Introduction
Metabolic/bariatric surgery has been developing intensely as a method of treating obesity and its systemic consequences. Among available surgical approaches, laparoscopic sleeve gastrectomy (LSG) is worth attention due to satisfactory body mass reduction and amelioration of co-morbidities. It is the most common bariatric procedure performed in Poland [1]. The final effect of treatment is a result of stomach volume reduction and, in consequence, restriction of food intake. Furthermore, recent research suggests that resection of the major part of a stomach (the fundus and body) leads to significant changes in gastrointestinal tract peristalsis, as well as neurohormonal and carbohydrate-fat balance [2–4]. Long-term metabolic effects depend on many factors and most of them are still unknown. The differences in outcomes of bariatric surgery may be the results of the patients’ gender, age, physical activity, preoperative weight loss and compliance with dietary recommendations. However, no clear conclusions regarding factors influencing the effect of LSG have been established.
Aim
The research has been conducted to evaluate gender-related differences in the results of laparoscopic sleeve gastrectomy in 6-month follow-up.
Material and methods
The study group included 101 patients who underwent surgery between January 2012 and December 2014. All patients provided written informed consent prior to the study and additional written informed consent was obtained before the surgical procedure. The study was approved by the Ethics Committee of the Medical University of Bialystok, Poland (No. R-I-002/438/2014) in accordance with the guidelines of the Helsinki Declaration and its later amendments.
Inclusion criteria for the surgical procedure were failure of weight loss after conservative treatment, body mass index (BMI) ≥ 40.0 kg/m2 or ≥ 35.0 kg/m2 with the presence of obesity-related co-morbidities, no alcohol or drug abuse as well as no active psychosis. All qualified patients underwent laparoscopic sleeve gastrectomy performed by the same operating team – the operator and 2 assistants. The procedure included dissection of the major curvature that started 2 or 6 cm from the pylorus and continued toward the left crus of diaphragm. 32 Fr or 40 Fr calibrating tubes were used to control the diameter of the remaining stomach. As the final step the leak test was performed using a 5% glucose solution and air.
The primary endpoint of the study was the influence of patient’s gender on postoperative weight loss. Secondary outcomes were differences in laboratory test results observed in the postoperative period with regard to patient’s gender. Patients were divided and analyzed in two groups: males vs. females.
Data were collected before the surgery, as well as 1, 3 and 6 months postoperatively. The measurements included body mass, BMI, fasting glucose and insulin concentrations, glycated hemoglobin level (HbA1c), alanine aminotransferase (ALT), aspartate aminotransferase (AST), total cholesterol and its fractions, triglycerides, and C-reactive protein (CRP).
The calculation of the percentage of excess weight loss (%EWL), the percentage of excess BMI loss (%EBMIL) and the homeostatic model assessment of insulin resistance index (HOMA-IR) was performed using the following formulas:
%EWL = (body mass before the surgery – body mass during follow-up)/(body mass before the surgery – ideal body mass) × 100.
In order to calculate ideal body mass, the following formulas were applied:
%EBMIL = (BMI before the surgery – BMI during follow-up)/(BMI before the surgery – 25) × 100.
HOMA-IR = glucose level (mg/dl)* insulin concentration (mU/l)/405; result > 2.6 confirmed insulin resistance.
Statistical analysis
Data analysis was conducted using Statistica v13.5 (StatSoft Inc., Tulsa, Oklahoma, USA). Continuous variables with normal distribution are presented as mean, standard deviation (SD) and 95% confidence interval (95% CI). Skewed variables are presented as median with inter-quartile range (IQR). Student’s t test was used to compare continuous variables between groups or the Mann-Whitney test for skewed ones. Dichotomous variables were analyzed with Pearson’s χ2 test. For repetitive observations, the repetitive measurements ANOVA with post-hoc Tukey’s test was used. In the case of skewed variables, Friedman’s ANOVA with its post-hoc test was used. A p-value of < 0.05 was considered to be statistically significant.
Results
Material
The cohort included 101 patients with the average age of 43 years. Women accounted for 52% of the cohort (n = 53), men for 48% (n = 48). The characteristics of the groups are presented in Table I. Patients reported a number of co-morbidities preoperatively, of which the most common were: type 2 diabetes (6 women – 11%, 11 men – 23%), depression (14 women – 26%, 7 men – 15%), hypertension (13 women – 25%, 12 men – 25%) and dyslipidemia (5 women – 9%, 7 men – 15%).
Table I
Characteristics of group differences at the time of LSG
Preoperative differences
As presented in Table I, age did not differ between males and females. Males had significantly higher body mass and BMI. Operative technique distribution was comparable between groups in Pearson’s χ2 test. Males presented with significantly higher glucose and insulin level; therefore also HOMA-IR was higher in male patients. Median triglycerides, ALT and AST were also significantly higher in male patients (Table I).
Primary outcome
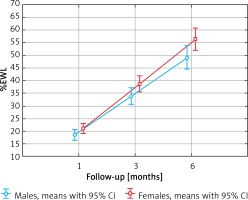
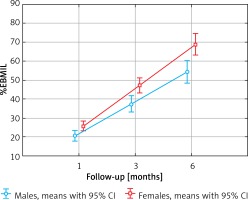
The influence of patient’s gender and operative technique was examined in repetitive measurements ANOVA of %EWL and %EBMIL, as presented in Table II. Main effects of repetitive measures ANOVA showed significant changes between measures both for %EWL (p < 0.001) and %EBMIL (p < 0.001), as well as a significant influence of patient’s gender on it (for %EWL p = 0.026, and for %EBMIL p = 0.001). Furthermore, post-hoc Tukey’s test was conducted. %EWL in 1 month and 3 months did not differ significantly between males and females (p = 0.904 and p = 0.335 respectively). Females had significantly higher %EWL after 6 months (p = 0.034). Means of %EWL with 95%CI are presented in Figure 1. %EBMIL did not differ between males and females at 1 month (p = 0.572). Females had significantly higher %EBMIL at 3 months and 6 months than males (p = 0.022 and p < 0.001 respectively). Operative technique did not change %EWL with significantly regard to patients’ gender as a predicting factor (p = 0.678). The same was for %EBMIL (p = 0.728) (Figure 2).
Table II
Repetitive measurements of %EWL and %EBMIL
Secondary outcomes
The analysis of carbohydrate profile showed a statistically significant decrease in glucose level in both men and women (median value after 6 months: female – 90 mg/dl, male – 93.00 mg/dl; p = 0.001). Among all analyzed additional parameters, the insulin and glucose levels showed statistically significant changes between men and women (p = 0.031 and p = 0.012 respectively). The assessment of lipid profile indicated statistically significant decreases in total cholesterol (p < 0.001), triglycerides (p < 0.001), and LDL (p = 0.05) and an increase in HDL level (p < 0.01). The total results of repetitive measurements of selected laboratory tests are presented in Table III.
Table III
Repetitive measurements of selected laboratory test results
Discussion
Due to the fact that obesity is not only a metabolic but also a social and economic issue, the world of medicine has been intensively looking for the best methods to cope with this problem [5]. Worldwide studies have repeatedly proven that bariatric surgery is the most effective treatment of morbid obesity and its co-morbidities in both short- and long-term observations. Implementation of bariatric procedures allows surgeons to achieve satisfactory weight loss and improvement in insulin, glucose and lipid metabolism [6].
The research examined metabolic differences after LSG according to the patients’ gender.
Postoperative dynamics of body mass reduction and BMI changes over the examined period were calculated using the %EWL and %EBMIL indicators. Significant weight loss calculated by %EWL and decrease in BMI estimated by %EBMIL were recorded in both groups during the observation period. Females had significantly higher %EWL and %EBMIL at 6 months postoperatively than males. Perrone et al. described better outcomes in terms of %EBMIL in males than in females after LSG in 5-year follow-up, which is opposite to our results. However, we observed our patients for a shorter time and the male group had a higher BMI preoperatively [7]. Study conducted by Binda et al. showed that lower age and preoperative weight loss are conducive to achieving higher %EWL, but no differences between genders have been found to be essential for the results [8].
A study published by Yuval et al. compared the dynamics of changes in body mass of patients after sleeve gastrectomy with the division into two groups depending on the size of the calibrating tube (< 40 Fr vs. ≥ 40 Fr) and found no statistically significant difference in %EWL [9]. Unsatisfactory body mass loss frequently forces surgeons to convert the surgical technique to more restrictive one [10]. However, there is a lack of clear proof in the worldwide literature for the statement that application of a smaller calibrating tube is associated with higher body mass. Regardless of the technique of LSG, lifestyle changes and following doctor’s recommendations result in satisfactory weight loss. Lombardo et al. proved that the weight regain rate was lower in patients who more frequently participated in follow-up visits [11]. According to the study conducted by Stroh et al., men present with higher incidence of co-morbidities such as type 2 diabetes, hypertension and dyslipidemia, which subsequently reduces the rate of postoperative amelioration [12]. In our study, we observed higher incidence of type 2 diabetes and dyslipidemia in males.
Alignment of disorders of carbohydrate metabolism coexisting with obesity after sleeve gastrectomy is mainly related to the reduction of body weight [13]. However, it has been proved that the improvement of the glycemic profile, insulin concentration or the level of glycated hemoglobin is observed before significant changes in weight and body composition. In our study, the dynamics of changes in insulin levels and HOMA-IR were examined, useful in the assessment of insulin resistance of peripheral tissues and the function of pancreatic beta cells. In our study, the largest decrease in insulin level was noted after the first month of observation (16.80 μU/dl vs. 9.00 μU/dl in women and 23.10 μU/dl vs. 10.85 μU/dl in men). The assessment of HOMA-IR between males and females showed normalization (values < 2.6) 6 months postoperatively, reaching a median of 1.35 for females and 1.63 for males. Rizello et al. observed a significant decrease of insulin resistance in some patients after sleeve gastrectomy 3 days after the surgery [14]. Thus, 15 days after the surgery in all patients glucose and insulin concentrations in serum as well as HOMA-IR significantly decreased before the occurrence of changes in body mass. Similar results were obtained by Catoi et al., who described a similar decreasing tendency in insulin concentration and HOMA-IR 7 days postoperatively. In their study, statistical significance was reached 30 days after the surgery [15]. Sharma et al. described a case of a 49-year-old obese patient (BMI 59 kg/m2) who had shown a fast (14 days after the surgery) decrease of insulin concentration, from the initial value of 49.5 μU/ml to 16.5 μU/ml. What is more, HOMA-IR was 4.6 after 14 days (initial value 8.82) and after 7 months it normalized to 2.4 [16].
Improvement of carbohydrate profile after sleeve gastrectomy is strictly connected with body mass reduction and changes in volume of fat tissue. However, recent studies prove that amelioration occurs in the early postoperative period. The explanation of this mechanism is probably connected with neurohormonal balance of the gastrointestinal tract. Resection of the majority of the stomach is connected with removal of cells producing ghrelin, which occur mostly in the fundus. According to different publications, the concentration of this hormone decreases after the surgery by 40–50% in comparison to the initial value [17–19]. Research conducted by Dardzinska et al., who compared the pre- and post-prandial changes in both isoforms of ghrelin in obese patients showed that sleeve gastrectomy leads to a decrease in des-acyl ghrelin levels [20]. In consequence, it reduces appetite and glucose concentration in serum, increases secretion of insulin and lowers insulin resistance. A further mechanism explaining the process of carbohydrate metabolism improvement is regulation of incretin hormones. The influence of bariatric surgery on normalization of glycemia probably explains the hindgut hypothesis, which is connected with accelerated contact of food with the distal part of the small bowel and as a result increases secretion of glucagon-like peptide-1 (GLP-1) and peptide YY (PYY).
It is believed that sleeve gastrectomy is associated with accelerated stomach emptying from undigested food and its fast passage through the duodenum and initial part of the small intestine [21, 22]. Moreover, after the surgery, decreased secretion of hydrochloric acid is observed, which directly stimulates secretion of PYY as well as the peptide releasing gastrin and as a consequence release of GLP-1 [23]. Karamanakos et al. reported that after sleeve gastrectomy both fasting and postprandial concentration of PYY increases significantly and ghrelin concentration decreases [19]. Basso et al. observed increases of GLP-1 and PYY in the early postoperative period, which is similar to the results of Peterli et al. [24, 25]. Increase of PYY and GLP-1 concentration is responsible for reduction of appetite and, most importantly, decrease of glucose, restoration of insulin sensitivity, glucagon secretion inhibition and as a consequence inhibition of hepatic gluconeogenesis, which beneficially influences parameters of carbohydrate balance until a body mass reduction occurs. Wroblewski et al. observed that weight loss rather than type of procedure is mostly responsible for hormonal variation in obese patients and indicates the leptin level as the best indicator of body mass changes [26].
Research has shown that even in 60% of patients, obesity is connected with steatosis, including 55% of the pediatric population [27–30]. Despite routine abdominal ultrasonography, which according to different authors has low sensitivity and specificity in recognition of liver steatosis, measures of aminotransferases activity have been performed in all patients [31].
In our study group, changes regarding lipid metabolism included increase of HDL and decrease of total cholesterol, LDL and triglycerides 6 months postoperatively. The increase of HDL cholesterol level has also been proved in the studies of Zhang et al. [32] and Wong et al. [33]. However, there is no relationship between cholesterol increase and changes in body weight.
It is now assumed that fat tissue is responsible for homeostasis of the human organism and it is an important metabolic organ. Furthermore, in the occurrence of insulin resistance, fat tissue macrophages play a significant role and are the source of pro- and anti-inflammatory cytokines. A strong correlation was demonstrated in obese patients between C-reactive protein concentration in serum and BMI, and furthermore, body mass loss causes CRP decrease [33–35]. Moreover, a rapid increase of CRP in the early postoperative period is a marker of complications after LSG [36]. In our examined group, the concentration of CRP did not exceed laboratory norms in any observation period.
The limitation of our study was selection bias. Patients were not matched with respect to baseline body mass and BMI, which were significantly higher in males. The distribution of coexisting diseases was also not comparable. Males were diagnosed with type 2 diabetes in 11 (23%) cases, and females in 6 (11%) cases, whereas dyslipidemia occurred in 5 (9%) women and 7 (15%) men. These differences may affect weight loss outcome and changes in metabolic profile parameters after LSG.
Conclusions
Essential changes of %EWL and %EBMIL as well as the influence of patients’ gender on the postoperative weight loss parameters were observed in the study. According to our research, obese females benefit more after LSG than obese males in the terms of postoperative body mass reduction. The study reveals that patients’ gender may be a predictor for LSG outcomes. However, further research with a larger group and better patients’ selection is needed to provide strong evidence of an association between gender and results of laparoscopic sleeve gastrectomy.