Over the last decade, awake craniotomy (AC) has become a commonly performed neurosurgical procedure for resection of brain tumors in eloquent areas [1]. By cortical and subcortical mapping functional areas can be localized in the awake patient, allowing maximal tumor resection up to functional boundaries [2, 3]. AC has been shown to result in fewer late neurological deficits [4, 5], shorter length of hospital stay [6, 7], and longer overall survival [5], the latter correlating directly with the extent of tumor resection [7]. However, the anesthesiological management for AC shows high variations between centers performing this procedure. So far, different anesthesiological approaches have been reported: asleep-awake-asleep (AAA), asleep-awake (AA), monitored anesthesia care (MAC) and conscious sedation (CS) [1, 6–16]. Not only applied techniques, but also used medication and airway devices, differ or have underlain changes and improvements over time [14, 17, 18]. Successful intraoperative mapping requires the patient to be fully awake and cooperative, challenging the anesthesiologist in charge. Extended knowledge in the field of neuroanesthesia, of used medication and of airway management is therefore required by the performing anesthesiologist. Strategies including local anesthesia for scalp blockade, advanced airway management, dedicated sedation protocols and skillful management of hemo-dynamics are essential [15].
At our institution, the asleep-awake technique has been used since 2006. By reviewing our experience with this technique we aimed to contribute to a further standardization of neuroanesthetic management in AC and thus increase patients’ safety.
METHODS
Study design and data collection
This observational retrospective single-center study was carried out in accordance with the Declaration of Helsinki. Ethical approval was provided by the local Ethics Committee of the UCT – University Hospital Frankfurt, Frankfurt, Germany (approval number SNO/10/2018). All patients of this study gave their written informed consent to the analysis of their clinical data and tumor tissue.
Between September 1st, 2006 and May 31st, 2018 data of all patients undergoing AC were collected and anonymized. Clinical information, including anesthesiological management, that had been extracted from institutional protocols, as well as surgical reports, was evaluated for each patient.
In our center, dexmedetomidine (DEX) was introduced as an adjunct for sedation in May 2016. Therefore, all patients who underwent AC since then were allocated to the DEX group (DG) and received propofol, remifentanil and DEX. Patients receiving AC before May 2016 were assigned to the standard group (SG), receiving anesthesia/sedation by propofol and remifentanil alone.
For all drug dosage, cumulative doses and periods of administration were registered from the beginning until the end of the surgical procedure, the latter being defined as the patient’s arrival at the intensive care unit (ICU).
Four different time periods/durations were documented and analyzed. Time to arousal characterized the duration between discontinuing the anesthetic medication until the patient was fully awake and cooperative. The awake phase represented the period during which the patient was awake while undergoing the surgery, i.e. either until reintubation was necessary or until the end of surgery. The duration of surgery was defined as the time between incision and suture. The total duration characterized the total perioperative period from arrival in the operating room until leaving the operating room for transport to the ICU.
Objects of primary airway evaluation were Mallampati classification, thyromental distance (according to Patil) and extent of reclination of the head. It was the choice of the responsible anesthetist whether to use a standard Macintosh endotracheal tube, laryngeal mask or laryngeal tube.
Anesthetic management for the asleep-awake technique
All patients underwent standard noninvasive monitoring (arterial blood pressure measurement, pulse oxi-metry and electrocardiography). After preoxygenation via a face mask for at least 3 minutes, sedation was induced either with fentanyl (Rotexmedica GmbH, Trittau, Germany) bolus (2–4 µg kg-1) or remifentanil (Aspen Pharma Trading Limited, Dublin, Ireland) as continuous infusion (0.25 µg kg-1 min-1), followed by propofol (Fresenius Kabi Deutschland GmbH, Bad Homburg, Germany) (3–5 mg kg-1 h-1). Then, either a laryngeal mask (LMA), laryngeal tube suction (LTS) or an endotracheal tube (ETT) was inserted for airway control. Patients intubated received rocuronium (Inresa Arzneimittel GmbH, Freiburg im Breisgau, Germany) (0.6 mg kg-1). Thereafter, sedation was provided by intravenous infusions of propofol (4–9 mg kg-1 h-1) and remifentanil (0.14–0.4 µg kg-1 min-1) to maintain anesthesia, and intra-arterial and a central venous catheters were placed. Sterofundin ISO (B. Braun, Melsungen, Germany) was used as continuous balanced crystalloid infusion as standard. If necessary, noradrenaline was administered to maintain mean arterial pressure above 70 mm Hg (9.3 kPa).
All patients were placed in a semi-lateral position, with the head fixed in a 3-pin skull clamp. Once positioned, infiltration of the pin sites, the operation field and an additional scalp nerve block were set, using ropivacaine (Fresenius Kabi) 40 mL 0.75% (300 mg) mixed with 200 µg of adrenaline (InfectoPharm Arzneimittel und Consilium GmbH, Heppenheim, Germany).
Surgical preparation, draping and a final control using the surgical safety checklist followed. After craniotomy, the dura was first soaked with 1% lidocaine (Astra Zeneca GmbH, Wedel, Germany) and then opened. Now, and in accordance with the surgeon, general anesthesia was discontinued. As soon as the patient breathed spontaneously and showed signs of being awake, the LMA, LTS or ETT was removed.
In patients of the DG, DEX was initiated simultaneously to the discontinuation of propofol infusion at the time of bone flap removal. DEX was administered until the end of dura opening.
Cooperative patients underwent extensive cortical and subcortical mapping throughout the whole surgical procedure in order to define all functional boundaries of tumor resection.
Any intraoperative seizures were treated by pouring cold water on the brain and, if necessary, additionally by intravenous administration of levetiracetam (Declimed GmbH, Hamburg, Germany). Patients showing inadequate awakening, persistent seizures or insufficient spontaneous breathing were not extubated. In case of bradypnea, hypoventilation, central respiratory depression or CO2 accumulation, reintubation was performed with the device of choice by the responsible anesthetist.
Intraoperative analgesia was performed by continuous administration of remifentanil and bolus injection of metamizole. Once the surgical procedure was completed, patients were brought awake and spontaneously breathing to the ICU for postoperative surveillance.
Statistical analyses
All statistical analyses were performed using SigmaPlot 12 (Systat Software GmbH, Erkrath, Germany). Depending on the distribution of data (determined via the Shapiro-Wilk test), Student’s t-test or the Mann-Whitney rank sum test was used to compare data. The c2 or Fisher exact test was used to detect differences between the proportions of patients with respect to categorical data. Values were expressed as number (count, percent), mean ± SD, or median (25th and 75th percentiles, IQR), as appropriate. All tests were two-sided, and the level of significance was set to P < 0.05.
RESULTS
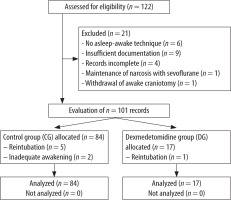
In total, 122 patients underwent AC between September 1st, 2006 and May 31st, 2018 in our Department of Neurosurgery. Due to missing data (Figure 1), 21 patients had to be excluded from the study, resulting in a final analysis of 101 patients.
After changing our standard operating procedure in May 2016 all patients undergoing AC received DEX as an adjunct, resulting in 17 patients allocated to the DG. Although the number of patients in the SG and DG differed, there were no statistically significant differences in their baseline characteristics (Table 1).
TABLE 1
Demographic data
Use of anesthetics
All anesthetics used during the surgical procedure are listed in Table 2. Compared to patients in the SG, patients in the DG showed a significantly shorter period of propofol administration (P = 0.008). However, the total dose of propofol was nearly identical in both groups (P = 0.789), as propofol was administered at a significantly increased rate in the DG compared to the SG (P < 0.001) (Table 2). Patients in both groups received propofol during the awake phase on an individual basis due to anxious behavior.
TABLE 2
Drug administration
SG – standard group, DG – dexmedetomidine group, Dex. – dexmedetomidine, GA – general anesthesia: phase of procedure between induction of narcosis until intraoperative awakening. Awake – phase of procedure
The period of remifentanil administration did not differ between groups (P = 0.484). Nevertheless, the patients in the DG received a cumulatively higher dose (P = 0.023) as a result of an increased infusion rate (P < 0.001). In the awakening phase, analgesia with remifentanil was performed in the SG over a significantly longer period of time (P = 0.032), but there was still no significant difference regarding total dosage comparing both groups (P = 0.464) (Table 2). Fentanyl as an opioid for induction of anesthesia was used in both groups. Its dosage also did not differ between groups (P = 0.446).
Intraoperative complications
In total, 12 patients (12%) showed intraoperative seizures, of whom eleven patients (13%) were treated in the SG and one patient (6%) in the DG, which did not result in significant group difference (P = 0.402).
In six patients (6%), all from the SG group, initial intubation difficulties occurred; in four patients the airway was finally secured with a laryngeal mask. In these four patients the preoperative airway evaluation revealed a Cormack and Lehane score of III. In one case an ETT was used and in one case a LTS. These two patients presented an unexpected difficult airway. During surgery, failed extubation occurred in two patients (2%), both treated in the SG. Most importantly, reintubation was required in six patients (6%), of whom five were in the SG and one in the DG. For reintubation LMA were used in five (83%) cases, and in one patient an ETT was inserted.
Time periods
Analyzing the duration of different periods within surgery revealed no significant differences between the two groups (Table 3). A non-significantly shorter duration of arousal was found in the DG (SG vs. DG; 15 min. vs. 12 min.; P = 0.271).
TABLE 3
Durations of different intraoperative periods
Airway management
No difference was found between the two groups regarding Mallampati classification, thyromental distance according to Patil (P = 0.554) or extent of reclination of the head (P = 0.827). Reintubation was necessary in three patients due to refractory intraoperative seizure and in three patients due to respiratory exhaustion. No reintubation was requested by any patient.
Table 4 shows characteristics of airway management including used devices for securing the airway.
TABLE 4
Airway management
DISCUSSION
For this observation over a period of 12 years, data from 101 patients undergoing AC for brain tumor surgery were evaluated. The anaesthesiological management of AA using propofol and remifentanil, optionally in combination with DEX, enabled sufficient protection of the respiratory tract in all patients. Further, all patients showed a rapid arousal for cortical mapping. The entire procedure was well tolerated by all patients and a precise neurological monitoring of the neurological condition was achieved as the patients stayed awake for the rest of the surgery.
Considerations regarding different anesthesiological techniques
According to the literature, different anesthetic techniques have been used for AC during the last decades. However, data for the use of GA for cerebral tumor surgery in eloquent areas show worse outcomes and higher total inpatient costs compared to awake craniotomies [19].
A meta-analysis evaluating the usefulness of intraoperative mapping confirmed that its use resulted in a reduction in morbidity and in an increase in extent of tumor resection compared to a cohort undergoing surgery without intraoperative mapping [4]. However, in order to perform reliable intraoperative mapping an awake and cooperative patient is needed. This can be achieved in different ways. For the asleep-awake-asleep technique, patients are under GA at the beginning, requiring a secure airway and controlled ventilation [8, 9, 15, 16]. On the one hand, this may be advantageous for the patient, not experiencing the stressful phase from placing on the table, clamping in the Mayfield holder, preparation for craniotomy itself and, later on, the intraoperative phase after cortical mapping. On the other hand, it may be difficult to re-secure the airway of a patient positioned in the Mayfield clamp because access to the patient’s head is often restricted and reclination of the head is impossible.
Different possibilities for securing the airway during AC have been evaluated. According to the meta-analysis of Stevanovic et al. most common is the LM, facemask or nasal cannula [15]. At our department, LMA and endotracheal tubes are the most commonly used devices, but in recent years there has been a trend towards more frequent use of LMA (Table 4). Furthermore, GA and reintubation immediately after tumor resection restricts prompt postoperative neurological examination. A first postoperative neurological examination will then only be possible after extubation in the intensive care unit. This leads to a variable time frame without any surveillance of neurological deficits. In our study, six patients needed reintubation, which could be performed without any complications.
Another approach to perform AC is monitored anesthesia care or the conscious sedation technique. During this procedure patients are kept in different levels of sedation with preserved spontaneous breathing [8, 9, 13, 15, 20], and therefore no airway safety is required. Nevertheless, keeping the patient in (deep) sedation with sufficient spontaneous breathing is challenging. An increase in paCO2 by bradypnea may result in raised intracerebral pressure leading to brain swelling and herniation after dural incision and opening. The patient’s hyperventilation may reduce this effect, but this requires the patient to be cooperative [7]. Moreover, since the patient is awake during the whole procedure, even in sedation, he might experience the surgical procedure as an increased stress level.
In our department, we have brought together the benefits of different techniques to achieve the best possible advantages in terms of neurological monitoring and patient comfort. The AA technique allows a stressless insertion of the central venous catheter and artery line, as well as positioning and placing the patient in the Mayfield clamp. Furthermore, controlled ventilation allows one to control the end-tidal CO2, which allows for a compensatory mechanism to react on brain swelling after dura incision. As mentioned earlier, the patients’ wakefulness after tumor resection provides an excellent possibility to assess his or her neurological condition. In addition, the prolonged awake period, which was a median of 210.0 minutes (IQR 165.0/262.5) in our studied cohort, was very well tolerated by all patients.
Medication
Besides techniques, also used medication differs between centers performing AC, and several different combinations have been reviewed [7–9, 11–13]. At an optimum, drugs should have a fast on- and offset, should not influence the patients’ neurocognitive function during intraoperative mapping, and should not have a depressive effect on the cardiovascular and respiratory system.
If GA is part of the performed technique, propofol and remifentanil [7, 8, 11, 12, 15, 16, 21] are most commonly used while volatile anesthetics and fentanyl are less common [9]. Fentanyl, as well as alfentanil and sufentanil, showed a proconvulsive effect, making remifentanil the opioid of choice for AC [13, 16]. Moreover, intravenous anesthesia by manually controlled infusion (MCI) should be rejected in favor of target-controlled infusion (TCI) when conducting AC. As demonstrated by Wang et al. TCI group patients have a significantly shorter intraoperative awakening time. The highest bispectral index score (BIS) values appeared during the awake phase in the TCI group and the mean heart rate and arterial pressure were more stable in the TCI group in comparison to the MCI group [16].
Using BIS monitoring during AC may be beneficial to shorten post-anesthetic recovery due to reduced cumulative doses of anesthetics. Conte et al. found higher BIS values at the end of the asleep phase associated with shorter time to LMA removal [21].
Most recently, DEX as an anxiolytic, sedative and anesthetic drug has been introduced in AC [13] and was used as an additive in the beginning [22, 23]. DEX is a lipophilic imidazole derivate that selectively binds on pre- and postsynaptic α2-receptors and acts as an agonist. It has an opioid-sparing effect and therefore results in less respiratory distress than propofol. Moreover, it has not only been demonstrated that DEX allows successful electrocorticography (ECoG) monitoring during AC [14], but also that DEX leads to a shorter duration of arousal compared to propofol [24], as could be confirmed by our data. Thus, patients of the DG were awake after 12.0 min (IQR 10.0/16.5) compared to 15.0 min (IQR 10.0/20.0) in patients of the SG.
Limitations
The main limitation of this study is its retrospective nature. With the introduction of DEX only recently, the number of patients receiving DEX did finally not allow for a powerful statistical analysis. Therefore, and above all due to patients’ heterogenous characteristics such as tumor histology, tumor location, tumor volume, age and clinical state, matching of patients receiving DEX with patients in the control group was not possible. Moreover, although surgery was conducted by the same dedicated team of neurosurgeons over the complete study period, different anesthesiological teams were involved in patients’ treatment. We propose to perform a large multicenter randomized controlled trial in order to further evaluate the AA technique in combination with DEX. This would allow us to correct for the patients’ inherent characteristics and eliminate possible bias.
CONCLUSIONS
All mentioned approaches have contributed to optimize the procedure for AC over the years. A notable benefit of the AA technique is the ability to assess the patients’ neurological state from arousal until the end of surgery and immediately thereafter. Thus, planned reintubation is no longer necessary, avoiding the risk of airway incidents in this context. The AA technique is safe and well tolerated by all patients. Furthermore, AA is beneficial to AAA or MAC, due to reduced stress during the initial phase of surgery. Using DEX as an adjunct to propofol and remifentanil the duration of patients’ arousal can be decreased under successful ECoG monitoring, and without influencing patients’ respiratory state, resulting in increased patients’ safety.