Introduction
The possible role of the immune system in controlling carcinogenesis was first suggested by Paul Ehrlich in 1909 [1]. Of note, at the beginning of the 20th century immunology, as a distinct scientific discipline, did not exist [2] and it was not possible to propose any idea for explanation of immune system-tumor relationships. The subject was not actively followed until the middle of the 1950s when it was evident that humoral acquired elements of the immune system are accompanied by equally important acquired cellular immunity [2, 3].
The early concept of cancer immunosurveillance
The role of the immune system in defense against cancer has been the subject of hot debates since the 1950s. At that time, the importance of cellular components of the immune system in mediating allograft rejection was proven [4, 5]. Moreover, in animal models of inbred strains of mice, immunity against transplantable tumors induced by carcinogens was observed [6]. Soon, antigenic differences between tumors and normal tissues were defined [7]. Based on these facts, the hypothesis of cancer immunosurveillance was proposed by Lewis Thomas and Sir Macfarlane Burnet in the late 1950s [8, 9]. The core of this concept was the statement that “In (…) animals (…) inheritable genetic changes must be common in somatic cells and a proportion of these changes will represent a step towards malignancy. (…) There should be some mechanisms for eliminating (…) dangerous mutant cells. (…) It is postulated that this mechanism is of immunological character” [8]. Not much later, Jacques Miller discovered the essential role of the thymus in development of cellular immunity [10]. He also showed that neonatally thymectomized (immunosuppressed) mice failed to reject allo- and xenogeneic skin and were more susceptible to carcinogen-induced tumor development in comparison to normal mice [11].
The immunosurveillance theory was not generally accepted. In fact, the significance of the immune system in anticancer defense remained unappreciated until the end of the last century. Several observations argued against the hypothesis. For example, nude (athymic, T-cell deficient) mice did not develop more cancers than normal mice [12, 13]. At present, we know that nude mice are not totally immunocompromised: their Tgd cell and NK cell functions are generally intact [14, 15]. Opponents of the hypothesis also showed that in immunoprivileged sites such as the anterior chamber of the eye as well as in the brain no excessive number of tumors was observed. In deeply immunosuppressed patients after renal transplantation, increased incidence of tumors was reported, but most of these tumors had viral etiology (lymphomas, Kaposi sarcomas, and other) [16]. Now, longitudinal observations show that in patients after transplantation, regularly taking immunosuppressive drugs, the incidence of all types of cancer is increased [17]. Opponents of the hypothesis also pointed out that patients with diseases associated with a deficient immune system did not experience increased cancer incidence. Some investigators postulated that components of the immune system could even promote neoplasia and speed up cancer growth. As proof of this they cited acceleration of tumor development in mice injected with tumor-immune serum in the phenomenon of tumor enhancement [18].
By the end of the 20th century, it was generally believed that the immune system protects against virally induced neoplasia but its role in preventing spontaneous cancers was controversial. In the landmark review paper by Hanahan and Weinberg in 2000, discussing the subject of carcinogenesis and tumor development, listing six hallmarks necessary for tumor growth, no mention was made as to the role of the immune system in the process of tumor initiation and progression [19].
At the beginning of the 21st century, the advancement of knowledge about the mechanisms of carcinogenesis led to a renaissance of the concept of cancer immunosurveillance. Renewed interest in this issue resulted from studies demonstrating presence of tumor specific antigens in spontaneously developing tumors in cancer patients [20] and from investigations in animal models showing promotion of tumor development in mice with either an interferon (IFN)-g or IFN-gR deficit [21-23] or in mice lacking perforin [24]. Perforin is an element of T cell and NK cell lytic granules, important for target cell killing, including tumor cells [25, 26]. Facilitated tumor development was also observed in RAG1-/- or RAG2-/- mice [22, 27]. These mice are characterized by a lack of recombination activity genes, which are necessary for production of T, B, and NKT cell receptors for antigens [28].
In 2001, experiments performed in Robert D. Schrei- ber’s group confirmed that immunosuppressed mice are not only more prone to carcinogen-induced tumorigenesis in comparison with immunocompetent mice. The experiments showed that tumors growing in immunocompetent vs. immunodeficient mice are also qualitative different. Tumor cells from primary tumors growing in immunocompetent mice, when injected into naïve wild-type recipients, formed progressively growing tumors in 100% of mice. In contrast, tumor cells from tumors explanted from immunodeficient mice, when injected into naïve wild-type animals, formed progressing tumors in half of mice but in the other half of mice the tumors finally regressed [22]. The investigators concluded that carcinogen-induced tumors from immunocompetent mice were less immunogenic and “more aggressive” in comparison to tumors from immunodeficient mice since the former tumors experienced an “editing” (shaping) process by the intact immune system. In this process, more antigenic nascent transformed cells were eliminated [22, 29].
Cancer immunoediting hypothesis
Based on the above-described investigations, the cancer immunoediting hypothesis was formulated [29, 30]. The hypothesis, at present widely accepted, postulates that the immune system – as a whole – controls not only tumor growth (in fact, quantity) but also tumor quality. The concept proposes that there are three phases of tumor-immune system relationships: elimination, equilibrium, and escape (Fig. 1).
Fig. 1
Relationships between transformed/tumor cells and the protective antitumor response – cancer immunoediting hypothesis. The process of cancer immunoediting consists of three stages: elimination, equilibrium, and escape. In the elimination phase, transformed cells are killed by antitumor effector mechanisms. Some of these cells can survive and enter the equilibrium phase in which variants of cancer cells are generated that can avoid an immune attack. In the escape phase, the tumor grows progressively due to promotion of local immunosuppression which allows the antitumor response to be evaded (see details in the text) (modified from [30])
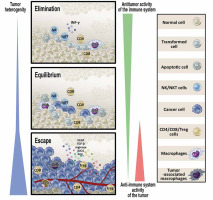
In the elimination phase, innate and adaptive arms of the immune system work in cooperation to identify and destroy transformed cells. These cells are recognized as dangerous and are deleted due to phenotypic changes: presentation of tumor neoantigens, expression of stress ligands, releasing excessive damage-associated molecular pattern (DAMP) molecules [31].
Occasionally, some potentially tumorigenic cells survive the elimination phase and enter the next stage – equilibrium. In this phase, cancer cells are kept dormant for years, decades, and even for the life of the host. It seems that, in contrast to the elimination phase, in the equilibrium phase innate immunity is not necessary to control latent tumor cells. Major mechanisms of protection include adaptive elements of the immune system: Th1 cells, CTLs and cytokines of type 1 immunity (IL-12, IL-2, IFN-g) [32, 33].
The escape phase is synonymous with clinical tumor appearance. This phase results from genetic instability of tumor cells favoring development of quickly proliferating cells “invisible” to the immune system [34]. In a parallel process, a proangiogenic microenvironment is shaped allowing progression of cells to macroscopically identified lesions. The final step of this phase is acquisition of the ability to invade tissues and form metastases [35]. The escape phase progresses quickly as occasional necrotic lesions generate inflammation, stimulating the production and release of proangiogenic factors, which promote the healing process and further progression of the tumor. Independently, cellular and humoral components (e.g., Treg cells, myeloid-derived suppressor cells, IL-10) prevent an effective antitumor response and aggravate immune inefficiency in the tumor microenvironment. To summarize, the lack of anticancer immune response and the creation of an optimal microenvironment is an active process shaped by tumor cells and is a result of various mechanisms. These mechanisms involve several phenomena [31, 36].
Resistance of tumor cells to the cytotoxic effect of cellular and humoral components of the immune system. An example is increased expression of prosurvival proteins allowing prevention of cancer cell destruction: BCL-2 [37], proteinase inhibitor 9 (PI-9, serpin B9, an inhibitor of granzyme B-mediated apoptosis) [38] and cellular FLICE inhibiting protein (c-FLIP, CFLAR) [39].
Ineffective induction of antitumor response (priming) and limited recognition of tumor cells by the immune system. These mechanisms include inefficient presentation of tumor neoantigens by dendritic cells (most of them are, in fact, tolerogenic) [40, 41], absence of signals mediated by costimulatory molecules, decreased expression of MHC molecules on neoplastic cells (e.g. due to a mutated gene for b2-microglobulin) [41-43], and counteracting the cytotoxic effect of T lymphocytes through PDL-1 expression on neoplastic cells and tumor-infiltrating cells [44].
Formation of an immunosuppressive tumor microenvironment. Both cellular and soluble mediators create a milieu in which the tumor can grow. Tumors, as a rule, are infiltrated with cells of evident immunosuppressive function. This is often due to chronic inflammation and results from chemotactic factors released from tumor cells [45, 46]. The best characterized suppressive cells include myeloid-derived suppressor cells (MDSC), regulatory T (Treg) cells, and type 2 macrophages [47]. These cells, as well as cancer cells, produce and release mediators that are effective in inhibiting the immune response. The most important are: indoleamine 2,3-dioxygenase (IDO) [48], arginase-1 [49], cyclooxygenase-2 (COX-2) [50], and cytokines: transforming growth factor β (TGF-b), interleukin (IL)-10, and vascular endothelial growth factor (VEGF) [51, 52]. The latter additionally promotes tumor development via stimulation of angiogenesis. Certain mediators, such as TGF-b and VEGF, are typical markers of chronic inflammation and are characteristic for the physiological process of wound healing [53], promoting development of Treg cells responsible for prevention of autoimmunity [54, 55]. Recent studies have also shown that the immunosuppressive effect of the tumor microenvironment may be due to limited availability of nutrients (glucose deprivation) and an excessive amount of tumor cell-derived metabolites (e.g., lactate, fatty acids). This imbalance may negatively affect differentiation, proliferation, and function of effector tumor-infiltrating T lymphocytes (TILs) [56, 57].
Tumor immune landscape and the Immunoscore concept
In clinical oncology, a positive correlation has been observed for years between the intensity of cellular infiltrations in the tumor and the prognosis. Experiments in mice that provided evidence supporting the role of adaptive immunity in cancer immunoediting were the rationale for in-depth clinical studies evaluating immune components in human tumors and looking for predictive and prognostic values of immune markers in the tumor. Early detailed studies were carried out, among others, on melanoma [58], esophageal cancer [59], and on ovarian cancer [60, 61]. Sato et al. [61] performed an immunohistochemical analysis of cancer tissue sections and found that the presence of intratumoral (intraepithelial) CD4+ T cells and high CD8+ T cell/Treg ratio had favorable prognostic value. The most comprehensive investigations have been conducted, however, on colorectal cancer by the group of French investigators headed by Jerome Galone and Franck Pages. They used immunohistochemical staining and gene expression profiling for characterization of tumor infiltrating immune cells and type of adaptive immunity in the tumor. They found that Th1 type of immunity and high density of CD3+, CD8+, and CD54RO+ (memory marker) cells, both in the center of the tumor and in the invasive margin, predicted good clinical outcome [62]. Results of the study became the basis for introducing the term immune contexture in oncology in the first decade of this century. This term defines the immune landscape in the tumor and includes four characteristic features: 1) quality of tumor infiltrating lymphocytes, 2) density of these cells in the tumor, 3) orientation of the immune response in the tumor, and 4) localization of components of the immune system in tumor and paratumor areas and presence or absence of tertiary lymphoid structures [63].
The obvious consequence of research on the immune contexture in tumors was to define the most optimal prognostic immune parameters and introduction of the term “Immunoscore”. The key parameters in the Immuno- score were amounts of cytotoxic lymphocytes (CTLs) CD3+CD8+, CD45RO+ cells, both in the tumor center and at the invasive margin. In general, Immunoscore = 0 characterized low density of the above-mentioned T cells in the center and periphery of the tumor, and Immunoscore = 4 defined high density of these cells in both regions [64, 65]. The Immunoscore was defined based on immunostained, formalin-fixed, paraffin-embedded slides.
In research by Pages et al. [66], strong prognostic value of the Immunoscore was demonstrated in patients with localized, early stages colorectal cancer (stages I and II, according to the TNM classification) [66]. Five-year survival of patients with the highest Immunoscore (high density of CD8+ and CD45RO+ cells in the center of the tumor and in the invasive margin) was 3 times higher when compared to patients with Immunoscore = 0 (86% vs. 28%). These observations were confirmed in international studies aimed at assessing the prognostic value of T-cell density in the tumor and cytotoxic T cell counts in patients with stage I, II, and III colon cancer [67]. In these studies, in which 14 oncological centers were included from different countries, CD3+ and CD8+ T cells were determined in the core tumor and invasive margin regions, based on paraffin sections processed by immunochemistry, by image analysis software with a dedicated Immunoscore module. CD3 and CD8 markers were chosen because previous investigations showed that these markers of tumor-infiltrating cells were the optimal combination for prognostic purposes. A three-tier categorization system was applied (Immunoscore: low, intermediate, and high). It was found that patients with a high Immunoscore had prolonged overall survival and disease-free survival (DFS). DFS at 5 years was observed in 75% of patients with a high Immunoscore, 70% of patients with an intermediate Immunoscore, and 57% of patients with a low Immunoscore. Of note, improvement of prediction for overall survival was observed when the Immunoscore was added to a model that combined all clinical variables [67].
Significance of the immune contexture in cancer prognosis: suggestion to improve the classification of tumor staging
The current assessment of tumor staging and spread is based on the tumor-node-metastasis (TNM) classification. This classification has strong prognostic significance and has been used in clinical oncology for decades. However, due to accepting the significant role of the immune system in cancer surveillance and development, there have been attempts to improve the TNM staging system. Pages and Galon proposed to introduce an “Immune” component (from the Immunoscore) to classic TNM staging, resulting in a new classification – TNM-Immune [67]. They suggested that determining the immune contexture in routine histopathological samples may be helpful in prognosis of cancer progression and could also be beneficial from a therapeutic point of view [64, 68, 69]. Their suggestions are in line with the current trend of defining an optimal immune landscape in the tumor, predicting a good response to immunotherapy with monoclonal antibodies from the group of immune check-point inhibitors [70].
However, there are several obstacles to the broad acceptance of the proposed classification (TNM-Immune). The most problematic issue concerns extremely heterogeneous immune infiltrates in the tumor microenvironment, especially in advanced forms of cancer (stages III and IV) [71-73]. Some tumors are heavily infiltrated while in others the density of infiltrating cells is low [74] and includes both “good” elements (cytotoxic T lymphocytes, NK cells) and “bad” components (e.g., myeloid-derived suppressor cells or M2 macrophages) [75, 76]. To complicate the picture, some “good” cells may be inactive (exhausted) and non-functional. Another question is the problematic protective role of the immune system in the most advanced stages of tumors and the interrelationship between the extracellular matrix in cancer and components of the immune system [77]. Very reliable and in-depth research concerning lung cancer, describing the immune landscape from preneoplasia to invasive adenocarcinoma, demonstrated gradual loss of immune effectiveness of antitumor defense, as the cancer progresses. These studies clearly show that in the escape phase of the process of cancer immunoediting, the tumor actively sculptures and shapes immunity, causing non-functionality of protective components of the immune system [78] (Fig. 1).
Conclusions
At present, the Immunoscore is unlikely to be widely used to define tumor stage of progression and as a prognostic indicator in clinical oncology. Resolving some problems can be difficult: time-consuming histochemical techniques, the need to incur additional costs, interlaboratory non-reproducibility of assays, etc. However, due to diagnostic advances and computed tomography screening, more and more tumors will be detected in the future in early stages of development [79, 80]. In these cases, in addition to regular monitoring of tumor behavior, assessment of the intratumoral immune milieu and introducing the Immunoscore staging could be helpful in optimal prediction of therapeutic strategies [81, 82].


