Introduction
Exposed endoscopic full-thickness resection (EFTR), previously reported as pure free-hands or standard EFTR, without laparoscopic assistance is a scarless natural orifice transluminal endoscopic surgery (NOTES) technique that is emerging as a promising approach, with good efficacy and safety in the resection of both muscularis propria (MP)-originating submucosal tumors (SMTs) and select epithelial tumors (ETs) unsuitable for conventional endoscopic resection [1–6]. In exposed EFTR, the full-thickness resection is followed by subsequent GI defect closure. Thus, the term “exposed” is derived from the temporary exposure of the peritoneal cavity to the GI lumen [7]. The mainstay of exposed EFTR is a safe wall defect closure after the excision in order to prevent peritonitis and further surgical interventions. Currently, this is achieved principally through the use of standard through-the-scope (TTS) clips [8] or clips combined with endoloops [9, 10]. Endoscopic suturing devices, such as the OverStitch Endoscopic Suturing System (ESS) (Apollo Endosurgery, Austin, Texas, USA), that allow placement of durable full-thickness suturing that incorporates the muscle layer and results in a more stable construct, have been successfully used to suture gastrointestinal (GI) wall defects [11, 12]. However, data regarding their use in the exposed EFTR procedure are limited.
Aim
This study aimed to investigate the efficacy, safety, and feasibility of exposed EFTR followed by defect closure using the OverStitch ESS for select GI lesions.
Material and methods
Study design and patients
This study was a retrospective, observational, open-label, single-arm, consecutive case series conducted at a single tertiary-referral center from September 2018 to July 2019. A consecutive cohort of patients who underwent exposed EFTR without laparoscopic assistance with subsequent and same-time defect closure using the OverStitch for GI SMTs or ETs unsuitable for conventional endoscopic resection was included in our study. The inclusion criteria for patient selection were as follows: (1) gastric or duodenal SMTs arising from the MP, as confirmed by endoscopic ultrasound (EUS), with tumor diameter larger than 20 mm and/or presence of symptoms, gastrointestinal stromal tumor proven by preoperative histopathological examination, evidence of tumor growth during follow-up or patients’ preference; (2) rectal ETs unsuitable for conventional endoscopic resection (e.g., recurrent adenomas with negative lifting sign, incomplete resected non-lifting adenomas and T1-carcinomas incidentally resected using standard endoscopic techniques and with histological submucosal infiltration depth that was not assessable); (3) rectal SMTs with preoperative histological diagnosis of neuroendocrine tumor.
Exclusion criteria were as follows: pregnancy, patients < 18 years of age, failure to provide informed consent, coagulopathy (international normalized ratio > 1.5 and/or platelets < 40000/μl), metastatic disease on computed tomography (CT) and/or EUS, lesion size > 50 mm, SMTs with high-risk EUS features (i.e., irregular borders, cystic spaces, ulceration, echogenic foci, and heterogeneity).
Data were extracted for demographics, lesion features, R0 resection (i.e., negative vertical and lateral resection margins at histology) status, operation time (i.e., from submucosal injection to endoscopic suturing closure completion), surgical conversions (due to technical unfeasibility of the procedure and/or occurrence of adverse events (AEs) not amenable to endoscopic treatment), major AEs (i.e., delayed bleeding, delayed perforation, peritonitis, abdominal abscess, and abdominal infection), and length of hospital stay. Descriptive statistical analysis of the data, including percentage and mean, are reported.
IRRB approval was not needed for the study, and written informed consent was obtained from all patients.
Endoscopic equipment and accessories
Full-thickness resection was performed using a standard single-channel endoscope with a transparent cap (GIF-1TH190 plus D-201-11804, Olympus Europe, Hamburg, Germany Olympus) attached to its front, and an O-type HybridKnife (Erbe Elektromedizin, Tübingen, Germany). A double-channel endoscope (GIF2T160; Olympus Europe, Hamburg, Germany) preloaded with an OverStitch ESS (Apollo Endosurgery, Austin, Texas, USA) was used for wall defect suturing closure.
Pre-operative evaluation and procedures
All patients underwent pre-operative EUS and CT in order to characterize the lesion and exclude malignancy. All the procedures were performed with the patients under general anesthesia by a single, experienced therapeutic endoscopist (A.G.), whose experiences included more than 500 upper and lower GI endoscopic submucosal dissections (ESDs), and more than 100 procedures involving the use of the ESS. In all the cases, an operating room with an on-call abdominal surgeon was preventively activated in case of exposed EFTR failure or intra-procedural AEs not amenable endoscopically. Informed consent for both endoscopic and surgical operations was obtained from the patients.
Only carbon dioxide was used for insufflation. All patients received a single dose of prophylactic broad spectrum antibiotic administered intravenously immediately before the procedure.
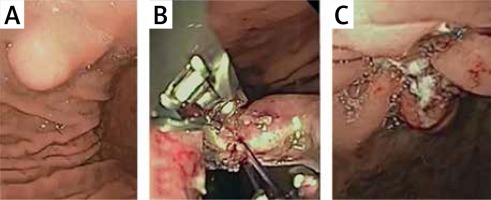
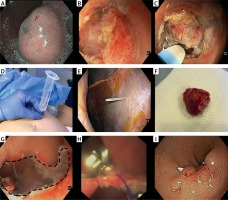
Exposed EFTR was performed as previously described [8]. Wall defect closure was achieved using the ESS in all cases. The procedure systematically involved (A) submucosal injection followed by precutting the mucosal and submucosal layer around the lesion; (B) exposed EFTR of the lesion and creation of an “active perforation” after draining intraluminal fluid; (C) capnoperitoneum management through a percutaneously inserted 20-gauge angiocatheter in the case of upper GI procedures; (D) single-channel endoscope removal and insertion of a double-channel endoscope preloaded with an ESS in order to perform full-thickness suturing closure by creation of either a continuous suture line or separated stitches (Photo 1).
Photo 1
Technical illustration of exposed endoscopic full-thickness resection (EFTR) followed by defect closure using endoscopic suturing system (ESS). A – Magnifying endoscopy with narrow-band imaging showing a submucosal lesion of the duodenal bulb with an erosion on the top. B – Precutting and removal of the mucosal and submucosal layer after submucosal injection, in order to expose the tumor. C – Exposed EFTR of the tumor and creation of “active perforation.” D, E – Capnoperitoneum management using percutaneously inserted angiocatheter. F – Macroscopic histological specimen. G – Duodenal wall defect after the full-thickness resection. H – Defect closure with the OverStitch ESS. I – final apposition of the tissue margins
Post-operative management and follow-up
All patients were admitted to the institute for post-procedure monitoring of AEs and kept nil per os the day of the operation. In cases of upper GI procedures, patients were allowed liquid diet on day 1, soft diet on day 3, and advancement of diet as tolerated thereafter. Conversely, soft diet was begun on day 1 following lower GI procedures. Post-EFTR medication included intravenous infusions of proton pump inhibitors (after upper GI procedures only) and antibiotics. Post-operative investigations to check for the adequacy of wall defect closure were not routinely performed. Patients were closely observed with clinical and laboratory examinations for any signs of peritonitis and/or GI bleeding. Patients were discharged home in the absence of specific complaints.
Follow-up endoscopy was performed 1 and 6 months after the procedure to observe the wound healing and exclude macroscopic recurrence. Subsequently, follow-up strategies were based on the results of histopathological examination.
Results
Seven patients (M : F 6 : 1) with a mean age of 56 ±14.5 years were identified. The indications were as follows: SMTs arising from the MP of the stomach (n = 2) and duodenum (n = 2), and from submucosa of the rectum (n = 1); ETs (n = 2) of the rectum, including one recurrent non-lifting adenoma in a post-endoscopic mucosal resection (EMR) scar, and one T1 carcinoma incidentally resected by EMR, without clear histopathological depth of submucosal invasion (Table I). The mean lesion size assessed by EUS was 25 ±9.9 mm (range: 15–40), and the mean procedure time 164 ±41 min (range: 110–254). Exposed EFTR was successfully performed in 6 of the 7 patients. Defect closure with the ESS was effective in all cases (6/6 patients). One case was converted into laparoscopic gastric wedge resection during the same anesthesia due to technical unfeasibility for completion of the resection. Histopathological examination showed grade 1 neuroendocrine tumors (n = 2) [13], low risk gastrointestinal stromal tumor (n = 1) [14], pancreatic heterotopia (n = 2), low-grade dysplasia adenoma (n = 1), and T2 adenocarcinoma (n = 1). We performed an R0 resection in all cases except for 1 case of rectal EFTR, previously treated by standard endoscopic mucosal resection and with evidence of invasive carcinoma infiltrating the MP. No major AEs were observed. Mean post-procedure hospitalization was 4 ±1.31 days (range: 3–7).
Table I
Clinicopathological characteristics and outcomes of exposed endoscopic full-thickness resection followed by defect closure with the endoscopic suturing system
At 1- and 6-month surveillance endoscopy, wound healing was detected in 6 out of 7 cases, including the gastric case converted into laparoscopic surgery. Neither macroscopic residual tumor nor tumor recurrence was observed (Photo 2). One-month endoscopic follow-up was not performed in the rectal case, in which subsequent surgery was done due to invasive adenocarcinoma. For the 2 cases of pancreatic heterotopia no further follow-up was done. For all the remaining cases, no local or systemic recurrence was observed during a mean follow-up period of 13.2 months (range: 9–18 months).
Discussion
EFTR was first described in 1998 by Suzuki et al. [15]. In 2001, the same group reported the effectiveness of EFTR using a ligation device for the treatment of 2 rectal tumors and 1 duodenal neuroendocrine tumor in humans [16]. A few years later, Ikeda et al. reported EFTR using the ESD technique on a porcine stomach [17], and Zhou et al. translated this technique into clinical practice (26 cases of gastric SMTs) [8].
Thereafter, exposed EFTR without laparoscopic control emerged as a promising, safe, and effective evolution technique for the resection of challenging ETs and deep SMTs through the GI tract, especially gastric MP-originating SMTs [1–6]. Exposed EFTR is an evolving “cut then close” NOTES technique providing the intentional creation of an active perforation for a complete endoscopic resection followed by wall patency restoration. Thus, effective defect closure is a crucial step of the procedure, with a key role in the final outcome. In this setting, different closure methods have been successfully reported in large series, mainly TTS clips, clips combined with endoloops, and over-the-scope (OTS) clips [8–10, 18]. The main limitation of TTS and OTS clip approaches is their inability to close wall defects larger than 10 and 25 mm, respectively [19].
Though technically demanding, effective larger defect closure with either different clips combined with endoloop techniques [9, 10] or the omental-patch method has been reported [20–22].
Following the advent of exposed EFTR, the non-exposed EFTR technique was introduced. Technically, the resection of the lesion is performed after the plication of the GI tract wall with dedicated full-thickness suturing devices, mainly represented by the OTSC system (Ovesco Endoscopy GmbH, Tübingen, Germany). The advantages of this “close then cut” technique consist principally in the potential avoidance of intra-peritoneal dissemination of tumor cells, and potential risk of infection. In addition, this approach has greater technical simplicity, with faster operating time. However, compared with exposed EFTR, the OTSC system is limited by a lower resection R0 rate, likely due to the impossibility of a “real-time” and direct visualization of the perimetral cutting margins. Also, the clip cannot be reverted once released, and is limited to small lesions (< 25 mm) [5].
In exposed EFTR, a full-thickness defect closure is strongly advised by the American Society for Gastrointestinal Endoscopy [7]. This is not achievable by standard clip closure methods, which allow mucosal and submucosal approximation only, due to the superficial bite of the clips [23]. Conversely, suturing closure by the use of ESS creates a full-thickness “surgical” suture through all layers of the GI wall, allowing secure closure of even large defects [24–26]. However, the efficacy and safety of OverStitch ESS for closure of post-EFTR wall defects have been reported only in a handful of case reports and 1 case series [27–32].
The concept of full-thickness suturing closure is of particular interest when performing exposed EFTR in ill-advised locations, such as the duodenum and colon-rectum. In fact, in these sites, ineffective defect closure is associated with higher morbidity and mortality than elsewhere in the GI tract. Indeed, evidence concerning exposed EFTR for duodenal or colorectal lesions is lacking due to concerns about safe and reliable defect closure achievement [25, 28, 33, 34].
In our case series wall defect closure using the Overstitch ESS was feasible and effective in all patients (6/6), including two duodenal defects (18 and 30 mm, respectively) and three rectal defects (40, 40, and 20 mm, respectively). No major AEs occurred, including delayed perforation and peritonitis. Only one gastric case was converted into laparoscopic wedge resection. After 75% of the resection, completion of the EFTR appeared technically unfeasible. Attempts at thread-with-clip countertraction, as previously reported [35, 36], were not effective in obtaining successful triangulation. Endoscopic peritoneal exploration showed the extraluminal growth pattern of the gastric SMT, and its close contact with the left liver lobe. The patient was previously informed that in case of EFTR failure, the standard laparoscopic approach would be performed during the same general anesthesia as a step-up approach. No major AEs occurred after the laparoscopy, R0 resection was achieved, and the patient was discharged on day 7.
In 2 cases, final histological diagnosis was heterotopic pancreas. In both cases pre-operative EUS-guided fine needle biopsy was performed. However, cytohistological examination was inconclusive, and both patients refused long-term endoscopic surveillance. Thus, exposed EFTR was proposed after multidisciplinary evaluation. As previously reported, indications for EFTR can include undiagnosed lesions in younger patients for whom the risk of resection might be outweighed by the benefit of avoiding long-term surveillance [37].
R1 resection was observed in only one rectal EFTR, which was performed for re-resection of a T1 carcinoma incidentally diagnosed in an EMR-treated lesion. Submucosal infiltration depth could not be determined accurately by the pathologist, and subsequent pre-operative EUS did not show clear MP infiltration. In this clinical setting, EFTR may represent an excellent diagnostic technique and, in the case of low-risk tumors (submucosal infiltration depth < 1000 µm, G1 or G2, absence of lymphatic vessel invasion, R0 resection), it is even therapeutic [38]. Thus, EFTR was performed. Unfortunately, the final histopathological examination was consistent with invasive adenocarcinoma infiltrating the MP and the patient underwent subsequent surgery.
Post-EFTR wall defect closure using the OverStitch was performed with either interrupted or continuous sutures according to the size, shape, and location of the defect [39].
The use of the OverStitch ESS for duodenal post-EFTR defect closure appeared feasible in experienced hands. However, compared with its use within the gastric or rectal cavity, endoscopic suturing within the duodenum seemed technically more demanding, mainly due to the limited working space.
In our opinion, the worldwide diffusion in clinical practice of exposed EFTR and other NOTES techniques during the last 15 years may be partly due to the unavailability of endoscopic suturing devices. The recent advent of such devices might represent a new start for NOTES procedures. ESS could be considered the natural evolution of the endosurgical technique of exposed EFTR, providing safe and effective full-thickness defect closure, and thus expanding its potential usefulness. Its role as a first-line endosurgical procedure, including in an eventual step-up approach for the management of select GI tumors in referral centers, appears particularly intriguing [40]. High morbidity and mortality associated with major surgery, especially of the duodenum, justify active research in this field.
Conclusions
Exposed EFTR without laparoscopic assistance, followed by suturing closure using the OverStitch, appears to be feasible, effective, and safe for the management of select epithelial and subepithelial tumors of the GI tract. Further prospective and randomized studies are needed to clarify the role of Overstitch ESS for post-EFTR wall defect closure.