The intranasal (IN) route to deliver medications is preferable because this method has advantages over the other methods, such as intravenous (IV), intramuscular, and subcutaneous. The advantages include painless and needleless application, rapid drug absorption, avoidance of gastrointestinal destruction, and hepatic first-pass metabolism [1, 2]. Previous studies have shown that the use of IN opiates is effective for mild to moderate sedation but not for deep sedation [3]. The most proper synthetic opiate medication for IN drug delivery is sufentanil because it has high lipid solubility, a short half-life (15 to 20 minutes), rapid onset of action causing rapid mucosal absorption, and large therapeutic index [4]. The administration of IN sufentanil provides preoperative sedation within 10 minutes, and it has been reported to cause less respiratory depression in comparison to fentanyl [5]. Intranasal administration of sufentanil induces no clinically significant change in vital signs. After IV sufentanil, respiratory depression may occur, and a clinically significant decrease in the arterial partial pressure of oxygen (PaO2) may be seen within 5 minutes [6]. Previously, it has been reported that IN delivery of benzodiazepines, such as midazolam, exhibits approximately 50–80% absolute bioavailability with a rapid onset. However, these studies also reported severe nasal irritation with its use [7, 8].
In this study, our aim was to compare the effects of IN application of 2 different doses of sufentanil or intravenous midazolam on cardiopulmonary safety profile, propofol consumption, and patient and endoscopist satisfaction and recovery during propofol-based sedation in patients undergoing colonoscopy.
METHODS
This was a prospective, randomized, double-blind study to compare 2 different doses of IN sufentanil and IV midazolam during propofol-based sedation for colonoscopy. This study was approved by the local Institutional Review Board.
A total of 121 consecutive patients scheduled for colonoscopy were enrolled to this study. Exclusion criteria were as follows: previous nasal surgery, acute or chronic nasal problems, known sensitivity or allergy to opiates, benzodiazepines, or propofol, patients with chronic pulmonary disease (COPD), obstructive sleep apnoea syndrome (OSAS), neurologic and psychiatric disorders, American Society of Anesthesiologists (ASA) physical classification status > III, age < 18 or > 65 years, pregnancy or breastfeeding, the risk of difficult intubation, a history of analgesic or narcotic abuse, and adverse events in previous sedations.
All patients fasted for 8 hours before the procedure. In the endoscopy room, all the patients received IV isotonic saline at a rate of 8 mL kg–1 h–1 and 3 L min–1 oxygen via a nasal cannula during the procedure. Monitoring of the patients including electrocardiography (ECG), noninvasive blood pressure (NIBP), heart rate (HR), respiratory rate (RR), and peri-pheral oxygen saturation (SpO2) was ensured with a monitoring device (Drager Infinity Delta, Drager Medical Systems Inc., Danvers, MA, USA) before the sedation until the hospital discharge. Moreover, bispectral index score (BIS) monitorization was performed during the procedure with a BIS Vista monitor (Covidien LLC, Mansfield, USA).
The patients were randomly allocated into 1 of 3 groups using sealed envelopes in a 1 : 1 : 1 ratio. An independent researcher prepared 3 sealed envelopes containing a code for each group. Before the patient was taken to the endoscopy room, the patient allocation was provided by selection of one of these envelopes by the anaesthetist who was unaware of coding. In group I sufentanil (Sufenta 50 µg mL-1, Janssen Pharmaceutica N.V. Belgium) IN 0.5 μg kg-1. In Group II sufentanil IN 0.25 μg kg-1, and in Group III IN 0.9% NaCl (placebo) and IV 0.04 mg kg–1 midazolam (Dormicum 5 mg 5 mL-1, Roche, Istanbul, Turkey) was administered. After 15 minutes, all patients received 0.5 mg kg–1 propofol (propofol 2%, Fresenius Kabi, Istanbul, Turkey) intravenously. The sedation level was evaluated in 2-minute intervals with Ramsey Sedation Score (RSS) (Appendix 1) and BIS. During follow-up, RSS was kept between 3 and 4 and BIS was kept between 66 and 85. When above these parameters, patients received intravenous propofol at a dose of 0.25 mg kg-1.
Patient characteristics, including age, gender, weight, height, body mass index (BMI), ASA class, co-existing disease, and propofol consumption and procedure-related times (endoscopy time, spontaneous eye-opening time, recovery time) were recorded.
The NIBP was measured every 5 minutes and HR, RR, and SpO2 were evaluated continuously throughout this study, and cardiopulmonary side effects (CPSEs) were assessed. CPSEs were defined as follows: apnoea (not breathing for more than 30 s), hypoventilation (RR < 8 min-1), hypoxaemia (SpO2 < 95% with supplemental oxygen), hypotension (decrease in mean arterial pressure [MAP] more than 20% compared to baseline value or systolic arterial pressure [SAP] < 90 mm Hg), bradycardia (HR < 50 beats/min), arrhythmia, and ST changes. Moreover, the lack of immobility during the endoscopy and postprocedural nausea/vomiting were also recorded.
After completion of the procedure, the patients were transferred to the recovery room. An observer evaluated the recovery status of the patients using the modified Aldrete scoring system (Appendix 2). The total score must be equal to or greater than 9 for the patient to be discharged from the recovery room. Before their discharge, the patients were asked by the study observer to evaluate overall satisfaction of the sedation, and the pain intensity and discomfort that they experienced during the procedure (by a 100-mm visual analogue scale (VAS); 0 = no pain, 100 = severe pain). Also, the endoscopist gave a report of his/her satisfaction from the procedure, including ease of insertion of the endoscopy, immobility of the patients and patients’ compliance and tolerance to the commands. The patient and doctor satisfaction were based on a 4-point rating, including a score of 1 = poor, 2 = fair, 3 = good, and 4 = excellent.
All colonoscopies were performed by the same gastroenterologist, who had more than 5 years of experience in gastrointestinal endoscopy. All sedations were performed by the same anaesthesiologist. The patients, endoscopist, and study observer who collected and recorded the data were unaware of the sedative agents and allocation of the groups. The study protocol is thus considered double-blinded, masked to observers.
Data were analysed using the SPSS software package for Windows (Statistical Package for Social Sciences, version 15.0, SPSS Inc., Chicago, IL, USA). Patients’ baseline preoperative characteristics were reported in values of mean and standard deviation (SD) or as represented in frequencies and percentages. Categorical variables were evaluated by the use of χ2 or Fisher exact test. The variables were investigated using the Kolmogorov-Smirnov/Shapiro-Wilk test to determine the normality of data distribution. Normally distributed variables were compared with one-way ANOVA between the groups, and Tukey’s test was used for post-hoc pairwise comparisons. When the variables were not normally distributed the Kruskal-Wallis test was used, and the Mann-Whitney U test was performed to test the significance of pairwise differences using Bonferroni correction to adjust for multiple comparisons. The primary endpoint of this study was cardiopulmonary safety, and the calculations of sample sizes were based on the incidence of CPEs. Based on a previous study, the incidence of CPEs was 28.49% with midazolam and propofol sedation for colonoscopy. Power analysis with α = 0.05 and β = 0.2 for determining the 50% reduction on CPEs with IN sufentanil revealed that each group required a minimum of 28 patients. A P-value of less than 0.05 was considered to show a statistically significant difference.
RESULTS
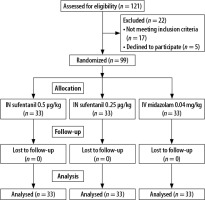
One hundred and twenty-one consecutive patients who were scheduled for elective outpatient colonoscopy were enrolled in this study. Twenty-two patients were excluded due to not meeting inclusion criteria (n = 17) and decline to participate (n = 5). Finally, 33 patients in all three groups were evaluated (Figure 1). All the patients completed this study without any complications related to the endoscopic procedure.
There were no statistically significant differences between the 3 groups concerning age, sex, height, weight, body mass index (BMI), ASA classification, and presence of co-existing disease (Table 1). The propofol dose that was required to maintain the desired level of the sedation was 52 ± 10 mg in group III, which was significantly higher than in group I (30 ± 8 mg) and group II (32 ± 8 mg) (P < 0.001, Table 1).
TABLE 1
Characteristics of the patients, propofol consumption, and BIS values
We found that endoscopy time was similar between the groups (P = 0.082, Table 2). It was observed that spontaneous eye opening time was 166.2 ± 11.8 s in group III, which was significantly longer than in group I (18.9 ± 6.4 s) and group II (17.9 ± 5.9 s) (P < 0.001, Table 2). The patients in group III had significantly longer recovery times (12.8 ± 2.3 min) compared with group I and II (3.7 ± 0.8 min and 3.9 ± 0.9 min, respectively) (P < 0.0001, Table 2). No significant difference was seen between group I and group II in propofol consumption (P = 0.397), endoscopy time (0.881), spontaneous eye opening time (0.501), and recovery time (P = 0.240) (Tables 1 and 2).
TABLE 2
Procedure-related times, adverse events, pain/discomfort, and satisfaction level
[i] SpO2 – peripheral oxygen saturation, MAP – mean arterial pressure, HR – heart rate, bpm – beats per minute, VAS – visual analogue scale (was expressed as cm). Values were expressed as mean ± SD, median (min–max) or percentage and number of patients. *Compared to group I and II. aOne-way analysis of variance test was used. bχ2 test was used. cKruskal-Wallis test was used
There was no patient who needed assisted ventilation due to respiratory depression. The HR and MAP were maintained within normal limits in all patients. Hypotension and bradycardia were not encountered during this study. We did not observe airway obstruction, arrhythmia, ST changes on ECG, permanent brain damage, or death in any patients. In 12 patients, SpO2 decreased to below 95%, which was treated by increasing the oxygen flow rate to 8 L/min, and they were all in group III (P < 0.001, Table 2). No patient had SpO2 below 90%. Lack of immobility was seen in 10 patients, and they were all in group III (P < 0.001). In group III, nausea was also seen in 2 patients, but not in group I and II. There was no nausea/vomiting in any patient.
When the RSS reached 3-4 the BIS value was 70.18 ± 6.55 in group III, which was significantly lower than group I (77.30 ± 1.83) and group II (77.18 ± 1.96) (P < 0.001). No significant difference was seen between group I and group II concerning the BIS.
The pain/discomfort experienced during the endoscopy evaluated by VAS was significantly higher in group III compared to other groups (P < 0.001), while there was no significant difference between group I and group II (P = 0.317) (Table 2). Patient satisfaction levels of the groups were close to each other (P = 0.195). Sixty-four patients classified the sedation as excellent (n = 21 in group I, n = 18 in group II, and n = 25 in group III) and the remaining 35 patients qualified as good (n = 12 in group I, n = 15 in group II, and n = 8 in group III) (Table 3). Our results showed that endoscopist satisfaction was significantly better for group I and II than for group III (P < 0.001), but there was no significant difference between group I and II (P = 0.279).
DISCUSSION
In this prospective, randomized, double-blind study, administration of the doses of 0.5 µg kg–1 and 0.25 µg kg–1 sufentanil intranasally and 0.04 mg kg–1 midazolam intravenously were compared before propofol-based sedation for colonoscopy. To our knowledge, this is the first trial to investigate this in the literature.
The main findings obtained from the present study were reported here. First, all sedation regimens were found to provide a safe and effective procedure. Second, the dose of propofol required to maintain sedation was higher in patients who received midazolam. Third, midazolam resulted in longer spontaneous eye opening and recovery times than both doses of sufentanil. Fourth, before induction of sedation with propofol, the administration of IV midazolam caused lower BIS values than IN sufentanil. Fifth, both doses of IN sufentanil had better pain control than IV midazolam during the procedure. And finally, endoscopist satisfaction was higher in the sufentanil groups than in the midazolam group.
The present study showed that both doses of sufentanil and midazolam with propofol had comparable haemodynamic safety, and cardiovascular changes were minimal. Although hypotension and bradycardia were not encountered in all groups of patients, midazolam was associated with a higher decrease in mean arterial pressure and heart rate compared to sufentanil, but this difference was not clinically significant. We also used SpO2 as a safety endpoint other than arterial blood pressure and heart rate. We found that all the patients who had a transient reduction of SpO2 below 95% were associated with midazolam, and the increase in oxygen flow rate was sufficient to improve SpO2.
It is well documented that propofol has significant consequences on haemodynamics and respiration, such as hypotension, bradycardia, and respiratory depression. Propofol causes a decrease in arterial pressure due to a drop in systemic vascular resistance, preload, and myocardial contractility, which could be attributed to a decrease in sympathetic nerve activity even at sedative doses [10, 11]. Moreover, propofol may also lead to bradycardia via interaction with atrial muscarinic cholinergic receptors and inhibition of arterial baroreflex response to hypotension [11, 12]. Respiratory depression may also be seen during propofol-based sedation in a dose-dependent manner. Large doses, rapid injection, and old age are the risk factors for propofol-induced haemodynamic alterations [10, 13, 14]. Because of the lack of analgesic efficacy when used as a single agent during endoscopic sedation, higher doses of propofol are required to maintain the desired level of sedation and increase cardiorespiratory side effects [15, 16]. The addition of an opioid and/or midazolam to propofol may help to reduce propofol consumption and, thus, the incidence of side effects [16–19].
In previous studies it has been revealed that the incidence of hypotension and bradycardia varies depending on the initial bolus dose of propofol, the definition of haemodynamic side effects, patient characteristics, monitorization techniques, and data collection methods when propofol and midazolam are used concomitantly [9, 20–22].
There are no clinical trials to directly compare the combination of propofol and IV midazolam or IN sufentanil. In a study in which sedation was performed with an IV loading dose of 2.5 mg midazolam before repeated doses of 20–40 mg propofol during colonoscopy, Delius et al. [19] reported that hypotension (< 90/50 mm Hg) and hypoxemia (SpO2 < 90%) were observed in 2 (1.7%) and 6 (5.2%) of 115 patients, respectively, but there was no bradycardia (< 50 bpm). In a prospective multicentre study, including 11,701 gastrointestinal endoscopy patients, Sieg et al. [21] found that few cases of hypotension (0.02%), bradycardia (0.05%), and hypoxaemia (0.5%) occurred for a sedation regimen consisting of 2–3 mg midazolam plus 10–20 mg propofol followed by boluses of 20–30 mg propofol. The incidence of side effects related to midazolam combined with propofol was comparable to previous studies, which also used similar propofol and midazolam doses for endoscopy sedation [19–21].
Sufentanil is a highly lipophilic opioid and is associated with an increased risk of hypoxaemia and apnoea. Deng et al. [23] reported that IV 0.1 µg kg–1 sufentanil during propofol sedation for colonoscopy is associated with respiratory depression (absent of end-tidal CO2, SpO2 < 90%, and respiratory rate < 6 breaths per minute) in approximately 30% of the patients. IN administration of sufentanil has some advantages such as ease of administration and rapid onset of action without severe cardiorespiratory side effects [5, 6, 24–26].
After the administration of IN sufentanil, time to reach peak plasma concentrations was found as 10 min in adults [6]. Therefore, in the present study we can speculate that IN sufentanil may reach peak plasma concentration at the onset of propofol induction.
In a prospective randomized study, Ayazoglu et al. [24] found that a combination of IN 0.1 µg sufentanil and propofol infusion (0.5–3 mg kg-1 h-1, 79.6 ± 9.31 mg total) during colonoscopy sedation resulted in a significant reduction in the MAP and HR values but not in SpO2. However, there was no severe hypotension, bradycardia, or hypoxaemia. A study with a limited number of patients showed that respiratory depression and haemodynamic alterations were not observed to be associated with administration of IN sufentanil 0.1–0.3 µg kg-1 combined with midazolam 5 mg [25]. In another study, Zhao et al. [26] found that the incidence of respiratory depression (hypoxemia) was 1.1%, and there were no cardiovascular side effects with the combination of propofol and IN sufentanil.
We found that midazolam was associated with longer spontaneous eye opening time and recovery time, and more than 30% propofol consumption compared to IN sufentanil. Furthermore, there was a higher incidence of motor response to colonoscope insertion or withdrawal in patients administered midazolam than in those given sufentanil. We can explain the differences between midazolam and sufentanil with both pharmacokinetics and pharmacodynamics. Although the presence of synergistic sedation between propofol and midazolam was confirmed in previous studies, neither propofol nor midazolam has analgesic activity [9, 14, 27, 28]. While synergistic interaction between propofol and sufentanil is more pronounced for analgesic efficacy, such as loss of motor response to noxious stimuli, than for hypnosis, and it has been shown clearly that providing adequate analgesia with opioids can reduce the need for sedation [28–30]. Vuyk et al. [31] reported that sufentanil increases plasma propofol concentrations due to a decrease in both the distribution and clearance of propofol. In another study, it was also found that propofol increases midazolam concentrations related to a reduction in the distribution and clearance of midazolam [32]. These pharmacokinetic interactions between propofol and sufentanil or midazolam may be another reason for the lower dose of propofol and shorter sedation-related times with the sufentanil pretreatment than the midazolam.
The results of this study show that IV midazolam administration before the induction of sedation with propofol causes lower BIS values compared to IN sufentanil although clinically similar sedation levels (RSS 3–4) are obtained in all groups. Similar to our results for moderate sedation with propofol and midazolam, the BIS level recommended by Delius et al. [20] is slightly above 73. Although there are no data about the direct effect of IN sufentanil on the BIS, it has been reported that the addition of intravenous sufentanil to propofol is associated with higher BIS values and lower propofol concentrations at loss of consciousness compared to propofol alone [33, 34]. Our findings are consistent with the studies mentioned above.
Balanced propofol sedation (BPS), which combines low doses of propofol with a benzodiazepine and/or an opioid to achieve moderate sedation, has gained increased interest recently [35]. Although the doses of propofol in the present study were comparable with previous studies, total propofol consumption was slightly lower in our study [9, 18–22, 27, 36–38]. It has been reported that BIS monitoring reduced the use of propofol in sedated patients [39]. We believe that monitoring the level of sedation by a BIS device provides a reduction in total propofol consumption compared to other studies.
The results of the present study demonstrate that pretreatment with both doses of IN sufentanil gives better pain control due to its own analgesic efficacy during colonoscopy compared to midazolam pretreatment. However, there was no difference between the groups regarding patient satisfaction. Although adequate conditions for the colonoscopy, which was defined as excellent or good regarding the satisfaction with the endoscopists, were comparable between the groups, because of the lack of immobility the number of endoscopist with excellent satisfaction was significantly lower in the midazolam group compared to the sufentanil groups.
There are several limitations in this study. We compared IN sufentanil and IV midazolam before the induction of sedation with propofol. However, the lack of an IV sufentanil group was the first limitation of the study. Secondly, the bioavailability of the intranasal sufentanil may vary person-to-person related to absorption from nasal mucosa and we did not measure plasma sufentanil levels. Thirdly, this study was performed in a single centre with a limited number of patients.
CONCLUSIONS
Our findings suggest that synergistic sedation can be achieved safely and effectively by administration of IN sufentanil or IV midazolam before propofol in patients undergoing colonoscopy. IN sufentanil, both 0.25 μg kg-1 and 0.5 μg kg-1, have better pain control and endoscopist satisfaction, and produce less respiratory depression than IV midazolam. However, IN sufentanil can be considered as a reasonable alternative to IV midazolam.