipoptotein (HDL) cholesterol were taken in a fasting state in the morning of the day of routine ambulatory transplant visit and were measured with routine laboratory methods. The estimated glomerular filtration rate (eGFR) was calculated by an abbreviated Modification of Diet in Renal Disease (MDRD) formula, and low-density lipoprotein (LDL) cholesterol was calculated using the Friedewald formula. In healthy people the same biochemical tests were measured on the day of ultrasonographic examination and eGFR was calculated. Systolic and diastolic blood pressure were measured on the arm without active or inactive arteriovenous fistula in an upright sitting position after at least 5 min rest using a mercury sphygmomanometer with appropriate cuff size. Two readings were recorded for each individual. The average of two readings was defined as the subject’s blood pressure.
High-resolution ultrasonography
Participants were examined in a fasting state, on the same day as the outpatient clinic visit, after taking blood pressure and blood samples, 12 h (or 24 h in the case of using once-daily prolonged release TAC formulation) after the last dose of calcineurin inhibitors in supine position, in a quiet, temperature-controlled (22–24°C) room, after at least 10 min of rest. All subjects were asked to refrain from using alcohol, non-steroidal anti-inflammatory drugs, nicotine or caffeine for 24 h before examination. β-blockers, diuretics and calcium channel blockers were withheld for 24 h before examination.
Endothelial-dependent and -independent vasodilation were assessed by high-resolution ultrasound (Vivid 7, General Electric Vingmed) with a 12 MHz linear transducer (M12L, General Electric Vingmed), according to recent guidelines [10]. The brachial artery in the nonfistula upper limb was imaged 5–10 cm above the antecubital fossa in the longitudinal plane. The transducer remained in a fixed position relative to the patient’s arm throughout the procedure. All measurements of artery diameter were taken with respect to the cardiac cycle, at the onset of the R wave (end of diastole) during simultaneous ECG recording. Media-adventitia interfaces on the near (anterior) and far (posterior) walls marked the boundaries of the artery diameter. The mean value of three consecutive measurements was calculated at each time point.
After obtaining baseline images of the brachial artery, transient ischemia for increased blood flow was induced by inflation of a blood pressure cuff around the forearm to 200 mm Hg for 5 min. The change in artery diameter was assessed 60 s after deflation of the cuff. Thereafter, a 15 min period was allowed for recovery of the vessel, after which additional baseline images of the brachial artery were obtained. Next, the artery dilation response after a sublingual dose of NTG spray (0.4 mg) was used as a measure of endothelium-independent vasodilatory capacity. The images used for measurement of the artery diameter were obtained at 60, 90, 120, 150 and 180 s of the test and the average of the maximum artery vasodilation observed in these five measurements was taken for analysis. The response of the vessel diameter to reactive hyperemia (FMD) and NMD was expressed as the percent change relative to the diameter immediately before cuff inflation and to the diameter immediately before drug administration, respectively. Measurements were made manually offline on a workstation by one blinded investigator. The intra-observer measurements’ reproducibility was 95%. A schematic representation of the tests used to assess the artery dilatation in the study (FMD and NMD) is presented in Table I.
Table I
Schematic representation of the arterial vessel reactivity tests: flow-mediated dilation (FMD) and endothelium-independent vasodilation (nitroglycerine-mediated dilation – NMD) used to assess artery dilatation in the study
| Autakoid potential | |||||
|---|---|---|---|---|---|
| HIGH | LOW | ||||
| Dialation potential | HIGH | NMD | o → O | NMD | o → O |
| FMD | o → O | FMD | o → o | ||
| LOW | NMD | O → O | NMD | O → O | |
| FMD | O → O | FMD | O → O | ||
Written informed consent was obtained from all subjects before entering the study, and the study protocol was approved by the local Ethics Committee.
Statistical analysis
All values are expressed as mean ± SD or SEM. Data were analyzed by the Shapiro-Wilk test to determine the distribution. Statistical analysis was performed using Student’s t-test or the Mann-Whitney test for continuous variables and using the χ2 test for non-continuous variables. The level of statistical significance was set at p < 0.05.
Results
The demographic and biochemical characteristics of the KTx patients and control group are summarized in Table II. The KTx patients were mean 5.1 ±2.3 years after transplantation and mean duration of hemodialysis before transplantation was 3.1 ±1.2 years. The mean eGFR of study patients was significantly lower than in the control group. Patients had good control of blood pressure and serum lipids (Table II).
Table II
Demographic and biochemical characteristics of KTx patients and healthy people
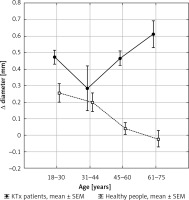
The mean vessel diameter before tests was significantly larger in healthy people than in KTx patients, mainly in patients over 45 years old. The vasodilative effect during transient ischemia was significantly less pronounced in KTx patients than in healthy people; only in patients younger than 30 years was it comparable (Table III). The ischemic reaction was age-related in both groups, but in KTx patients it was clearly weaker than in healthy people (Figure 1). In the group of the youngest healthy volunteers the ischemic test caused dilation of the brachial artery by an average of 8.4 ±4.4%, while lower values were recorded in the group of KTx patients (3.9 ±4.7%); the difference was not statistically significant due to pronounced dispersion of results in KTx patients. In the following age groups the disappearance of the ischemic test’s vasodilation effect was observed, which was more pronounced in KTx recipients (Figure 1, Table III). The data of the FMD test from 2 patients (one from the 46–60 age group and the other from over 60 years) were not included in the analysis because of a marked pathological reaction (vasoconstriction), different than in other patients.
Table III
Basal vessel diameter and the changes of diameter in endothelial-dependent test (flow-mediated dilation, FMD – ischemic test) and endothelium-independent test (nitroglycerine-mediated dilatation NMD) in KTx patients and in healthy people
Figure 1
Vasodilative reaction of the artery wall in response to transient ischemia (flow-mediated dilation, FMD) in KTx patients and in healthy people depending on age
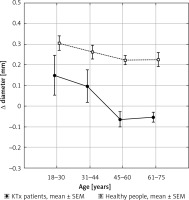
The dilation of the brachial artery after a dose of NTG in KTx recipients was comparable to that in healthy people, and a decrease of artery wall reactivity in the NMD test in all study subjects from both groups was observed in relation to the subjects’ age. In the successive age groups gradual weakening of the vasodilation effect was observed (Table III). The time of the vasodilation reaction after the NO donor was delayed with the age of the subject and positively correlated with age in all study subjects, but statistically significantly only in healthy people. The reaction in KTx patients was preserved but slightly weaker, and the curve ran flatter (Figure 2). The changes in the KTx group were more varied, resulting in a poorer correlation with the group of volunteers (Figure 2).
Figure 2
Time of occurrence of vasodilation reaction after one dose of nitroglycerin as a determinant of endothelium-independent vasodilation (nitroglycerine-mediated dilation, NMD) in KTx patients and in healthy people depending on age
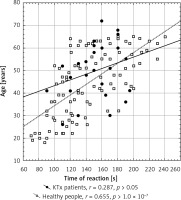
The relationship between the results of both tests are shown after subtraction of the results of the FMD and the NMD. The data obtained in the two groups divided into the age ranges adopted are presented in Figure 3. The differences between the healthy group and the KTx patients are most pronounced in subjects above 40 years of age. Figure 3 illustrates that in healthy people the endothelial-dependent and -independent reactions get weaker with age but both are preserved. In KTx recipients FMD is clearly weaker in each age range in comparison with the healthy group with the NMD reaction preserved.
Discussion
There is increasing evidence to show that kidney transplantation improves arterial function (endothelial-dependent and independent dilation) in CKD patients [6, 7]. In our study we assessed the vasodilatory function in patients in the long term after KTx and compared it with age-related healthy people. Our study showed that patients in the long term after KTx have more pronounced endothelial dysfunction in comparison with age-related healthy people. The endothelial independent vasodilation was not worse. This phenomenon may be explained by the fact that patients after KTx are still exposed to endothelial damage factors such as inflammation, oxidative stress, dyslipidemia, hypertension or hyperglycemia. The treatment with immunosuppressant agents is associated with additional harmful factors for the cardiovascular system. Most KTx patients have impaired kidney function with eGFR < 60 ml/min/1.72 m2. Furthermore, all of our KTx patients had a history of long kidney failure before kidney replacement therapy (approximately 3.2 years of hemodialysis before KTx). Previous studies showed improvement of FMD after KTX [11–13]. Sharma et al. observed an improvement of FMD from 9.1% to 15.7%, 3 months after KTx in 60 Indian patients. Impaired FMD was present in 26.8% in pre-transplant CKD patients and only in 3.3% after transplantation [14]. Similarly, Yilmaz et al. observed significant improvement of FMD among 161 chronic hemodialysis patients at 6 months after transplantation [15]. It thus indicates that KTx reverses endothelial dysfunction present in end-stage renal disease patients. On the other hand, Hornum et al. did not observe changes of FMD among 40 patients 1 year after KTx, but they noted a significant improvement of NTG-induced vasodilation [6]. The improvement of NMD was observed in the study by Yilmaz et al. [15], but not by Sharma et al. [14]. Maybe the changes of endothelial independent vasodilation require longer time after KTx. This hypothesis would be supported by our findings; we observed approximately 5 years after transplantation NMD values in KTx patients comparable to those observed in age-related healthy people. The endothelium-independent vasodilation in our KTx group was similar to that in healthy volunteers and the vasodilation potential disappeared with age like in healthy people. This would agree with the known effect of nitrates, which decrease the vasomotor tone of peripheral resistive conduit arteries. Therefore, it could be possible that transplantation has a positive effect on physical properties of this part of the circulatory network. The comparable vasodilatory effect of NTG on the vascular wall in healthy and KTx subjects may account for inhibition of adverse developments in the arterial wall connected with uremic toxemia such as intimal and medial calcification and highlight a potential link between improvement of CKD-mineral bone disease and reduction in CVD risk in patients after KTx. Despite improvement in the vasodilatory effect after KTx reported by other authors, we found that the values continue to be lower when compared to healthy people and a state of persistent endothelial dysfunction may contribute to an increased CVD risk in this population.
The limitation of our study is the small sample size with quite large age distribution among KTx patients resulting in the statistical power that does not allow for clinically relevant conclusions to be drawn. Our study is cross sectional and we assessed FMD and NMD only once without any observational period, and the values of the parameters before transplantation were unknown; therefore we were not able to assess whether they improved after KTx. Furthermore, we compared KTx findings with healthy people, not treated for hyperlipidemia or hypertension. The control group had better eGFR, significantly lower BMI, and lower diastolic and systolic blood pressure than the study population. On the other hand, some drugs such as statins or calcium channel blockers may improve endothelial function [16, 17]. Our KTx population was quite homogeneous; only patients with well-controlled hypertension were included in the study, all of them were treated with calcium channel blockers, none with renin-angiotensin-aldosterone inhibitors and all were treated with statins due to hyperlipidemia. Recent studies have confirmed the beneficial role of statins on FMD [18]. Only patients treated with TAC as part of an immunosuppressive scheme were included in the study to avoid the differences of cyclosporine and tacrolimus impact on the endothelium function [19, 20]. All of our patients were previously hemodialyzed and received a graft from a cadaveric donor.
In conclusion, both endothelial-dependent and -independent arterial wall reactions decrease with age. The endothelial-dependent vascular response is more attenuated in KTx patients than the independent one. The endothelium-independent vasodilation in KTx patients is comparable with that in healthy people.