Introduction
Functional mitral regurgitation (FMR) is a common finding in patients with heart failure due to reduced ejection fraction (HFrEF) [1]. One recent study reported a 20% prevalence of severe MR in HFrEF patients under optimal medical therapy (OMT) [2]. In HFrEF, severe FMR is associated with adverse outcome in terms of more severe heart failure symptoms, more frequent rehospitalization for heart failure and increased mortality [3].
In heart failure with preserved ejection fraction (HFpEF), FMR also occurs and is associated with adverse outcomes as well, independent from coexisting atrial fibrillation [4–6].
Current guidelines approve percutaneous edge-to-edge repair in patients with symptomatic FMR despite OMT after evaluation by the heart team with a level IIb C indication [1], based on early studies [7, 8]. More recent data provide conflicting evidence whether the MitraClip (MC) procedure can attenuate heart failure and improve prognosis [9, 10]. However, in a selected population of patients in the COAPT trial [9], MC was associated with significantly improved survival (33% relative risk reduction for all-cause mortality), compared to FMR patients under OMT alone.
Catheter-based mitral annuloplasty with the Carillon system (CS) represents another option for percutaneous therapy in FMR [11]. So far, limited evidence regarding safety, feasibility and especially therapeutic effects of the CS device has been available [12, 13]. Recently, the randomized, blinded and sham-controlled Reduce-FMR trial from 2019 [14] assigned 120 patients with HFrEF and severe FMR to either OMT and a sham procedure or implantation of a CS device with blinded follow-up for mitral regurgitation volume and left ventricular dimensions. After 12 months, MR regurgitation volume decreased significantly, as well as left ventricular end-diastolic diameters. Moreover, a nonsignificant trend towards improved heart failure symptoms, reduced rehospitalization rates and mortality could be observed, although the study was neither designed nor powered for these endpoints.
Whether these competing devices, MC and CS, provide comparable effectiveness in terms of reduction of FMR and impact on prognosis has not been investigated separately so far in a head-to-head comparison study. Therefore, the aim of the present study was to describe the effectiveness in MR reduction as well as all-cause mortality after either MC or CS as endpoints in a non-randomized single-center retrospective study.
Material and methods
Patients
Inclusion criteria for this single-center retrospective study were a history of interventional therapy for severe FMR or combined etiology with predominant functional MR by either MC or CS between 2013 and 2018 at Goethe University Hospital, Frankfurt, Germany. All patients suffered from symptomatic mitral regurgitation 2+ or 3 despite optimized medical therapy according to current guidelines at the time of invasive therapy. Assignment to the individual therapy was non-randomized and based on the choice of the operating cardiologist after an interdisciplinary heart team consensus favoring an interventional approach.
Exclusion criteria for CS were a history of or indication for cardiac resynchronization therapy with a left ventricular pacing lead in the coronary sinus, calcified mitral annulus, or prior mitral valve surgery. Exclusion criteria for MC were defined according to the consensus paper on AV valve therapy of the German Cardiac Society [15].
All patients gave their written informed consent on data collection and publication. The study was approved by the local ethics committee (protocol No. 512/15). The study was conducted according to the Declaration of Helsinki of 1975.
Patients were followed by telephone calls, contact with their general practitioner or request at a civil registry office.
Methods
Echocardiographic studies at baseline and follow-up were obtained according to the guidelines of the European Society of Cardiology [16], quantifying MR in three grades of severity by integrative assessment of qualitative, semi-quantitative and quantitative parameters.
In brief, MC was performed under general anesthesia after transfemoral venous access and transseptal puncture with access to the left atrium by TOE-guided grasping of the mitral leaflets with the MitraClip device and creating a novel double mitral orifice in order to reduce the severity of FMR [7].
CS was performed in conscious sedation by transjugular retrograde venous access of the coronary sinus and placement of an individually sized device for approximation of the mitral leaflets by reducing the mitral anulus diameter. Simultaneously, coronary angiography of the left circumflex and right coronary artery was performed in order to detect any significant coronary impingement by the device [11].
Echocardiographic follow-up was performed immediately after the procedure, and 6 months thereafter. Information about the vital status 12 months after therapy and thereafter was obtained by phone calls with the patients or requests of survival to the local authorities.
Statistical analysis
All statistical analysis was performed with SPSS 13.0.
If not depicted otherwise, continuous variables are given as mean ± standard error, categorical variables as frequencies and percentages. Differences between variables were calculated using the χ2 test for categorical variables and one-way ANOVA test for continuous variables. Multivariate analysis was performed by stepwise forwards Cox proportional hazards ratio regression analysis. All p-values are reported two-sided with a level of significance of p < 0.05. The Kaplan-Meier estimator was used for survival analysis and the log rank test.
Results
N = 117 patients underwent treatment with the MitraClip, n = 37 patients were treated with the Carillon. No patients were lost to follow-up.
Patient characteristics and procedural data
Baseline characteristics of the two groups are depicted in Table I.
Table I
Baseline characteristics
[i] Data are presented as n (%) or mean ± standard deviation. ACE – angiotensin-converting enzyme inhibitor, ARB – angiotensin-receptor blocker, BMI – body mass index, BP – blood pressure, CABG – coronary artery bypass graft surgery, EROA – effective regurgitant orifice area, GFR – glomerular filtration rate estimated by MDRD formula, ICD – implantable cardioverter-defibrillator, LVEF – left ventricular ejection fraction, LVEDD – left ventricular end-diastolic diameter, MRA – mineralocorticoid receptor antagonist, NT-proBNP – N-terminal pro-B-type natriuretic peptide, NYHA – New York Heart Association, PAP – pulmonary artery pressure.
Patients in the CS group were significantly more often male, with a lower number of implanted ICDs. Ischemic heart disease was the leading cause of functional MR in both groups. Guideline-directed heart failure medication was uptitrated to the maximum tolerable dose in both groups before device implantation.
Both severity of FMR and degree of heart failure were more pronounced in the MC group, regarding lower left ventricle ejection fraction (LVEF) and higher NT-proBNP values as well as slightly but significantly higher grades of MR reflected by vena contracta width, effective regurgitant orifice area (EROA), regurgitant volume and left ventricular end-diastolic diameter (LVEDD) (Table I) in the MC group.
Device implantation succeeded in 112 (95.7%) patients of the MC group and 31 (83.8%) patients of the CS group (p = 0.024). The lower rate of procedural success in the CS group was due to impingement of a relevant branch of the left circumflex artery, dissection of the coronary sinus or unavailability of a suitable device for individual CS anatomy.
Echocardiographic follow-up
First echocardiographic follow-up after therapy was obtained in 97 (86.8%) patients of the MC group after a median of 6 days (IQR = 11.5 days) and 26 (83.9%) patients of the CS group (Table II) after a median of 28.5 days (IQR = 89.8 days) (Table II).
Table II
Echocardiographic follow-up
In both groups, MR degree, EROA and regurgitation volume as well as systolic pulmonary artery pressure were significantly reduced. Moreover, LVEDD diminished significantly in both groups as a marker of inverse remodeling. Although the grade of FMR was statistically significant between the MC and CS group in this short-term follow-up, no significant differences between the deltas of the single echocardiographic parameters vena contracta width, EROA and systolic pulmonary artery pressure between the MC and CS group were detected (Table II).
Echocardiographic follow-up at 6 months was available in 38 patients of the MC group (33.9%) and 11 patients of the CS group (35.5%) after a median of 189 (IQR = 51.5) days and 200 (IQR 48) days, respectively (Table II).
In both groups, grade of MR as well as vena contracta width, EROA and regurgitation volume remained reduced compared to baseline parameters, with an increase in LVEDD in the MC group. Systolic PA pressure showed a significantly more pronounced reduction in the CS group, compared to the MC group (Table II).
For the whole study population as well as the subgroup of patients treated with MC, no predictors of procedural success defined as reduction of FMR by at least one grade could be detected. Only in the subgroup analysis of the CS group was a higher grade of pre-interventional tricuspid regurgitation significantly associated with therapeutic failure despite proper device implantation (patients with MR reduction by one grade or more: TR 1.1 ±0.7 versus a TR of 1.9 ±0.6 in patients with MR reduction of less than one grade, p = 0.005).
Survival during follow-up
Within 12 months after therapy, 14 patients of the CS group and 73 patients of the MC group died, accounting for a 1-year mortality of 45.2% and 65.2%, respectively (Figure 1).
Figure 1
Kaplan-Meier plot of cumulative survival in years for the MitraClip group (green line) and Carillon group (blue line). Log rank p was 0.001
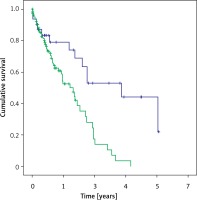
During follow-up of up to 5 years, survival curves diverged significantly, with a median survival of 1.66 years in the MC group and 3.92 years in the Carillon group, p = 0.01 in log rank testing. Five-year survival rates were 0% in the MC group and 6.5% in the Carillon group.
Univariate analysis of baseline demographic parameters and survival revealed that end stage renal disease necessitating hemodialysis was significantly associated with adverse outcome in the MC group. Only 2 of the 10 MC patients with hemodialysis at baseline reached median survival, whereas 8 patients died before (p = 0.057).
In multivariate analysis including all parameters significantly different between the Carillon and MitraClip groups at baseline, only left ventricular end-diastolic diameter persisted as an independent predictor of mortality during follow-up (hazard ratio = 1.06, 95% confidence interval: 1.015–1.056, p = 0.012).
Discussion
This is the first study to describe the differential effects of MC or CS on the degree of functional mitral regurgitation and long-term survival in an unselected, non-randomized all-comers population. However, due to the limitations described below, its contribution is predominantly hypothesis-generating.
Our study cohort displays similar characteristics comparable to populations of FMR patients examined in large contemporary randomized trials [9, 10]. Moreover, the reduction of MR severity corresponds to the therapeutic effects of both devices described before [9, 14].
Due to the limited number of follow-up examinations, we cannot provide a complete course of FMR grade especially for the CS group, which might be of particular interest as the CS system is well described to bear later responsiveness in FMR reduction, potentially dependent on the angle between the coronary sinus and mitral valve plane [17].
The 1-year mortality rates of 45.2% and 65.2% in the CS and MC groups of our study are higher than all-cause mortality seen in COAPT with 19.1% in the device group and 23.2% in the control group. A recent analysis of pooled Carillon data reports 83.6% 1-year survival and 56.2% survival after 4 and 5 years [18].
This difference in mortality can only partially be explained by the introduction of angiotensin receptor neprilysin inhibitor (ARNI) for pharmacological therapy in contemporary heart failure patients [19–21], its effects on FMR [22] or emerging usage of defibrillator/resynchronization therapy [23]. However, a mean EuroSCORE II [24] of 10.7 in patients with pronounced left ventricular remodeling reflected by a mean LVEF of 37% and a mean LVEDD of 61 mm with FMR grade 3 displays a cardiac high-risk collective at the time of treatment between 2013 and 2018 that might not have been included in a contemporary randomized trial.
The striking difference in all-cause mortality between the MC and CS groups during the long-term follow-up has to be interpreted with caution. As pointed out before, only limited numbers of follow-up echocardiographic examinations are available (Table II). In a subset of CS patients (n = 9) with long-term follow-up up to 2 years, FMR remained reduced to an average MI grade of 1.94 ±0.63. We cannot provide similar follow-up data for the MC group. As the vast majority of MC and CS patients did not undergo prespecified echocardiographic long-term follow-up, we cannot exclude that a substantial proportion of patients from any therapy group deteriorated in FMR grade over time with a potential adverse impact on survival. Due to the limited follow-up in our cohort, we cannot reproduce the beneficial impact of successful CS therapy within short-term follow-up on long-term survival as recently described [18]. However, in multivariate analysis, left ventricular end diastolic diameter persisted as an independent predictor of mortality, indicating that the differences in mortality are mainly driven by selection bias of patients, with more pronounced heart failure in the MC group.
This study bears a number of substantial limitations to be taken into account when interpreting the data:
Due to the retrospective design of the study with uncoordinated echocardiographic follow-up, we were not able to provide full information about the course of mitral regurgitation after therapy in all patients for both groups. Allocation of patients to either MC or CS was not randomized but based on operators’ choice, potentially influenced by personal experience and facilities’ capacity for either MC or CS at the time of therapy. Both therapy groups included patients first to be treated with MC and CS at the institution. We can therefore not exclude that contemporary patients might benefit more from therapy as the learning curve for either therapy has to be taken into account for procedural success.
The impact of both therapies on the degree and course of heart failure is an important issue. Due to the lack of data, we are not able to provide important heart failure parameters such as NYHA class, NT-proBNP levels or 6-minute walking tests during follow-up.
We can only provide overall mortality and have no insights into the causes of death, especially cardiovascular mortality. The sample size of the study population does not allow robust conclusions about the differential impact on mortality by the examined treatments for MR.
In conclusion, the less invasive Carillon approach appears to bear comparable beneficial short-term effects on the grade of FMR and LV remodeling and a comparable 1-year survival outcome, compared to the MitraClip.
As interventional mitral valve therapies are emerging, differential effects and long-term clinical and survival efficacies of evidence-based device therapies such as the MitraClip and Carillon have to be investigated in properly randomized upcoming clinical trials.


