Introduction
Adipose tissue affects the immune system leading to the development of an inflammatory process of a peculiar presentation and low intensity. Its pathogenesis is complex and involves multiple pathways. Fat cells, mainly visceral, behave as immune cells. They produce and release a large number of proinflammatory adipokines and cytokines such as leptin, adiponectin, resistin, tumor necrosis factor-α (TNF-α), interleukin (IL)-1β and IL-6, monocyte chemoattractant protein-1 (MCP-1), macrophage migration inhibitory factors (MIF), C-reactive protein (CRP), and plasminogen activator inhibitor-1 (PAI-1), which activate further proinflammatory pathways and promote oxidative stress to impact negatively surrounding tissues. The production of other cytokines, chemokines, vasoactive amines, and prothrombotic factors increases, the expression of cell adhesion molecules is enhanced, while neutrophils and monocytes are recruited from the marginated pool [1-5]. Moreover, cytokines activate the hematopoietic system, and stimulate the growth of granulocyte and monocyte progenitor cells and thrombopoiesis [6]. Activation of T lymphocytes and their polarization into Th1 lymphocytes are responsible for the cellular immunity response [2, 4]. These inflammatory reactions accompanying obesity are reinforced by adipose tissue macrophages and lymphocytes [2, 3]. The visceral adiposity is also metabolically active. Most secreted adipocytokines and cytokines are involved in development of insulin resistance, disturbances in lipid and carbohydrate metabolism and hypertension [3]. The low-grade chronic inflammatory process concomitant with numerous metabolic and hemodynamic disturbances is considered to be one of the major players of the metabolic syndrome and atherosclerosis process [1, 3].
Elevated inflammatory biomarkers are consistent with the ongoing inflammatory process and some of them are routinely assessed in clinical practice. Overweight and obese patients have a higher total leukocyte count compared to their lean peers, which is mostly the result of the increase in the monocyte count [7]. Neutrophil, lymphocyte and eosinophil counts may also be elevated [8-10]. Greater adiposity leads to a higher CRP level [1, 8, 11-13]. Some studies in obese adults showed that weight loss improved inflammation parameters [3, 14]. Limited investigations regarding the impact on lifestyle changes on an inflammatory process in obese children have been carried out [8, 15-17].
The purpose of the retrospective study was to analyze the leukocyte profile and C-reactive protein concentration in overweight and obese adolescents compared to normal weight peers and their changes after 6 to 12 months of dietary intervention.
Material and methods
We analyzed retrospectively medical records of overweight, obese and normal weight children over the age of 10, who were hospitalized in the Department of Paediatrics and Endocrinology at the Medical University of Warsaw, Poland from 2015 to 2017. Exclusion criteria included overweight and obesity due to hormonal or genetic disorders and secondary to central nervous system diseases and immune system disturbances. None of the patients included in this study had a positive history of allergy or symptoms of acute infection over the last 3 weeks before hospitalization, and none of them used any medications. All patients were nonsmokers. Overweight and obesity were defined by the percentile age- and sex-specific BMI curves based on a nationally representative group of children aged 3-18 years of the OLAF and OLA project [18]. Finally, 99 children aged from 10.0 to 17.5 years (median age 13.6 years): 20 overweight (OW: 8 girls and 12 boys) and 79 obese (O: 49 girls and 30 boys) were enrolled in the study. At the first hospitalization (the baseline visit), all of the children underwent dietary intervention which included the analysis of their diet as conducted by dietitians, the typical level of physical activity and correction of nutritional mistakes with recommendations for a healthy lifestyle. The energy value of the recommended diet was tailored individually based on the age, sex and level of physical activity, according to Polish recommendations (supplementary material). Eighty-two children in the study group (16 OW/66 O) volunteered for a scheduled re-hospitalization follow-up 6 to 12 months after the nutrition counseling.
The control group consisted of 42 normal weight children who were age- and sex-matched (24 girls and 18 boys) aged from 10.0 to 17.9 years (median age 14.4 years), who were evaluated only once (the baseline visit).
Based on body mass index standard deviation score (BMI SDS) changes between the baseline and the follow-up visit (ΔBMI SDS = BMI SDSat follow-up visit – BMI SDSat baseline), the overweight and obese children were divided into three groups:
children who lost weight – ΔBMI SDS < 0 (n = 45),
children who gained weight – ΔBMI SDS > 0 (n = 22),
children with unchanged weight – ΔBMI SDS = 0 (n = 15).
The study protocol was approved by the Bioethical Committee at the Medical University of Warsaw, Poland.
Supplementary material
The assessment of the nutritional habits of children and adolescents was carried out (with the parents/legal guardians present) based on an interview encompassing 3 days with one weekend day. The information concerned the number and distribution of meals during the day, the composition of meals and how they were prepared, the number and type of snacks consumed, and the amount and type of drinks consumed. Portion sizes were assessed using the “Album of photographs of food products and dishes” [19]. The collected data also concerned the typical level of physical activity.
Food energy and the content of nutrients in the menus were estimated using the “Energia” program comprising appropriate coefficients of loss resulting from technological processes. The obtained data were compared against consumption reference values for the Polish population appropriate for the age, sex and physical activity level of the participants [20]. Based on Polish recommendations of the National Food and Nutrition Institute [20], the patient obtained information about correct nutritional behaviors and physical activity. Dietary intervention was based on The Healthy Eating and Physical Activity Pyramid for children and adolescent aged 4-18 years, for example: 5 meals throughout the day (intervals between meals 3-4 hours), eliminating highly processed products (e.g. white flour, sugar, salt, fast food products, sweet drinks), replacing animal fat with vegetable oil, eating more fish and low-fat meat instead of red meat, drinking natural water, more vegetables and fruits per day, as well as daily physical activity at school and beyond. Recommendations were given individually based on the patient’s current health.
Anthropometric measurements
All children underwent anthropometric measurements conducted by a qualified anthropologist. Anthropometric measurements were taken with the participants wearing only underwear, standing in the anatomical position, upright, with the head in the Frankfort horizontal plane. Body weight (kg) was measured by means of medical scales to the nearest 0.1 kg, height (cm) was measured using a stadiometer (Holtain Limited) to the nearest 0.1 cm. Waist (WC, cm) and hip circumference (HC, cm) were measured by an elastic measuring tape, according to WHO recommendations [21]. The skinfold thickness (mm) was measured under the triceps brachii muscle and under the inferior scapular angle by means of a skinfold caliper Harpenden type on the non-dominant side of the body. Based on these measurements, BMI, the waist-to-hip ratio (WHR), the waist-to-height ratio (WHtR) and the percentage of fat tissue (% BFM) using the Slaughter formula were calculated [22]. The degree of obesity expressed as BMI SDS was calculated using the LMS method, to normalize skewness of the distribution of BMI [23]. The threshold of obesity was set at BMI SDS ≥ 2, and the threshold of overweight was set at BMI SDS ≥ 1 and < 2 [21]. WHtR exceeding 0.5 was assumed to define abdominal obesity. The norm for body fat percentage was set at 19% for girls and 15% for boys [22].
Blood examination
Blood samples were collected after overnight fasting and analyzed by standard methods. Complete blood counts were obtained by an automated blood cell counter (XN-1000, Sysmex). Total cholesterol (TC, mg/dl), triglycerides (TG, mg/dl), and high-density lipoprotein cholesterol (HDL-C, mg/dl) concentrations were determined by the colorimetric enzymatic method using the Vitros 5600 analyzer (Ortho Clinical Diagnostic). Low-density lipoprotein cholesterol (LDL-C) was calculated using the Friedewald formula [LDL-C = TC – (HDL + TG/5)]. Concentration of CRP (mg/dl) was determined using a fixed-point immune-rate method; concentration of fasting glucose (mg/dl) was measured by the glucose oxidase colorimetric method, both on the Vitros 5600 analyzer (Ortho Clinical Diagnostic). Fasting insulin (μIU/ml) concentration was evaluated by the chemiluminescent enzyme immunoassay method on the Immulite 2000 XPi (Siemens).
The homeostasis model assessment – insulin resistance (HOMA-IR) and the TG/HDL ratio were calculated [24, 25]. Lipid profile and glucose metabolism parameters were evaluated based on the recommendations of the American Heart Association (AHA, 2007) [26] and the Polish Diabetes Association (PTD, 2018) [27]. Fasting insulin level ≥ 15 μIU/ml was considered elevated [24]. The HOMA-IR value ≥ 3.16 [28] and the TG/HDL-C ratio ≥ 3 [25] were closely correlated with insulin resistance (IR).
Statistical analysis
Detailed statistical calculations were performed using Statistica 13.1. Data distribution was checked by the Shapiro-Wilk test. The data were presented as the mean ±standard deviation (SD) or the median with the interquartile range, as appropriate. Differences in anthropological and biochemical parameters between the overweight and obese group and the control group were examined using the t-test for normally distributed data or the Mann-Whitney U test when the distribution was different from normal. In order to assess the relationship between analyzed variables in overweight and obese children, the Spearman correlation coefficient was used. Comparison between the same parameters at the baseline and at the follow-up visit was obtained using the Wilcoxon test. Independent predictors, i.e. WBC (white blood cell), neutrophil and monocyte counts at baseline and their changes within the observation period, were analyzed using the backward stepwise regression analysis. A p-value < 0.05 was statistically significant.
Results
The characteristics of anthropometric and biochemical parameters in overweight and obese children (study group) and their normal peers (control group) are presented in Table 1.
Table 1
Comparison of anthropometric measurements, hematological parameters, lipid profile and CRP concentration between overweight and obese children (study group) and normal weight children (control group)
[i] Data are presented as mean ±standard deviation (SD) or median values with interquartile range as appropriate; BMI – body mass index, BMI SDS – body mass index standard deviation score, WC – waist circumference, HC – hip circumference, WHR – waist-to-hip ratio, WHtR – waist-to-height ratio, % BFM – % of body fat mass, WBC – white blood cells, RBC – red blood cells, Hb – hemoglobin, CRP – C-reactive protein, TC – total cholesterol, HDL-C – high-density lipoprotein cholesterol, LDL-C – low-density lipoprotein cholesterol, TG – triglycerides, ns – non-significant.
All study group children were found to have a high body fat percentage. In 85% of them, their WHtR met the criteria for diagnosing abdominal obesity. They had an atherogenic lipid profile. Almost half of the children had an elevated TG/HDL ratio (48 individuals; 48.5% of the group), fasting insulin concentration (48; 48.5%) and HOMA-IR (42; 42.4%). Overweight and obese children had higher WBC, neutrophil and monocyte counts than the controls. We also noted a tendency for an elevated lymphocyte count. Moreover, the red blood cell (RBC) count and CRP concentration were higher in the study group compared to the control group. The platelet count did not reveal any significant differences.
Relationship between the leukocyte profile, CRP concentration and anthropometric and biochemical parameters in overweight and obese children at the baseline visit
The results of the Spearman correlation analysis of the total leukocyte and neutrophil counts, CRP concentration and selected anthropometric and biochemical parameters in overweight and obese children are shown in Table 2. The baseline WBC count poorly correlated only with the baseline BMI SDS. Considering biochemical parameters, the baseline WBC count weakly correlated with baseline CRP, fasting insulin, TG, HDL-C concentrations, TG/HDL-C ratio and HOMA-IR. The baseline neutrophil count correlated with body weight, WC, HC, % BMF, BMI, BMI SDS and with the same baseline biochemical parameters. The monocyte count correlated only with WC (rs = 0.208, p = 0.017). Among analyzed parameters, the lymphocyte count correlated only with TG levels (rs = 0.218, p = 0.034). The CRP concentration correlated with almost all anthropometric parameters describing fatness, but not with carbohydrate and lipid parameters. We did not find any significant relationship between the leucocyte profile, CRP concentration and WHR, fasting glucose, TC and LDL-C concentrations at baseline.
Table 2
Correlations between white blood cells, neutrophil count, CRP concentration and selected anthropometric and biochemical parameters in overweight and obese children at the baseline visit
[i] Data are presented as rs – Spearman correlation coefficient and below p-value or ns – non- significant; BMI – body mass index, WC – waist circumference, HC – hip circumference, WHtR – waist- to-height ratio, % BFM – % of body fat mass, WBC – white blood cells, CRP – C-reactive protein, TG – triglycerides, HDL-C – high-density lipoprotein cholesterol, TG/HDL-C – triglycerides to high-density lipoprotein cholesterol ratio, HOMA-IR – homeostasis model assessment – insulin resistance
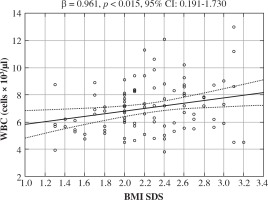
The differences derived from the comparison between variables were confirmed by the backward stepwise regression analysis. Six models were proposed in order to evaluate the relationship between the baseline WBC count (models 1 and 2), the baseline neutrophil count (models 3 and 4) and the baseline monocyte count (models 5 and 6), which were considered as dependent variables and selected independent variables (models 1, 3 and 5: BMI SDS, % BFM, WC and WHtR; models 2, 4, and 6: CRP and fasting insulin concentrations, HOMA-IR and TG/HDL ratio). The baseline BMI SDS and fasting insulin concentration were identified as independent predictors of both the baseline WBC count (model 1: β = 0.961, 95% CI: 0.191-1.730, R2 = 0.061, F = 6.15, p < 0.015, Fig. 1; model 2: β = 0.040, 95% CI: 0.011-0.067, R2 = 0.10, F = 7.93, p < 0.006) and the baseline neutrophil count (model 3: β = 1.04, 95% CI: 0.467-1.613, R2 = 0.121, F = 12.99, p < 0.01; model 4: β = 0.031, 95% CI: 0.009-0.053, R2 = 0.105, F = 8.34, p < 0.005). No correlation between the baseline monocyte count in models 5 and 6 was found.
Changes in the leukocyte profile, CRP concentration in overweight and obese children within the observation period and the relationship with anthropometric and biochemical parameters
Eighty-two children were present at the follow-up visit. Forty-five of them (8 OW/37 O) lost weight (∆BMI SDS > 0), 22 (3 OW/19 O) gained weight (∆BMI SDS < 0) and in 15 (5 OW/10 O) BMI SDS (∆BMI SDS = 0) remained unchanged.
The characteristics of anthropological measurements and laboratory parameters are shown in Table 3. In children who lost weight, a decline in BMI SDS was correlated, as expected, with a decrease in other anthropometric parameters. A decrease in the WBC count (p = 0.011), the neutrophil count (p = 0.014) and the monocyte count (p = 0.014) compared to baseline values was found. The leucocyte profile in overweight and obese children after reduction of body weight was comparable to the control group. The CRP (p = 0.009), fasting insulin (p = 0.029), TC (p < 0.001), LDL-C (p < 0.001) and TG concentrations (p = 0.046) decreased at the follow-up visit compared to baseline measurements. However, in children who gained weight and whose weight remained unchanged, we did not observe any changes in blood and biochemical parameters, but it could result from the small number of participants in these groups. We only found a tendency to a higher fasting glucose level and insulin concentration in children who gained body weight (p = 0.05, p = 0.067, respectively).
Table 3
Changes in anthropometric measurements, hematological parameters, CRP concentration, lipid profile and HOMA-IR in overweight and obese children according to changes in BMI SDS at the follow-up visit
| Variable | Overweight and obese group – baseline visit (n = 99) | Overweight and obese children – the follow-up visit (n = 82) | |||||
|---|---|---|---|---|---|---|---|
| Lost weight (n = 45) | Gain weight (n = 22) | Body weight unchanged (n = 15) | |||||
| baseline visit | follow-up visit | baseline visit | follow-up visit | baseline visit | follow-up visit | ||
| Height (cm) | 164.71 ±11.15 | 163.84 ±12.15 | 165.89 ±11.44 | 164.09 ±8.06 | 166.59 ±7.84 | 164.22 ±14.34 | 167.04 ±13.35 |
| Body weight (kg) | 83.50 ±20.10 | 82.34 ±19.12 | 79.42 ±18.48*** | 81.62 ±14.98 | 88.40 ±13.31** | 83.87 ±28.47 | 86.46 ±26.44*** |
| BMI (kg/m2) | 30.37 ±4.55 | 30.27 ±4.04 | 28.47 ±4.11*** | 30.22 ±4.74 | 31.86 ±4.64*** | 30.28 ±5.52 | 30.28 ±4.86 |
| BMI SDS | 2.24 ±0.46 | 2.27 ±0.44 | 1.93 ±0.54*** | 2.24 ±0.46 | 2.40 ±0.48*** | 2.14 ±0.50 | 2.13 ±0.49 |
| WC (cm) | 91.0 (82.0-95.0) | 91.0 (83.0-94.0) | 86.0 (77.0-94.0)*** | 88.0 (82.0-94.0) | 92.0 (87.0-97.0)** | 90.0 (79.0-101.0) | 89.50 (81.0-99.0) |
| HC (cm) | 105.0 (98.0-112.5) | 106.0 (90.0-111.0) | 103.0 (93.0-110.0)*** | 104.0 (99.0-114.0) | 110.0 (100.0-118.0)*** | 103.0 (95.0-112.5) | 104.0 (97.0-115.0) |
| WHR | 0.85 ±0.05 | 0.85 ±0.05 | 0.83 ±0.05*** | 0.84 ±0.05 | 0.83 ±0.05 | 0.87±0.05 | 0.86 ±0.05 |
| WHtR | 0.54 (0.51-0.58) | 0.53 (0.51-0.57) | 0.51 (0.48-0.59) | 0.54 (0.49-0.58) | 0.54 (0.50-0.60)** | 0.55 (0.51-0.59) | 0.54 (0.50-0.60) |
| % BFM | 35.65 (32.40-41.10) | 37.40 (33.70-40.30) | 33.20 (29.90-35.90)*** | 35.40 (35.00-39.70) | 36.70 (32.10-40.30) | 34.30 (31.00-40.00) | 34.30 (29.40-37.00) |
| WBC (cells × 103/μl) | 6.90 (5.68-8.24) | 6.90 (6.10-8.58) | 6.20 (5.70-7.49)* | 6.50 (5.61-7.50) | 6.95 (5.95-8.15) | 7.70 (5.26-8.74) | 6.99 (4.93-8.36) |
| Neutrophils (cells × 103/μl) | 3.50 (2.64-4.30) | 3.70 (3.02-5.00) | 3.36 (2.70-3.74)* | 3.15 (2.50-4.00) | 3.30 (2.56-4.32) | 3.50 (2.20-4.83) | 3.33 (2.14-4.70) |
| Monocytes (cells × 103/μl) | 0.54 (0.50-0.61) | 0.51 (0.50-0.65) | 0.47 (0.40-0.60)* | 0.53 (0.40-0.66) | 0.57 (0.48-0.69) | 0.55 (0.50-0.77) | 0.58 (0.48-0.75) |
| Eosinophils (cells × 103/μl) | 0.20 (0.10-0.30) | 0.14 (0.10-0.30) | 0.20 (0.10-0.25) | 0.14 (0.10-0.26) | 0.18 (0.11-0.25) | 0.26 (0.10-0.36) | 0.17 (0.10-0.35) |
| Basophils (cells × 103/μl) | 0.01 (0.00-0.04) | 0.01 (0.00-0.07) | 0.01 (0.00-0.03) | 0.01 (0.00-0.03) | 0.02 (0.00-0.03) | 0.02 (0.00-0.04) | 0.01 (0.00-0.02) |
| Lymphocytes (cells × 103/μl) | 2.50 (2.14-2.90) | 2.40 (2.10-2.71) | 2.20 (1.90-2.59) | 2.45 (2.00-2.90) | 2.50 (2.10-3.17) | 2.80 (2.30-3.14) | 2.60 (2.13-2.92) |
| Platelets (cells × 103/μl) | 268.42 ±55.19 | 262.62 ±51.41 | 259.12 ±51.57 | 264.22 ±62.83 | 266.45 ±69.24 | 279.33 ±48.0 | 259.13 ±55.3 |
| RBC(cells × 106/μl) | 5.01 (4.73-5.20) | 5.03 (4.80-5.23) | 4.86 (4.67-5.19) | 4.96 (4.71-5.17) | 4.97 (4.70-5.12) | 5.08 (4.74-5.26) | 4.99 (4.68-5.30) |
| Hb (g/dl) | 13.80 (13.20-14.50) | 13.80 (13.20-14.40) | 13.70 (13.00-14.30) | 13.45 (12.60-14.10) | 13.55 (12.95-14.35) | 14.20 (0.50-0.75) | 14.0 (13.30-15.20) |
| CRP (mg/dl) | 0.5 (0.5-0.8) | 0.6 (0.5-0.9) | 0.5 (0.5-0.6)** | 0.5 (0.5-0.6) | 0.5 (0.5- 0,6) | 0.5 (0.5-0.7) | 0.5 (0.5-0.5) |
| Fasting glucose (mg/dl) | 83.65 ±8.96 | 84.25 ±10.45 | 85.87 ±6.48 | 82.88 ±7.33 | 86.34 ±6.85 | 84.56 ±6.64 | 84.48 ±4.89 |
| Fasting insulin (μlU/ml) | 14.15 (8.56-22.10) | 15.80 (11.20-28.70) | 13.90 (9.84-23.0)* | 12.0 (7.60-21.80) | 15.50 (10.90-23.90) | 13.10 (7.36-24.90) | 13.0 (11.5-21.90) |
| TC (mg/dl) | 166.02 ±29.12 | 166.09 ±31.55 | 150.78 ±24.01*** | 165.75 ±28.61 | 167.77 ±31.03 | 166.78 ±22.95 | 161.0 ±24.6 |
| HDL-C (mg/dl) | 43.0 (38.0-48.0) | 43.0 (38.0-47.0) | 43.0 (39.0-46.0) | 39.1 (33.0-44.0) | 40.0 (37.0-48.0) | 44.0 (40.0-50.0) | 42.5 (40.0-48.0) |
| LDL-C (mg/dl) | 95.18 ±26.36 | 95.99 ±26.50 | 82.86 ±18.18** | 92.44 ±28.86 | 95.86 ±26.69 | 95.92 ±23.14 | 90.24 ±21.92 |
| TG (mg/dl) | 116.0 (86.0-161.0) | 118.5 (91.0-164.0) | 113.0 (74.0-144.0)* | 122.0 (99.0-161.0) | 124.0 (102.0-193.0) | 98.1 (79.0-155.0) | 113.5 (90.0-143.0) |
| TG/HDL-C | 2.58 (1.91-4.06) | 2.75 (2.00-3.97) | 2.77 (1.66-3.34) | 3.78 (2.01-4.46) | 2.91 (2.00-5.12) | 2.15 (1.67-3.69) | 2.53 (2.04 -2.95) |
| HOMA-IR | 2.92 (1.79-5.24) | 3.06 (2.12-6.19) | 3.03 (2.00-4.41) | 2.48 (1.56-4.98) | 3.27 (2.52-4.98) | 2.78 (1.53-5.96) | 2.83 (2.40-4.59) |
Data are presented as mean ±standard deviation (SD) or median with interquartile range as appropriate, BMI – body mass index, WC – waist circumference, HC – hip circumference, WHR – waist-to-hip ratio, WHtR – waist-to-height ratio, % BFM – % of body fat mass, WBC – white blood cells, RBC – red blood cells, Hb – hemoglobin, TC – total cholesterol, TG – triglycerides, HDL-C – high-density lipoprotein cholesterol, LDL-C – low-density lipoprotein cholesterol, TG/HDL-C – triglycerides to high-density lipoprotein cholesterol ratio, HOMA-IR – homeostasis model assessment – insulin resistance
In the three groups considered together, the changes in the WBC count at the follow-up visit (ΔWBC = WBCat follow-up visit – WBCat baseline) correlated with changes in BMI (ΔBMI = BMIat follow-up visit – BMIat baseline; p = 0.007) and BMI SDS (ΔBMI SDS = BMI SDSat follow-up visit – BMI SDSat baseline; p = 0.002). A similar positive relationship was also found between Δneutrophil count (Δneutrophil count = neutrophil countat follow-up visit – neutrophil countat baseline) and Δmonocyte count (Δmonocyte count = monocyte countat follow-up visit – monocyte countat baseline) and ΔBMI (p = 0.022; p = 0.001, respectively) and ΔBMI SDS (p = 0.032; p = 0.017). Moreover, changes in the monocyte count correlated with changes in WHtR (ΔWHtR = WHtRat follow-up visit – WHtRat baseline; p = 0.002). Considering some biochemical parameters, changes in leukocytes correlated significantly with Δinsulin concentration (Δ insulin = insulinat follow-up visit – insulinat baseline; p = 0.025), ΔHOMA-IR (ΔHOMA-IR = HOMA-IRat follow-up visit – HOMA-IRat baseline; p = 0.022), ΔTC (ΔTC = TCat follow-up visit – TCat baseline; p = 0.014), ΔTG (ΔTG = TGat follow-up visit – TGat baseline; p = 0.02), and ΔTG/HDL-C ratio (ΔTG/HDL-C = TG/HDL-Cat follow-up visit – TG/HDL-Cat baseline; p = 0.011). Changes in the neutrophil count were associated with ΔTG (p = 0.011), ΔTG/HDL-C ratio (p = 0.009) and ΔCRP (Δ CRP = CRPat follow-up visit – CRPat baseline; p = 0.005). Changes in the monocyte count correlated significantly only with ΔTG (p = 0.026). No correlation between changes in the CRP concentration and analyzed anthropometric and biochemical parameters was found.
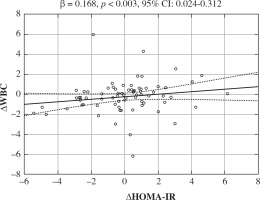
In order to evaluate independent relationships between ΔWBC count (model 7), ∆neutrophil count (model 8) and Δmonocyte count (model 9), which were considered as dependent variables, and changes in selected anthropometric and biochemical parameters during 6-12 months of the observation period, which were considered as independent variables, backward stepwise regression models were used (model 7 including ∆BMI SDS, ∆insulin concentration, ∆HOMA-IR and ∆TG/HDL ratio; models 8 and 9 including ∆BMI SDS, ∆insulin concentration, ∆TG/HDL ratio and ∆CRP). In models 7 and 8, ∆HOMA-IR (β = 0.168, 95% CI: 0.024-0.312, R2 = 0.175, F = 6.26, p < 0.003, Fig. 2; β = 0.135, 95% CI: 0.023-0.246, R2 = 0.192, F = 7.03, p < 0.002, respectively) and ∆TG/HDL ratio (β = 0.305, 95% CI: 0.074-0.534, R2 = 0.175, F = 6.26, p < 0.003; β = 0.256, 95% CI: 0.077-0.434, R2 = 0.192, F = 7.03, p < 0.002) were identified as independent predictors of both ∆WBC count and ∆neutrophils. In model 9, ΔHOMA-IR had a significant impact on Δmonocyte count (β = 0.719, 95% CI: 0.079-1.360, R2 = 0.116, F = 1.87, p < 0.03).
Discussion
Our study confirmed previous observations that accumulation of fat mass leads to development of low-grade systemic inflammation. Overweight and obese children had a higher peripheral total leukocyte count compared to their lean peers [9, 10, 29]. It is mainly related to the elevated monocyte count [7]. Some researchers have demonstrated an increased count of WBC subfractions such as neutrophils, lymphocytes or eosinophils [9-11]. In our study, overweight and obese teenagers had elevated monocyte and neutrophil counts, and a slightly increased lymphocyte count. A higher count of leukocytes is reported also in obese children with non-alcoholic fatty liver disease (NAFLD) [7] and type 2 diabetes [30], which increases the prevalence of metabolic syndrome [31]. Likewise, a chronic inflammatory process activates thrombogenic factors and leads to an increase in the platelet number in patients with excess fat mass [10, 12, 32], but this process has not been confirmed in our study. Differences in WBC, neutrophil, lymphocyte and platelet counts were not always found in other studies [33, 34].
Numerous studies have shown that changes in the leukocyte profile are associated with indices of excess adiposity [9, 10, 32, 35, 36]. In two cross-sectional studies including a representative sample of the Korean pediatric population and Italian overweight and obese children, the mean values of BMI, BMI z-score, waist circumference and WHtR increased along with the increase in WBC count quartile [37, 38]. In another study, a higher neutrophil count in 77 obese children correlated also with waist circumference and WHR [11]. Our study has brought similar results. The WBC count in overweight and obese children correlated with BMI SDS, the monocyte count with waist circumference and the neutrophil count with all anthropometric measurements relating to fatness. However, in the multiple regression analysis, the only independent variable significantly impacting total leukocyte and neutrophil counts was BMI SDS. The data can explain the mechanism leading to the changes in the WBC count and its subfractions. Visceral fat is generally much more proinflammatory than subcutaneous adipose tissue. It produces a huge number of proinflammatory adipokines and cytokines which are responsible for the development of an inflammatory process. IL-6 stimulates differentiation and proliferation of multipotential hematopoietic progenitors into granulocytes and macrophages, accelerates neutrophil release from the bone marrow and induces their demargination from the marginal pool [6]. Moreover, IL-6, whose synthesis is induced by both TNF-α and IL-1, also regulates CRP production in the liver [5, 29]. Visser et al. found, after the adjustment of factors influencing CRP, that in overweight boys and girls, the CRP concentration was elevated 3.74 times and 3.17 times, respectively, compared to their normal weight peers [29]. Furthermore, the common signaling paths in the production of neutrophils and CRP could elucidate the link between total leukocyte and neutrophil counts and CRP concentration found in the literature and in our study [11, 12]. Leptin plays an important role in controlling immunity and inflammation. Beside neutrophil, monocyte and macrophage activation, leptin also stimulates human lymphocyte T cell proliferative responses, and increased Th1 and suppressed Th2 cytokine production regulates functions of natural killer (NK) cells and induces B lymphocytes to produce proinflammatory cytokines [2, 4]. These alterations in lymphocyte homeostasis are confirmed by clinical trials [4, 9]. The tendency to an elevated lymphocyte count, which was observed in our study in overweight and obese children, may be partially explained by the proinflammatory nature of leptin.
Proinflammatory adipokines and cytokines are involved in the development of insulin resistance [3]. They have a direct negative impact on the insulin signaling pathway and indirect via stimulation of inflammatory pathways and changes in the glucose and lipid homeostasis [2]. In the above-mentioned cross-sectional studies, it was found that the mean values of typical cardiometabolic risk factors (such as triglyceride and insulin concentrations, glucose concentration at 120 minutes of the oral glucose tolerance test, HOMA-IR and WBISI, whole body insulin sensitivity index, and the TG/HDL-C ratio) and increase in the prevalence of insulin resistance rose proportionally with the increase of the WBC count quartile [37, 38]. In our study, the baseline WBC count in overweight and obese individuals correlated with lipid profile and the insulin resistance index, wherein the fasting insulin concentration was their independent predictor. Another study confirmed the relationship between total leukocyte count, their subfractions and insulin resistance [7]. Moreover, Di Bonito et al. observed increased thickness of the common carotid intima media (cIMT) as well as the elevation of the left ventricular mass index (LVMi) and relative wall thickness (RWT) together with increased quartiles of WBC count. Furthermore, children with WBC ≥ 8 700 cell/mm3 are at 1.3 to 2.5-fold greater risk of glucose metabolism disturbance and vascular and cardiac damage [37]. These results make us aware of their public significance. Chronic low-grade inflammation contributes to the development of atherosclerosis in the pediatric population. Studies in adults have revealed that a high leukocyte count is an independent cardiovascular risk factor [39].
Moreover, our study showed that the proinflammatory state is reversible. Elevated counts of total leukocytes, monocytes and neutrophils and CRP concentration in overweight and obese children decreased consistently with their weight reduction and also in the case of the leucocyte profile, achieving values comparable to the control group. In all overweight and obese children, ΔWBC, Δneutrophil and Δmonocyte counts were dependent on ΔBMI, ΔBMI SDS and also ∆HOMA-IR 6 to 12 months after dietary intervention. Studies among children and adolescents are limited and focused on the assessment of other inflammatory parameters. Balagopal et al. found that the elevated blood concentration of CRP, fibrinogen and IL-6 in 21 obese adolescents was reduced after 3 months of moderate physical activity and diet-based lifestyle following an intervention only. At the baseline visit, these inflammatory parameters correlated not only with indices of body fatness but also with the insulin resistance index HOMA-IR and their reduction accompanied improvement of HOMA-IR [15]. In a study including 43 obese children aged 7-11, Ferguson et al. did not find any evidence of the reduction of fibrinogen, PAI-1, or D-dimer concentrations after 4 months of physical training. However, children with a greater percentage of body fat and concentrations of hemostatic factors at baseline had a greater reduction in fibrinogen and D-dimer concentration after physical training compared to those with lower values [16]. A similar conclusion was reached by Barbeau [8]. In a group of 74 obese teenagers participating in three different 8-month interventions relating to lifestyle education (LSE), LSE and moderate-intensity physical training, and LSE with high-intensity physical training, changes in CRP, fibrinogen, D-dimer and PAI-1 concentrations were mainly associated with their baseline values, but not dependent on adiposity and physical activity [8]. Future investigations comprising a bigger number of participants and longer duration of interventions are needed to understand better changes in immunological and inflammatory processes in obesity. It is assumed that long-term care for the obese pediatric population may reduce metabolic and inflammatory disturbances.
Conclusions
Routine tests in pediatric care showed that accumulation of fat mass leads to the development of a low-grade inflammation state. Overweight and obese children have an elevated leukocyte count with increased neutrophil and monocyte counts and increased CRP concentration compared to their normal weight peers. The intensity of this inflammatory process is dependent on the degree of obesity and metabolic disturbances accompanying obesity, especially insulin resistance. Reduction of fat mass can improve lipid profile and fasting insulin concentration and, importantly, decrease serological biomarkers of inflammation. Changes in the insulin resistance index HOMA-IR are an independent predictor of changes in WBC, neutrophil and monocyte counts after a 6-12-month observation period.