Introduction
Colorectal laterally spreading tumors (LSTs) were first described in 1993 [1]. The lesions represent a special subtype of colorectal neoplasms, which are defined as more than 10 mm in diameter, and grow laterally and circumferentially typically rather than vertically along the wall of the colorectum [2]. The LSTs are subdivided into granular (LST-G) and non-granular (LST-NG) types based on the endoscopic finding, and the granular type consists of homogeneous (LST-G-H) or nodular-mixed (LST-G-M) morphology while the non-granular type is flat-elevated (LST-NG-F) or pseudo-depressed (LST-NG-PD) [3]. The LSTs have been recognized as a significant precursor of advanced colorectal cancer (CRC) [4]. In addition, LSTs are prone to be overlooked in colonoscopy because of the unique morphological features, and are technically challenging in endoscopic resection (ER), which probably causes incomplete en bloc resection and local recurrence. Hence, the endoscopic diagnosis and treatment of LSTs are greatly essential for colorectal cancer prevention. To date, ER, including endoscopic mucosal resection (EMR) and endoscopic submucosal dissection (ESD), has been indicated for the lesions. The ESD has many predominant advantages over EMR for large lesions [5]. A variety of high-frequency electric knives have been developed for ESD, such as the Hook knife [6], insulation-tipped diathermic knife (IT knife) [7], Flush knife [8], Flex knife [9], bipolar needle knife (B-knife) [10], SB knife [11], and Triangle tip knife (TT knife) [12], etc. The various knives have their strengths as well as shortcomings. They need to be used in combination in most cases. The Dual knife is a relatively novel type of ESD knife produced in 2009, which can be used for completing the whole procedure of ESD.
We report herein the results of a clinical study in which only the Dual knife was used for the ESD procedure of colorectal LSTs.
Aim
Colorectal LSTs are a special subtype of colorectal neoplasms. We aimed to evaluate the clinical features and outcomes of ESD for colorectal LSTs only using the Dual knife from our experience.
Material and methods
Participants
The study subjects were 162 patients (each patient had 1 lesion) with colorectal LSTs treated by ESD (only using the Dual knife) by a single endoscopist at our hospital between June 2015 and January 2018. The study protocol was conducted in accordance with the tenets of the revised Declaration of Helsinki (1983) and was approved by the ethics committee of our institution. Informed consent was obtained from all the patients.
Bowel preparation
Bowel preparation involved a patient drinking 2–3 l of polyethylene glycol (PEG) electrolyte solution before the procedure, then an assessment of the quality of bowel cleanliness on the basis of the Boston Bowel Preparation Scale (BBPS); a BBPS score ≥ 5 was satisfactory [13].
Settings of colorectal ESD
All procedures were performed using a colonoscope (CF-H260; Olympus, Japan) with a transparent cap (D-201-13404; Olympus, Japan) and a gastroscope (GIF-H260J; Olympus, Japan) with a distal attachment (ND-201-11804; Olympus, Japan). A Dual knife (KD-650Q; Olympus, Japan) was used during all ESD procedures. A high-frequency electrosurgical generator (VIO 200S; ERBE, Germany) was set up. An injection needle (NM-200L-0423; Olympus, Japan) was used to lift up the lesion. Hot biopsy forceps (FD-1U-1; Olympus, Japan) or endoscopic metal clips (HX-610-090L; Olympus, Japan) were used to stop bleeding. A mixture of 100 ml of 10% glycerol solution containing 2 ml of methylthioninium chloride and 1 mg of 0.002% epinephrine was used as the injection solution [14].
The ESD procedures
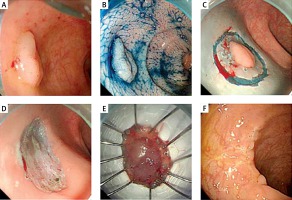
The ESD was carried out as follows: 1) Marking: It could assist to indicate the margin of lesions during the procedure. However, marking was not usually used in the colorectal LSTs because the range of lesions was generally clear (Photo 1 A). 2) Submucosal injection: the injection solution mentioned above is locally injected into the submucosa of a lesion through the injection needle (Photo 1 B). An additional submucosal injection was repeated as needed. 3) Incising the mucosa: After submucosal injection, the mucosal layer around the lesion (3–5 mm outside lesion edge) was incised circumferentially using a Dual knife with an endo-cut Q mode (effect 3, duration 2, interval 2) (Photo 1 C). 4) Dissecting the submucosa: The submucosal layer beneath the lesion was then dissected using the Dual knife with a force-coagulation mode (effect 3, 40 W). 5) Treatment of the post-ESD resection site: careful examination and coagulation of exposed visible vessels (with little or no overlying submucosa) in the post-ESD site with hot biopsy forceps (Photo 1 D). Endoscopic metal clips were used for suturing the resection bed as appropriate. Finally, the lesion was resected completely and retrieved endoscopically.
Photo 1
Endoscopic submucosal dissection (ESD) procedures, performed with a Dual knife. A – A 20 mm flat-elevated nongranular type of laterally spreading tumor (LST-NG-F) located in the rectum. B – Lesion margins were delineated with 0.4% indigo-carmine spraying before ESD. C – Following injection of 10% glycerol solution containing methylthioninium chloride and 0.002% epinephrine into the submucosal layer, a circumferential incision was made using the Dual knife. D – An ESD was then carried out using the Dual knife. The ulcer bed is represented here after the successful en bloc resection. E – The resected specimen was 20 mm × 20 mm in diameter and histology revealed a tubular adenoma. F – Follow-up endoscopy 6 months after ESD showed no local persistence or recurrence
Management after ESD
All patients routinely underwent the post-operative hospital stay, the fasting period and infusion. A reasonable diet was given after confirming the absence of inflammatory findings, such as fever, abdominal pain, and level of serum C-reactive protein (CRP), while inspecting delayed bleeding and delayed perforation. Both the length of the fasting period and hospitalization should be considered with respect to each specific situation. All the patients with colorectal ESD received a follow-up endoscopy 6 to 31 months after the resection to check for any local recurrence on the basis of endoscopic and pathological assessments (Photo 1 F).
Histopathology
Resected specimens were pinned flat on the corkboard with sufficient tension (Photo 1 E). Subsequently, the specimens were immersed in 10% formalin, embedded in paraffin, serially sectioned into pieces at intervals of 2 mm, and stained with hematoxylin and eosin. Finally, the following parameters – the histopathological type, resection margins (lateral and basal) of the lesions, and the invasive depth of the lesions – were evaluated by an expert pathologist with regard to the Vienna classification of gastrointestinal epithelial neoplasia [15].
Variables investigated
The variables were investigated as follows: patients’ clinical features (including sex and age), tumor location, tumor size, pathologic type, LST subtypes, ESD procedure times, en bloc rate and complications (including bleeding and perforation). The local recurrence rate after complete en bloc resection was also assessed.
Results
A total of 162 patients with colorectal LSTs treated by ESD using the Dual knife were enrolled in the study. The clinicopathological characteristics and outcome of ESD treatment for LSTs in this study are presented in Table I. The mean age of the patients was 53.9 years (range: 24–81 years), with a male to female ratio of 1.35 : 1 (93 /69). The mean diameter of lesions was 46 mm (12–100 mm), including 125 cases that were more than 20 mm in diameter and 37 cases that were less than 20 mm. The distribution of LST subtypes was 61 LST-G-H, 57 LST-G-M, 39 LST-NG-F, and 5 LST-NG-PD. Overall, the most common location was the rectum (74, 45.7%), followed by the descending colon (31, 19.1%), the sigmoid colon (24, 14.8%), the transverse colon (17, 10.5%), the ascending colon (11, 6.8%) and cecum (5, 3.1%). The most common histological type was tubular adenoma with 63 (38.9%) cases, followed by villous adenoma with 34 (21.0%) cases, tubulovillous adenoma with 28 (17.3%) cases, high-grade intraepithelial neoplasia (HGIN) with 24 (14.8%) cases, inflammatory polyp with 9 (5.6%) cases, serrated polyp with 3 (1.8%) cases, and submucosal invasive carcinoma with 1 (0.6%) case (additional radical procedures were reserved for them) (Table I). In 161 patients, we obtained complete dissection of the LST by using the Dual knife and the complete histological evaluation. The mean operating time was 56 min (range: 12–135 min) (Table I). One perforation (0.6%) with 3 mm × 3 mm occurred during the procedure, and the patient recovered after being treated with endoscopic metal clips. One case of delayed bleeding (0.6%) was observed on postoperative day (POD) 3, and the patient underwent endoscopic clipping and achieved successful hemostasis. Blood transfusion was not required for the patient. No case of local persistence and recurrence or metastasis was observed at a follow-up endoscopy 6 to 31 months after the en bloc resection.
Table I
Clinicopathological features and outcomes of ESD treatment for colorectal LSTs with Dual knife only in this study
Discussion
Colorectal LSTs are classified into LST-Gs and LST-NGs, according to macroscopic findings, which are an important kind of precursor lesions of colorectal carcinoma. Furthermore, the carcinoma incidence rate and the submucosal invasion rate were reported to be higher in LST-NG than in LST-G [4]. Thus, we recognize that morphological classification for LST is significant for planning appropriate treatment strategies for patients. The EMR, piecemeal EMR, or ESD for such LSTs is an effective treatment. Piecemeal EMR is associated with higher local recurrence rates, ranging from 10% to 23.5%, as compared with EMR, whose local recurrence rates vary from 0% to 9.1% for en bloc resections, and from 0% to 3% following ESD [16–18]. Moreover, LST-NGs and residual or locally recurrent lesions (after prior endoscopic treatment) are considered technically difficult to treat because of the presence of submucosal fibrosis. Consequently, for colorectal LSTs, especially larger than 20 mm in diameter, ESD is a greatly appropriate choice that enables the en bloc resection regardless of the size of the lesion, accurate histological evaluation, and a reduced possibility of residual lesions or local recurrence.
The ESD has been the first-line endoscopic treatment for superficial gastrointestinal tract tumors [19]. The devices applied in ESD are crucial for performing the procedure successfully. Nowadays, a large number of knives have been developed for ESD. Representative knives that are frequently applied in colorectal ESD are as follows: Hook knife, IT knife, Flush knife, Flex knife, Dual knife, SB knife, B-knife, and Triangle tip knife. Of these, the Dual knife is one of the most constantly used knives and has some advantageous functions including incision, dissection, and hemostasis. The knife is equipped with a minute disk tip and lengths of 1.5 mm and 2 mm. Currently, there are 3 types of Dual knife: KD-650Q, KD-650U, and KD-650L. Of these, the KD-650Q is usually used for ESD of colorectal tumors with a disk tip (the length of 1.5 mm). Then the knife with a length of 1.5 mm is usually used for ESD of colorectal tumors. When small blood vessels are exposed or bleeding occurs during the ESD procedures, colonoscopists can usually prevent bleeding or stop bleeding successfully with the electrocoagulation mode of the Dual knife, instead of changing hemostatic devices. Furthermore, it can keep the endoscopic views clear, avoid complications, and decrease the procedure time. For large vessels or small vessels that cannot achieve hemostasis, hemostatic forceps or endoscopic metal clips should be used.
In our study, we reported our encouraging experience of ESD by using the Dual knife only for colorectal LSTs. Thereby we summarized some technical skills of application of the Dual knife during the ESD procedure on the basis of our experiences with it as follows: 1) Keep an appropriate working distance between the tip of the knife and the tip of the endoscope, then the tip of the knife is pressed softly on the mucosa and the cutting line is under the direct visualization during mucosal cutting, avoiding the blind operation. 2) Control appropriate tension with regard to mucosal thickness, tumor location, and degree of mucosa prone to bleeding during mucosal incision. For lesions located in the rectum with thick mucosa, we can increase properly the incising tension and power setting. On the other hand, for mucosa prone to bleeding or thin mucosa, tension and power setting are decreased to prevent deep incising. 3) During submucosal dissection, the thickness of the submucosal cushion should be maintained continuously. As the incision proceeds, dissection is carried out. Dissecting submucosa of a lesion is better performed in the longitudinal direction with the aid of gravity or the transparent cap, avoiding damage to the muscular layer. 4) Mucosal incision and submucosal dissection are sometimes very difficult during the final stage of ESD procedures. Therefore, we ought to adjust the position and direction of the tip of the Dual knife and focus on the target tissue, avoiding damage to the surrounding mucosa.
For these recent cases, all the colorectal ESD was completed with the Dual knife, which reduces the impact of exchanging attachments on the operation time. Thus, utilization of the Dual knife for colorectal ESD is worthy of clinical widespread application. Furthermore, expert endoscopists are essential for carrying out high-quality ESD with the Dual knife. As with all the other new technologies, a learning curve exists in the process of ESD [20–22]. Systematic teaching within gastroenterology training procedures, intensive short courses, advanced endoscopy equipment, and computer-based training patterns may assist inexperienced endoscopists to learn endoscopic techniques.
Our experience was limited by the fact that it was a retrospective study that was carried out at a single hospital with a finite number of patients. Apart from that, diverse therapeutic instruments and procedures (injection solutions, coagulation models, etc.) would contribute to increased variability in the observed outcomes.
Conclusions
With regard to the colorectal LSTs, ESD using the Dual knife is safe, feasible, and minimally invasive. However, to confirm our results, larger trials with a longer follow-up are required together with improvement of the technique and of the technical devices. A prospective multicenter trial is needed to confirm the usefulness of the Dual knife in comparison to other ESD knives.









