Current issue
Archive
Manuscripts accepted
About the Journal
Editorial office
Editorial board
Abstracting and indexing
Subscription
Contact
Ethical standards and procedures
Most read articles
Instructions for authors
Article Processing Charge (APC)
Regulations of paying article processing charge (APC)
NEUROSURGERY / CLINICAL RESEARCH
Association between oxidative stress biomarkers and concentrations of some metal ions in the blood of patients with brain tumors and hydrocephalus
1
Department of Surgery, Faculty of Medicine, University of Belgrade, Belgrade, Serbia
2
Clinical Center of Serbia, Neurosurgery Division, Belgrade, Serbia
3
Department of Physiology, Institute for Biological Research “Siniša Stanković”, University of Belgrade, Belgrade, Serbia
4
Faculty of Chemistry, University of Belgrade, Belgrade, Serbia
Submission date: 2018-03-29
Final revision date: 2018-06-14
Acceptance date: 2018-06-28
Online publication date: 2019-08-22
Publication date: 2020-05-26
Arch Med Sci 2020;16(4):811-819
KEYWORDS
TOPICS
ABSTRACT
Introduction:
Any substance that induces production of free radicals can be a potential cause of brain damage. The aim of our study was to investigate the relationship between some metal ions and oxidative stress biomarkers in the blood of patients with brain tumor and hydrocephalus.
Material and methods:
Our study included 27 control subjects, 24 patients with brain tumor and 21 patients with hydrocephalus. The activities of superoxide dismutase (CuZn SOD), catalase (CAT), glutathione peroxidase (GSH-Px), glutathione reductase (GR), glutathione S-transferase (GST) and acetyl cholinesterase (AChE), as well as concentrations of reduced glutathione (GSH), lipid peroxides (TBARS) and sulfhydryl groups (SH) were analyzed in the plasma and red blood cells (RBCs) of patients. We also determined the concentrations of Mn, Ni, Co, Cu, Zn, As, Se, Cd, Hg and Fe.
Results:
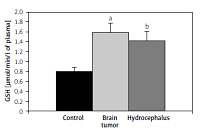
The higher activity of SOD and concentration of GSH in both investigated groups could indicate higher oxidative stress. We also observed decreased levels of SH groups in both groups of patients. In both groups of patients we detected decreased concentrations of Ni, Co, Zn and Fe (and Mn in brain tumor patients) and increased concentrations of As, Se and Cd in the blood. Interestingly, we observed a higher concentration of Cd in both plasma and RBCs of hydrocephalus patients compared to the patients with brain tumor.
Conclusions:
There are strong correlations between some metal ion concentrations and certain oxidative stress biomarkers in the blood of patients, which supports our hypothesis, but the observed trend needs to be further investigated.
Any substance that induces production of free radicals can be a potential cause of brain damage. The aim of our study was to investigate the relationship between some metal ions and oxidative stress biomarkers in the blood of patients with brain tumor and hydrocephalus.
Material and methods:
Our study included 27 control subjects, 24 patients with brain tumor and 21 patients with hydrocephalus. The activities of superoxide dismutase (CuZn SOD), catalase (CAT), glutathione peroxidase (GSH-Px), glutathione reductase (GR), glutathione S-transferase (GST) and acetyl cholinesterase (AChE), as well as concentrations of reduced glutathione (GSH), lipid peroxides (TBARS) and sulfhydryl groups (SH) were analyzed in the plasma and red blood cells (RBCs) of patients. We also determined the concentrations of Mn, Ni, Co, Cu, Zn, As, Se, Cd, Hg and Fe.
Results:
The higher activity of SOD and concentration of GSH in both investigated groups could indicate higher oxidative stress. We also observed decreased levels of SH groups in both groups of patients. In both groups of patients we detected decreased concentrations of Ni, Co, Zn and Fe (and Mn in brain tumor patients) and increased concentrations of As, Se and Cd in the blood. Interestingly, we observed a higher concentration of Cd in both plasma and RBCs of hydrocephalus patients compared to the patients with brain tumor.
Conclusions:
There are strong correlations between some metal ion concentrations and certain oxidative stress biomarkers in the blood of patients, which supports our hypothesis, but the observed trend needs to be further investigated.
Share
RELATED ARTICLE
We process personal data collected when visiting the website. The function of obtaining information about users and their behavior is carried out by voluntarily entered information in forms and saving cookies in end devices. Data, including cookies, are used to provide services, improve the user experience and to analyze the traffic in accordance with the Privacy policy. Data are also collected and processed by Google Analytics tool (more).
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.