One of the initial, most noticeable, signs of ageing is a baggy, floppy eyelid. Blepharoplasty, or eyelid surgery, can be a great method to mend the eye area and rejuvenate a patient’s appearance. The usual concern for patients considering blepharoplasty surgery is the choice of anaesthesia [1].
Blepharoplasty can be performed under local infiltration anaesthesia with or without sedation (twilight anaesthesia) or general anaesthesia depending upon the surgical plan, the patient and surgeon preferences, and duration of surgery. A simple upper or lower eyelid blepharoplasty can be performed under local anaesthesia. Other more invasive procedures may need intravenous sedation, or general anaesthesia.
With local anaesthesia, the patient feels the injections of the anaesthetic and may feel pain during surgery, which necessitates the administration of more local anaesthetics [2–4].
When the local is combined with twilight anaes-thesia, the patient will not remember the experience at all, and the surgery is more comfortable. However, it may have unpredictable effects, especially in the elderly, causing respiratory depression, a loss of airway control, hypotension, prolonged recovery and sometimes restlessness [5].
General anaesthesia necessitates securing the patient’s airway either with an endotracheal tube or a laryngeal mask airway (LMA); however, both carry the disadvantage of postoperative sore throat. A nasopharyngeal airway (NPA) can be an alternative without the risk of postoperative sore throat [6].
The primary end point of our study was the incidence of sore throat with the use of NPA compared to LMA in patients receiving general anaesthesia during blepharoplasty.
Secondary endpoints included: intraoperative haemodynamic stability, failure rate, recovery time, postoperative complications and patient and surgeon satisfaction.
METHODS
After local medical ethical committee approval (dated 2nd of January 2018; approval number: FMASU R 48/2017; registered at www.clinicaltrials.gov under number NCT03510949) and obtaining written informed consent, 148 adult ASA II–III patients aged between 40 and 60 years old, who were admitted to the Department of Ophthalmology for four-lid blepharoplasty, were enrolled in this prospective randomized study. The study was conducted from February 2018 to August 2018.
Exclusion criteria were: uncontrolled hypertension (systolic blood pressure > 160 mm Hg) or ischaemic heart disease, pre-existing coagulation defects or anticoagulation medication, end stage renal disease, recent upper respiratory tract infections, asthma, chronic obstructive pulmonary disease and smoking, history of obstructive sleep apnoea, chronic drug or alcohol abuse, morbid obesity, i.e. body mass index (BMI) > 40 kg m-2 or BMI > 35 kg m-2 and experiencing obesity-related health conditions, such as high blood pressure or diabetes, and allergy to any of the drugs used in the study.
Upon arrival at the induction room, an intravenous 22 G catheter was inserted, and all patients were pre-medicated with intravenous midazolam 0.02 mg kg-1, granisetron 2 mg and ranitidine 30 mg given 15 min prior to surgery.
Standard monitors were applied, including ECG, pulse oximetry, non-invasive blood pressure monitoring and capnography.
After 5 min of preoxygenation, general anaesthesia was induced in all patients by intravenous administration of fentanyl 0.5 μg kg-1 and propofol 1–1.5 mg kg-1 titrated to a loss of verbal response.
Patients were then randomly and evenly divided into two equal groups (74 patients each) using a computerized random number generator. The allocation sequence was concealed in sequentially numbered, opaque, sealed envelopes. Each envelope was opened just before the corresponding patient’s surgery.
In group N, patients’ nasal mucosa was anaesthetized and vasoconstricted using a mixture of lidocaine and phenylephrine (1 mL of phenylephrine 1% in 3 mL of lidocaine 4%). The topical anaesthetic/vasoconstrictor solution was applied to both nasal cavities. Water-based lubricant was applied to the tip of the NPA of appropriate size. The NasoSafe NPA (Flexicare, Mountain Ash, UK) was then advanced into the more patent nostril along the septum horizontally, following the natural curvature of the floor of the nasopharyngeal cavity and rotated 90o to lie in the nasopharynx. The nasopharyngeal airway was connected to the breathing circuit using a properly sized adaptor of endotracheal tube. To identify the more patent nostril, we asked the patient to close his/her mouth and inhale through the nose with one side closed, and alternate. The patient was then asked to point out the side he/she inhaled more easily with.
In group L (control group), a mixture of water- based lubricant and lidocaine 4% gel was applied to the tip of the LMA of appropriate size. The LarySeal Blue LMA (Flexicare, Mountain Ash, UK) was introduced along the hard palate towards the hypopha-rynx until resistance was felt. Once in place, the cuff was inflated till airtightness (maximum 20 mL of air) without holding the LMA to allow it to acquire its natural position.
Anaesthesia was maintained by sevoflurane 3–4% in 5–6 L min-1 oxygen (50%). Fresh gas flow was limited to the minimal amount necessary to prevent rebreathing. Patients were allowed to breathe spontaneously using Jackson-Rees’ modification of Ayre’s t-piece. Propofol intravenous infusion 0.5–1 mg kg-1 h-1 (adjusted according to vital parameters) was administered to ensure patient immobility. Propofol infusion was used with sevoflurane to lower the concentration of inhaled sevoflurane, and to benefit from its antiemetic effect.
All patients received local infiltration anaesthesia given by the surgeon: a mixture of 1 : 1 lidocaine 2% and bupivacaine 0.5% with 1/100,000 adrenaline, with a total dose of 8–10 mL according to eyelid fullness. All patients were operated on by the same surgeon.
Mean arterial pressure (MAP), heart rate (HR), end tidal CO2 (EtCO2) and oxygen saturation (SpO2) were recorded on arrival at the induction room (baseline), after induction, and every 5 min throughout the surgery.
If significant adverse respiratory events occurred (defined as: desaturation SpO2 ≤ 85% or hypercarbia EtCO2 ≥ 50 mm Hg), the FiO2 was increased. If no response was observed, repositioning of the tube was attempted. If these steps failed, the LMA patients were converted to mechanical ventilation, while in NPA patients, the NPA was removed and replaced by LMA. If the above measures failed, a rescue endotracheal tube was inserted to replace the NPA or LMA. The incidence of failure was recorded, and new patients were recruited for the study instead.
The operative time was measured from the start of skin incision to the end of skin closure.
At the conclusion of surgery, sevoflurane and propofol infusion were discontinued, and patients inhaled 100% O2 until spontaneous recovery. Recovery time was recorded (defined as the time from discontinuation of anaesthetic drugs until response to a verbal command).
The incidence of postoperative sore throat, unsteadiness (dizziness), nausea and vomiting was recorded (in terms of absent/present) as well as patient and surgeon satisfaction.
Patient and surgeon satisfaction was assessed using a 7-point scale: 1 – extremely satisfied, 2 – satis-fied, 3 – somewhat satisfied, 4 – neither satisfied nor dissatisfied, 5 – somewhat dissatisfied, 6 – dissatisfied and 7 – extremely dissatisfied [7]. Patient satisfaction was assessed immediately before discharge from the PACU (post-anaesthesia care unit).
Data were recorded by an anaesthesia nurse who did not participate in the study.
Statistical analysis
Assuming that the incidence of sore throat is 17.5% with a laryngeal mask (LM) and 3.3% with a face-mask (FM) [8], at least 74 patients in each group were needed to detect this difference. A maxi-mum error of 5% and statistical power of 80% were considered.
Data were collected, coded, tabulated, and then analysed using the SPSS v.16.0 statistical package. Numerical variables are presented as the mean and standard deviation and were compared using a t-test, while ordinal and categorical variables are presented as the frequency and percent and were compared using the Mann-Whitney test or the c2 test as appropriate. Repeated measures variables were analysed using a repeated measures general linear model (GLM). Any difference with a P-value < 0.05 was considered statistically significant.
RESULTS
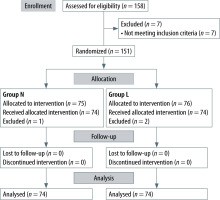
Of the 158 patients assessed for eligibility, 151 patients were enrolled in this study (5 were excluded because of uncontrollable hypertension, 1 for smoking and 1 for a history of obstructive sleep apnoea) (Figure 1). Patients excluded because of failure to maintain anaesthesia with NPA or LMA were replaced by the same number of successful patients.
Patient characteristics and operative time were comparable between the groups (Table 1).
TABLE 1
Patients’ characteristics
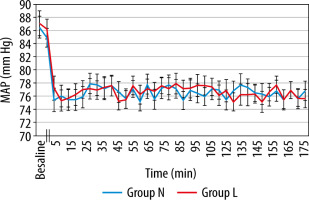
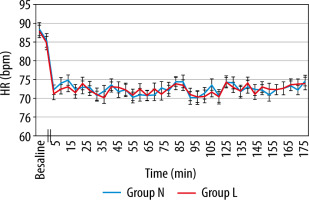
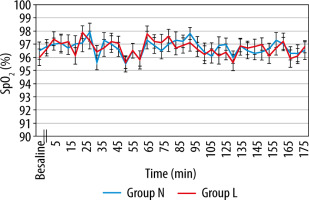
MAP and HR, in both groups, showed a slight decline after induction and thereafter compared to baseline values. However, this decrease was found to be statistically insignificant (P value > 0.05) (Figures 2 and 3). Also, there was no significant difference between the groups regarding SpO2 and EtCO2 (Figures 4 and 5).
One case was recorded as a failure in the NPA group (group N) (1.3%), in which NPA was replaced by LMA before the start of surgery. In the LMA group (group L), two cases were recorded as failures (2.7%), and the LMA was replaced by an endotracheal tube (ETT) before beginning the surgery.
Recovery time was significantly shorter in group N (10.3 ± 2.84 min) than in group L (12.6 ± 2.65 min) (P-value < 0.001). However, this difference (average 2 min) was not clinically significant.
Incidence of postoperative sore throat was significantly lower in group N (4.0%) than in group L (17.6%) with a difference of 13.5%, 95% CI (3.5–24.1%), P < 0.015. Although the incidence of unsteadiness (dizziness), nausea and vomiting was lower in group N, this was statistically insignificant (Table 2).
TABLE 2
Postoperative complications
| Parameter | Group N | Group L | P-value |
|---|---|---|---|
| Sore throat, n (%) | 3 (4.0) | 13(17.6) | 0.015 |
| Unsteadiness, n (%) | 1 (1.3) | 2 (2.7) | 1.000 |
| Nausea/vomiting, n (%) | 18 (24.3) | 22 (29.7) | 0.579 |
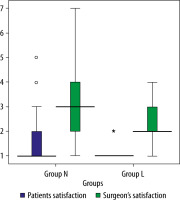
Patient and surgeon satisfaction were significantly higher in group N than in group L (Figure 6). Forty-four patients in group N scored 1 on the patient satisfaction score compared to 2 in group L. No patient in group N scored 7 compared to 1 in group L (P-value < 0.001). Regarding surgeon satisfaction, the surgeon scored 1 in most of the cases in group N (69 patients) compared to scoring 2 in most of the cases in group L (41 patients) (P-value < 0.001).
DISCUSSION
Postoperative sore throat is a common complication following general anaesthesia with a reported incidence of 62%. Although it has often been regarded as a relatively minor complication, its avoidance is of great importance to patients. The incidence of postoperative sore throat is lower with supraglottic airway devices (SADs) than with ETTs; nonetheless, postoperative sore throat still remains significant (49%) and is affected by the type of supraglottic device used [9]. Evidence suggests that there is a slight difference in the incidence of postoperative sore throat between first- and second-generation SADs, with the exception of the i-gel type, possibly due to the absence of an inflatable cuff [10].
Despite various manoeuvres to lower the incidence of sore throat using LMA, no special method has been found completely efficient [11]. Monitoring intracuff pressure can reduce laryngopharyngeal complications by 70% [12, 13]. However, other studies argued against the relation between intracuff pressure and incidence of sore throat [14], relating it more to the type of ventilation [15].
In this study, we found that the incidence of postoperative sore throat following the use of a nasopharyngeal airway was significantly lower than that with the use of a laryngeal mask airway. The pain was described by the patients as mild discomfort with no analgesics required. Pain due to NPA may be attributed to dehydration due to limited fluid intake preoperatively and the fact that we did not use humidity moisture exchangers in the gas delivery circuit.
Although many studies have compared the incidence of sore throat following endotracheal intubation and different types of LMAs, no study has compared the incidence of sore throat between LMAs and NPAs.
In a prospective study assessing postoperative sore throat following ambulatory surgery in 5264 patients, 12.1% reported sore throat. Of these patients, 45.4% had tracheal intubation, 17.5% had an LMA and 3.3% had a face mask (FM) [8].
Another study compared the incidence of sore throat after standard anaesthesia using three different methods of airway management: FM, LM, and LM with an insertion aid. The incidence of sore throat was found to be significantly less with FM (8%) than with LM when used without the insertion aid (28.5%) (P < 0.02) [16].
Alexander and Leach [17] compared the incidence of sore throat between LMA, ETT and FM in 321 patients. They concluded that the incidence of sore throat was similar for the LMA and FM.
Another study comparing the use of a reinforced laryngeal mask, a standard LM and a nasal mask in paediatric outpatient dental surgery found that the incidence of sore throat was comparable between the three groups (0, 5 and 2 patients respectively) [18].
However, other studies have shown some contradictory results concerning the incidence of sore throat with LMA. In a study comparing the laryngeal and gastro-intestinal complications of using ETT and LMA after elective orthopaedic operations in 80 patients, there was no significant difference between the two groups regarding sore throat [19].
Another study using ProSeal LMAs compared to ETTs for routine laparoscopic cholecystectomy enrolled a total of 200 patients (100 in each group). The incidence of sore throat was slightly higher in the LMA group (7%) than in the ETT group (5%) [20].
Cuffed oropharyngeal airway (COPA) was compared to LMA in 120 anaesthetized patients. The incidence of sore throat and jaw pain was found to be higher with COPA [21].
In this study, we found that the recovery time was significantly shorter when using NPA than when using LMA. This may be because LMA requires greater depth of anaesthesia [22].
In a study comparing NPA to LMA during diagnostic flexible fibre-optic bronchoscopy in children, the recovery time was significantly shorter in the NPA group, with no difference in haemodynamic status or SpO2 [23].
On the other hand, Watcha et al. [24] found no difference in recovery time between LMA and FM with a Guedel airway during paediatric myringotomy.
In the present study, we found that the incidence of failure was comparable between the groups. The procedure was recognized as a failure before the start of surgery, which allowed alternative measures to be taken without interrupting the surgical procedure. Patient and surgeon satisfaction were significantly better in the NPA group, with shorter recovery times and fewer postoperative complications.
LIMITATIONS
Using a humidifier in the gas delivery system might have lowered the incidence of sore throat with NPA. Additionally, we believe that monitoring LMA cuff pressure, as well as inflating the cuff with minimum pressure required for airtightness, might have influenced the incidence of sore throat with LMA, or at least eliminated it as a contributing factor.
A total of 10 patients were excluded from our study; only 3 of them were excluded after allocation. Although these patients may pose a potential selection bias, we believe that their small number (1 patient in group N and 2 patients in group L) compared to the relatively large sample size (74 patients in each group) likely did not affect the results significantly. We believe the results are still reliable as all the allocation envelopes were re-randomized each time an envelope was opened but the patient was excluded.