Introduction
Priority pathogens are biological infectious agents characterized by particular virulence and drug resistance. One of the most important families of priority pathogens is Enterobacteriaceae spp. such as Klebsiella pneumoniae and Escherichia coli producing extended-spectrum β-lactamases (ESBL (+)) [1, 2]. The incidence of infections caused by ESBL (+) pathogens is increasing every year, mainly because of inadequate antimicrobial therapy [3–6]. ESBL (+) pathogens are becoming a major challenge in clinical practice, causing hospital-acquired infections in patients who have undergone multiple hospitalizations as well as community-acquired infections in outpatients [3, 4, 7–13].
Extended-spectrum β-lactamases is a rapidly evolving group of enzymes capable of decomposing the majority of β-lactam antibiotics such as third-generation cephalosporins and aztreonam, but not active towards cephamycins, carbapenems and β-lactamase inhibitors [5, 6, 11, 12]. Penicillins with β-lactamase inhibitors reach high concentrations in urine and can be used in treatment of urinary tract infections (UTIs) caused by ESBL (+) pathogens [8]. Carbapenems still remain the gold standard of treatment for serious pediatric ESBL (+) infections [1, 12, 14]. Ertapenem and imipenem are considered to be the most effective antibiotics against ESBL (+) E. coli strains [15], but in some cases after antibiogram results the therapy can be changed to non-carbapenem alternatives such as fluoroquinolones, aminoglycosides, nitrofurans, co-trimoxazole [1, 4, 8, 14].
Due to plasmid location of ESBL genes there is a risk of gene transfer to another resistant strain and development of plasmid-mediated multidrug resistance, which limits the therapeutic possibilities [1, 12, 14].
Extended-spectrum β-lactamases (+) strain infections are becoming a growing problem in empirical treatment for doctors around the world. In our study we want to show the demographics, risk factors and treatment outcomes of pediatric patients infected by ESBL (+) pathogens from 5-year experience in our clinic.
Material and methods
The medical documentation data of patients hospitalized in the Department of Pediatrics, Pediatric Nephrology and Allergology, Military Institute of Medicine, Warsaw, Poland between January 2011 and December 2015 were included in the retrospective analysis.
Records of ESBL (+) bacterial infections were separated among documentation of all hospitalized children and subjected to a detailed analysis. Age, gender, severity of infection, urinalysis, antibiotic selection according to the antibiogram and outcome were considered. A correlation between the presence of ESBL (+) bacteria in the analyzed sample and clinical status and indicators of inflammation was also included. The children fulfilling the criteria for symptomatic infections and asymptomatic carriers were separated.
Identification of isolates was performed by an automated system for identification, VITEK2. All the strains were classified as ESBL (+) by the method of double discs.
Statistical analysis
The results are presented as the mean value and percentages. The obtained data were analyzed by means of Microsoft Excel and analytics software Statistica. The resemblance of a particular variable distribution to the normal distribution was tested by Shapiro-Wilk and Wilkinson tests.
Results
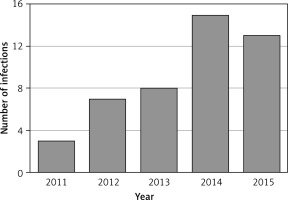
Among 8015 hospitalizations and 537 positive cultures of biological material performed in the 5-year-period, 46 ESBL (+) infections were recorded, which constituted 0.57% of all hospitalizations and 8.5% of all positive microbiological tests. The analysis revealed an increasing trend in the number of ESBL (+) infections between January 2011 and December 2015 (Figure 1).
The mean age of analyzed children was 5.35 years and 65.2% of patients were female. 43.5% of patients were asymptomatic carriers. 76.1% of patients had a history of previous hospitalization and in 17 (37%) cases there was more than one hospitalization. The mean time from the last to current hospitalization was 2.3 months. 71.7% of patients had urinary tract structural or functional abnormalities such as urolithiasis, horseshoe kidney, dilation of the renal pelvis and calyces, recurrent urinary tract infections, vesicoureteral reflux, hypercalciuria, nephrotic syndrome, urosepsis in anamnesis and neurogenic bladder. 60.9% of the patients had a history of urinary tract related procedures such as URSL, ESWL, urography, cystoscopy, cystography, laparotomy, retrograde intrarenal surgery and urodynamic testing. The mean time between the last procedure and ESBL (+) urine culture was 7 months; however, if we rule out one diacritical case (8 years from the last procedure) the mean time changes to 3.3 months. 28.26% suffered from chronic illness other than urinary system diseases and anomalies, especially neurological pathologies such as myelomeningocele, epilepsy, cerebral palsy, paresis, mitochondrial encephalopathy, as well as asthma and allergy. Death has not been reported as a result of ESBL (+) infections. All patients were treated with good results and discharged from the clinic in good clinical condition. Results are summarized in Table I.
Table I
Characteristics of the study group (n = 46)
Due to the nephrological character of the department ESBL (+) pathogens were mostly identified from urine cultures and only in 1 case from wound swab culture. The two most common pathogens were E. coli ESBL (+) and K. pneumoniae ESBL (+). Only in one case did the results of microbiological tests reveal the presence of two causative priority pathogens (Table II).
Table II
Etiology of ESBL (+) infections
Depending on laboratory results and clinical state the empirical antibiotic monotherapy was cefuroxime in 19.6%, amikacin in 8.7% and amoxicillin with clavulanic acid in 6.5% out of 46 cases. In 9 (19.6%) cases more than one drug was used in the therapy (Table III). After obtaining the results from bacterial culture the therapy was modified according to antibiogram results. In all cases ESBL (+) bacteria were susceptible to carbapenems. The resistance to fluoroquinolones was about 25%, to aminoglycosides 10%. The resistance to cephalosporins was approximately 55%, to the combination of β-lactam and β-lactamase inhibitors e.g. piperacillin-tazobactam was approximately 40% and to co-trimoxazole 35%. In 13 cases of asymptomatic carrier state antibiotic treatment was not implemented.
Discussion
Infections caused by ESBL (+) pathogens continue to be associated with significant morbidity and mortality worldwide [16]. The rapid development of resistance mechanisms of ESBL (+) bacteria limits the therapeutic possibilities and increases the cost of treatment. The spread of ESBL (+) strains is an epidemiological risk for patients by prolonging the hospital stay and increasing mortality, as well as for hospitals by causing the necessity of expensive antibiotics usage [1, 4].
Urinary tract infections (UTI) are after respiratory tract infections the second most common type of infection affecting children [6]. By the age of 7 up to 8.4% of girls and 1.7% of boys have been diagnosed with UTI at least once [5, 6]. Numerous factors which predispose children to UTI have already been defined. The most important of them are structural and functional abnormalities of the urinary tract such as vesicoureteral reflux, short urethra in girls and narrow foreskin of male infants [5, 6]. Furthermore, UTI is the most common infection caused by ESBL (+) bacteria, but we have limited knowledge regarding the clinical epidemiology of these infections. Although well characterized in adults, risk factors, outcomes, therapies, and control measures for ESBL (+) bacteria are less appreciated in children [16].
Reports regarding risk factors for infection or colonization by ESBL (+) bacteria in children were mostly obtained from pediatric intensive care units and neonatal intensive care units. The reports described younger gestational age, low birth weight, prolonged mechanical ventilation, length of hospital stay, invasive devices, and antibiotic use, and maternal-child transmission as the most common risk factors of the ESBL (+) infections. Most studies emphasized that beyond the neonatal period ESBL (+) pathogen infection risk factors for children and adults are similar (e.g. foreign travel, recent UTI, recent antibiotic treatment, numerous hospitalizations, intrafamilial transmission) [17–24]. However, a two-center case-control study of ESBL (+) infection risk factors in children from Chicago identified underlying neurological conditions as a potential risk factor specific to children [25–27]. The same correlation was observed in our study. Our study revealed urinary tract instrumentation as a potential risk factor of ESBL (+) infection or colonization. To our knowledge, this correlation has not been reported in English literature yet.
The analysis of our results revealed a fivefold increase in ESBL (+) pathogens isolated in microbiological tests during the observed period (2011–2015). Other studies analyzing the results of microbiological tests in terms of isolation of priority pathogen frequency also showed a significant increase in the percentage of isolation of ESBL (+) strains over the last 20 years [3, 8, 12, 28]. A multicenter study carried out in Spain in 2000 (GEIH-BLEE Project 2000) and 2006 (GEIH-project BLEE, 2006) showed an eightfold increase in ESBL (+) infections with E. coli and a twofold increase in ESBL (+) infections with K. pneumoniae [29]. An analysis of microbiological tests performed by Samet et al. in patients of all departments of a hospital in Gdansk (Poland) conducted in 2001–2003 showed that the frequency of isolation of ESBL (+) did not change significantly during the period, and ranged from 3% to 4.5%. However, in 1999 this incidence was 1.6%, while in 2001 it was 3.2%. The authors of the study suspect that these results may be caused by hospitalizations of asymptomatic carriers [9]. The problem of bacterial ESBL (+) infection in the Pediatrics, Nephrology and Allergology Department of the Military Medical Institute in Warsaw in 2011–2015 concerned from 2.8% to 8.8% of all patients who underwent microbiological tests. The incidence was comparable to the data available in the literature [4, 8, 9].
Nearly half of the children with isolated priority uropathogens were asymptomatic carriers. The results of urine cultures of these patients were obtained after patients’ discharge. It was recommended to perform control urine culture. In the case of re-cultured priority pathogens, treatment should be considered. Carrier state of ESBL (+) strains was mainly associated with previous long-term stays in hospitals, which correlates with our findings [16, 30–33]. Treatment should be considered individually, because antibiotic therapy may promote antibiotic resistance and predispose to symptomatic infection [34, 35].
The source of ESBL (+) bacteria presence in the human organism has not been definitively identified yet. The antibiotics used for humans are also used in veterinary medicine, agriculture and in the livestock breeding [36]. Therefore, food-producing animals and foods of animal origin are under suspicion for being transmission vectors for colonization and infection of humans with ESBL (+) Enterobacteriaceae [37, 38].
ESBL (+) bacteria have become a leading antibiotic-resistant threat in children. Options for treatment of ESBL (+) bacterial infections are limited, given that fewer antibiotics are approved for use in children [1]. ESBL (+) strains frequently possess co-resistance genes conferring resistance to aminoglycosides, fluoroquinolones, and trimethoprim/sulfamethoxazole, among others, limiting therapeutic options, especially with oral agents [12, 16]. In our study the susceptibility to fluoroquinolones was about 75%, and to aminoglycosides 90%, which was good news in the light of decreasing sensitivity noted worldwide. Therefore, these two groups can be considering as possible therapeutic options in the future.
In conclusion, the results confirm the rising trend of the ESBL (+) infections among hospital-acquired infections during the observed period. ESBL (+) bacteria were isolated primarily in previously hospitalized children with particular reference to urinary tract instrumentation during hospitalizations. The study showed that patients with urogenital disorders and other chronic diseases are more susceptible to these priority pathogen infections. Each bacterial ESBL (+) infection requires cooperation between clinicians, microbiologists and hospital epidemiologists. This allows for early detection of epidemics and the implementation of appropriate action. The epidemiological surveillance of colonized patients is crucial for the prevention of local epidemics.
The study provides an analysis of multiple cases of ESBL (+) bacteria infections and reveals the most important infection risk factors and treatment options. However, the study was performed in a nephrological department that determines the type of ESBL (+) pathogen infections involved in the analysis. More studies are needed to assess the incidence and virulence of ESBL (+) bacteria that might be accomplished by analyzing data from different departments.