Barotrauma has long been known as a complication of mechanical ventilation in the intensive care unit (ICU), and has been associated with prolonged hospitalization, increased morbidity and poorer outcomes [1]. However, this complication has gained renewed prominence during the coronavirus 2019 (COVID-19) pandemic, due to reports of considerably increased incidence amongst COVID-19 patients [2–4]. Manifestations of barotrauma have been noted even in non-mechanically ventilated patients – patients with mild or moderate disease, who are usually considered at lower risk for baro-trauma [3]. The incidence of barotrauma and associated manifestations such as pneumothorax can range from 1% of hospitalised COVID-19 patients to 15% in mechanically ventilated COVID-19 patients [3, 5]. This represents a marked increase in incidence from pre-COVID times, when studies revealed a barotrauma incidence rate of 2.9% in mechanically ventilated patients [1]. A combination of disease-induced lung frailty, raised airway pressures caused by oxygen therapy and persistent coughing have been implicated as possible culprits for this phenomenon [6]. There has also been a sharp increase in the use of ventilation strategies such as high-flow nasal cannula (HFNC) oxygen therapy and non-invasive ventilation (NIV) in an effort to avoid invasive mechanical ventilation, and the resultant morbidity and mortality [7]. Our study aimed to describe the incidence of barotrauma, the affected patient subset, mode of ventilation and outcome of patients developing this complication, and compare these characteristics with those of patients who did not develop barotrauma, in the ICUs of a dedicated COVID-19 care centre in India.
METHODS
The data for the cases and controls in the present study were collected from a prospective patient cohort, approved by the institutional ethics committee (IEC-291/17.04.2020). As this was a retrospective observational study, the requirement for written informed consent was waived. The study included all adult patients with confirmed COVID-19 infection, admitted between October 1st and December 31st 2020 in the four ICUs of our institution, a tertiary level dedicated COVID hospital. Cases were defined as those patients amongst the studied cohort who showed evidence of a ‘barotrauma event’ (defined as presence of at least one of pneumothorax [PTX], pneumomediastinum [PM], subcutaneous emphysema [SCE] or bronchopleural fistula [BPF]). PTX was defined as the presence of air in the pleural space, PM as air in the mediastinal space, SCE as air in the skin or subcutaneous tissue and BPF as an abnormal connection between the bronchi and pleural space. Diagnosis was made by an experienced clinician based on either clinical examination or imaging (chest radiography, computed tomography [CT] or ultrasonography) or a combination of both. The control group was constituted from the remaining patients in the studied cohort, i.e. COVID-19 patients admitted to the same ICUs during the same period, who did not develop barotrauma. Each case was matched to a patient belonging to the same gender and age range (age of case ±5 years) from amongst the non-barotrauma patients. If multiple possible matches were found for a case, one was selected randomly by an independent data gatherer unaware of the associated risk factors. Thus a 1 : 1 matched case-control cohort was created.
Details of both groups were noted, including age, gender, comorbidities, duration of ICU stay, patient outcome (death/discharge), mode of ventilation (oxygen by face mask [FM], high flow nasal cannula [HFNC], non-invasive ventilation via biphasic positive airway pressure [NIV] or invasive mechanical ventilation [IMV]), and duration spent on each mode. If the patient had been on multiple modes of oxygen therapy during ICU stay, the mode applied for the longest time was defined as the predominant mode. In the barotrauma patient group, additional details noted included the day of development of the barotrauma event, nature of the complication (PTX/PM/SCE/BPF), mode of oxygen therapy and fraction of inspired oxygen (FiO2) at the time of the event, as well as insertion of an intercostal drain. If the patient developed more than one form of barotrauma, the day of the earliest event was noted as the day of the event. Information was abstracted from the prospective cohort on a standard form by two authors, transferred to a Microsoft Excel sheet and analysed subsequently using R Software.
The continuous variables were presented as mean with standard deviation or median with interquartile range, while the categorical variables were presented as frequency and percentages. The Mann-Whitney U-test was applied for analysis of continuous variables and the χ2 test and Fisher exact test for categorical variables. A P-value < 0.05 was considered statistically significant.
RESULTS
A total of 827 patients were admitted in the ICUs of our institution during the three-month study period (Figure 1). Of these, 30 patients (3.6%) developed barotrauma events. We observed an overall ICU mechanical ventilation rate of 41%, HFNC rate of 20.5%, NIV rate of 10.2% and FM rate of 27.9% during the study period. This reflected that 3.24% of all mechanically ventilated patients, and 3.89% of non-mechanically ventilated patients, in our ICUs developed a barotrauma event. Of these 30 patients, most patients were male (22/30, 73.3%), and middle-aged (median age 55.5 years; IQR 28.3). The distribution of comorbidities amongst the baro-trauma and non-barotrauma groups was similar (Table 1). The most common systemic comorbidity within the barotrauma group was diabetes (13/30), followed by hypertension (11/30).
FIGURE 1
Flowchart showing cases, controls and outcomes
ICU – intensive care unit, PTX – pneumothorax, SCE – subcutaneous emphysema, PM – pneumomediastinum, BPF – bronchopleural fistula
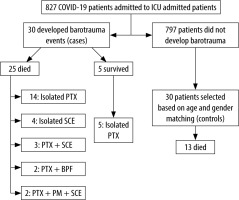
TABLE 1
Demographic and clinical characteristics of the two groups
The mortality rate amongst the patients with barotrauma was nearly double that of the control group (83.3% [25/30] in the barotrauma group vs. 43.3% [13/30] in the non-barotrauma group, P < 0.001). The overall all-cause mortality rate amongst ICU patients in the study period was 36.5% (302/827). We noted that odds of survival decreased by 85% if barotrauma occurred during the clinical course (OR 0.15; 95% CI: 0.46–0.51).
The predominant modes of oxygen therapy used during the ICU stay were comparable between the two groups (Table 2). However, a comparison of the duration spent on each mode revealed significantly longer time spent on HFNC (6.7 ± 9.46 vs. 1.73 ± 3.31 days, P = 0.04), and IMV (9.54 ± 9.71 vs. 0.867 ± 3.06 days, P < 0.001) amongst patients who developed barotrauma than those who did not. We found that patients with barotrauma spent nearly a week longer in the ICU (median ICU stay duration 15.5 days [IQR 9–26] in the barotrauma group vs. 9 days [IQR 7–18] in the control group, P = 0.014).
TABLE 2
Predominant mode of oxygen therapy in the two groups
| Predominant mode of oxygen therapy | Patients with barotrauma (n = 30) | Patients without barotrauma (n = 30) |
|---|---|---|
| FM | 5 | 2 |
| NRBM | 0 | 4 |
| HFNC | 8 | 10 |
| NIV | 6 | 3 |
| IMV | 11 | 11 |
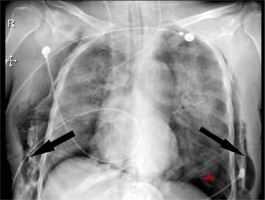
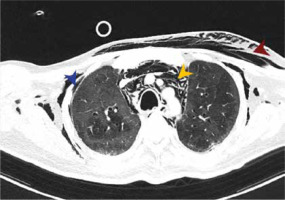
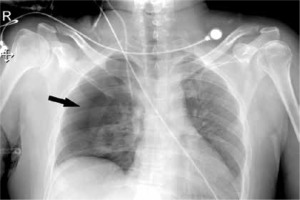
We observed 38 barotrauma events in the 30 patients (Figure 1). The most common event was PTX (Figure 2), seen in 26 out of 30 patients (86.6%). Nearly one-third of the patients had SCE (9/30), while seven patients had multiple events (Figure 3), including two patients who had a combination of PTX, PM and SCE (Figure 4). Intercostal drains (ICDs) were inserted in 24 of the 30 patients (80%), while the rest were managed conservatively. Two-thirds of patients (20/30) in this group developed barotrauma while on IMV (Table 3). The mean FiO2 at the time of the barotrauma event was 66.9% ± 19%. Preexisting conditions affecting the respiratory system were seen in over a third (11/30) of the barotrauma patients, which was statistically comparable to the non-barotrauma group (5/30, P = 0.80). These conditions included bronchial asthma, chronic obstructive pulmonary disease, interstitial lung disease, post-surgical pleural injury, lung malignancy and tuberculosis. The median onset of barotrauma was 9 (IQR 4–13.5) days after ICU admission.
TABLE 3
Mode of oxygen therapy at the time of barotrauma (n = 30)
| Mode of oxygen | Counts | % of total |
|---|---|---|
| FM | 3 | 10.0 |
| NRBM | 0 | 0 |
| HFNC | 4 | 13.3 |
| NIV | 3 | 10.0 |
| IMV | 20 | 66.7 |
FIGURE 2
Image shows right-sided pneumothorax in a 22-year-old male patient on day 4 of ICU stay. He underwent intercostal drain insertion and had a favourable outcome
DISCUSSION
We found that 3.6% (30/827) of admitted patients in the ICU cohort developed barotrauma. Patients with barotrauma were more likely to have a longer ICU stay (median 15.5 vs. 9 days) and higher mortality rate (83.3% vs. 34.2%). Literature reports a baro-trauma incidence rate varying from less than 1% in all hospitalised COVID-19 patients, to values as high as 13.6%, 15% and 17% in mechanically ventilated patients with COVID-19 illness [3–5, 8, 9]. We observed a lower barotrauma incidence rate, which reflects our studied cohort, i.e., ICU patients on all modes of ventilation, not only mechanically ventilated patients.
We observed a higher mortality rate amongst the barotrauma-affected patient subset as compared to the control group (83.3% vs. 34.2% respectively, P < 0.001). We found that occurrence of barotrauma decreased the odds of survival by 85% (OR 0.15; 95% CI: 0.46–0.51). Some authors report an insignificant difference or a modest increase in mortality rates in COVID-19 patients with barotrauma [3, 5]. On the other hand, Abdallat et al. [10] found a mortality rate of 64% in COVID-19 patients on IMV with barotrauma, compared to 25–50% in non-baro-trauma patients on IMV. The authors hypothesised that the findings reflected a worse disease process and prognosis. Similarly, McGuiness et al. [5] found barotrauma to be an independent risk factor for death in COVID-19 (OR = 2.2; P = 0.03), as well as prolonged hospital stay (OR = 0.92; P < 0.001), which was similar to our findings. We believe that the high mortality rate seen in our patient subset was multifactorial. Firstly, we studied only ICU patients, i.e., patients with more severe disease at admission. This is corroborated by our finding that these patients had a high FiO2 requirement (mean 66.7% ± 19%) at the time of barotrauma. Secondly, the associated use of IMV may be a factor in the high mortality rate. The mortality rate amongst mechanically ventilated patients with barotrauma is known to be significantly higher compared to those without [1]. Similarly, requirement of IMV in COVID-19 patients has been associated with poorer outcomes [11]. Two-thirds of our patients (20/30) developed barotrauma while on IMV, which may be one of the reasons for the high mortality. Thirdly, the presence of multiple barotrauma events may be a factor in the poor outcome. This is supported by our finding that all five patients with favourable outcomes in our study had isolated PTX, while all seven with multiple events died. We hypothesise that the pre-existing severe disease, along with IMV use, and development of multiple barotrauma events, may be responsible for the poor outcomes in our studied patients.
Prolonged ICU stay in patients with barotrauma is also a well-documented trend in the literature [1].
Barotrauma frequently necessitates interventions such as insertion of an ICD, or conservative management for SCE. In addition, barotrauma may cause an acute deterioration in respiratory and oxygenation parameters. These factors may be responsible for additional days of ICU stay. We found a significant association between the development of barotrauma and the duration spent on IMV and HFNC during the course of the ICU stay (9.54 vs. 0.867 days on IMV and 6.71 vs. 1.73 days on HFNC for the barotrauma and non-barotrauma group, respectively). Mechanical ventilation in COVID-19 patients relies on lung-protective ventilation, which includes a low tidal volume, high respiratory rate and high PEEP [12, 13]. It is thought that increasing the PEEP to maintain oxygenation in acute respiratory distress syndrome (ARDS) may be at the expense of increasing airway pressure and a higher risk of barotrauma [10]. While barotrauma in mechanically ventilated patients is well known, the association between the use of HFNC and barotrauma has not been studied sufficiently. HFNC has emerged as an effective mode of oxygen therapy in COVID-19 patients [7]. It is often considered a ‘safer’ mode of ventilation, and may delay the need for IMV [7]. However, HFNC is known to increase the mean airway pressure and has been associated with barotrauma in the paediatric population [14–16]. COVID-19 induced alveolar destruction, exacerbated by steroid use and the resultant transpulmonary pressure swings during mechanical ventilation, in addition to persistent coughing may lead to rupture of peripheral, peripleural alveoli, resulting in barotrauma [6, 17–20].
We found no significant difference in the incidence of pre-existing respiratory conditions between the two groups. Martinelli et al. [3] and Abdallat et al. [10] observed a few patients with respiratory comorbidities in their cohort of patients developing pneumothorax. Lemmers et al. [4] similarly found a lower incidence of chronic obstructive pulmonary disease in their cohort of patients. Thus, it appears that COVID-19 induced lung frailty, rather than pre-existing disease processes, is associated with increased barotrauma, as elaborated above.
The study has some limitations. This was a retrospective observational case control study with a limited sample size, which precludes establishing an association between various studied factors. Additionally, although we selected a hospital cohort as the control group, the possibility of selection bias cannot be ruled out. However, since both groups were sourced from the same base population during the same period, the control group can be considered representative of the population that yielded the cases. Secondly, though the presence of systemic comorbidities was noted, these were not graded according to severity. Thus, a deeper analysis of the impact of these factors on outcome was not possible. Thirdly, there may have been a number of missed barotrauma events, since computed tomographs, which are more sensitive at identifying small pneumothoraxes, were done only when clinically indicated. Finally, since the exact mechanism of barotrauma in COVID-19 is still poorly understood, further research is warranted in this direction.
CONCLUSIONS
Barotrauma in COVID-19 ICU patients is a significant problem. Barotrauma is associated with prolonged ICU stay and considerably higher odds of mortality. There appears to be no correlation between respiratory comorbidities and the presence of barotrauma. Barotrauma is associated with longer duration spent on IMV and HFNC. The association between HFNC and barotrauma is underrecognised, and further studies are required to validate it.