Introduction
The formation of DNA adducts with the mutagenic metabolites of benzo[a]pyrene (Bp) and estradiol (Es) is a critical event for breast carcinogenesis [1-4]. These low-weight compounds being conjugated with macromolecular carriers turn into haptens and are able to induce specific immune reactions. It was shown that antibodies (Abs) to Bp, Es and progesterone (Pg) could modulate the levels of these compounds in the blood serum and some tissues after immunization of animals [4-8]. Previously the concept of immunological disbalance between Abs to chemical carcinogens and endogenous steroids stimulating and inhibiting the initiation and promotion events of carcinogenesis was postulated [9]. Associations of Abs to Bp, Es and Pg with breast and lung cancers were revealed [10, 11]. It was supposed that Abs to Es could stimulate but Abs to Pg could inhibit the carcinogenesis by modifying the concentrations of these hormones in the blood serum, and corresponding anti-idiotypic Abs could act the same way by the binding to membrane Es and Pg receptors on the target cells. This assumption was confirmed partially by revealing the Abs specific to Es receptors (likely the anti-idiotypic Abs to Es) in the blood serum of breast cancer patients (BCP), which acted as Es agonists functioning as cancer-promoting factors [12].
However, the influence of specific Abs on the steroid hormones levels in the blood serum of healthy donors and cancer patients was not studied. The participation of Abs to environmental carcinogens in the modulation of hormone levels by specific Abs remained unknown.
The aim of this study was to reveal the expected cooperative actions of Abs to Es and Pg with the corresponding anti-idiotypic Abs on the Es and Pg concentrations in the blood serum of postmenopausal healthy women (HW) and BCP. The concept of immune-hormonal network with participation of Abs to Bp will be proposed.
Material and methods
Patients
The study was performed in 525 non-smoking postmenopausal women, of whom 443 were primarily diagnosed with invasive breast cancer (BC) in the Regional Clinical Oncology Dispensary (Kemerovo, Russian Federation). Each case diagnosis was confirmed morphologically in the oncology dispensary. Data related to tumor size, grade, and stage were collected from surgical pathology reports. 82 HW without breast pathology were included for comparison. The median ages of participants were 63 (ranging from 42 to 85) for BCP and 58 (ranging from 43 to 80) for HW.
The study protocol conforms to the ethical guidelines of the 1975 Declaration of Helsinki and was approved by the ethics committee of the Institute of Human Ecology of the Siberian Branch of the Russian Academy of Sciences (protocol No. 48/1). All women provided informed consent.
Inclusion and exclusion criteria
Inclusion criteria: for BCP group – patients with primarily diagnosed invasive breast cancer, non-smoking postmenopausal women, patients were aged over 40 years old; for control group – healthy postmenopausal women without breast pathology, non-smoking, women were aged above 40 years old.
Exclusion criteria: patients with other types of tumor, smoking women, young women under 40 years old.
Immunoassay of idiotypic Abs1 and anti-idiotypic Abs2 to estradiol and progesterone
Abs1 and Abs2 to Es and Pg were detected by solid phase non-competitive enzyme-linked immunosorbent assays. Microtiter wells were coated with 2 µg/ml Es or Pg conjugated with bovine serum albumin (BSA) (Amresco, USA) in 100 µl of phosphate-buffered saline (PBS), pH 7.4 (Amresco, USA) at room temperature overnight for detection of Abs1. The Es-BSA conjugate was obtained from Sigma-Aldrich (Germany). Pg-BSA conjugate was obtained by conjugation of hemi-glutarate of 21-hydroxyprogesterone (Sigma-Aldrich, Germany) and BSA by the carbodiimide method. Analysis of anti-idiotypic Abs2 to Es and Pg (IgG-Es2, IgG-Pg2) was performed on commercial kits of ImmunoFA-Estradiol and ImmunoFA-PG (Immunotech, Russia) with monoclonal Abs against Es and Pg immobilized on plastic. Coated wells were blocked for 30 min in blocking buffer saline (0.5% BSA in PBS, 0.05% Tween 20). The serum samples were diluted at 1/20 for Abs1 and 1/100 for Abs2 in blocking buffer saline and were incubated with coated antigens (100 µl/well) for 1 h at 37°C. Bound Abs were detected by goat anti-human immunoglobulin (Ig) A or IgG antibody labeled with horseradish peroxidase (1/10000 dilution, Novex, USA). After each assay step wells were washed 4-5 times with 250 µl/well of washing buffer saline (PBS, 0.05% Tween 20). The amounts of bound Abs were determined through enzymatic reaction with the chromogenic substrate 3,3’,5,5’-tetramethylbenzidine (TMB) (Sigma-Aldrich, Germany). The reaction was then terminated by addition of 2 N HCl and absorbance was measured at 450 nm. All measurements were conducted in duplicate.
The levels of Abs1 and Abs2 to Es or Pg were expressed in arbitrary units and calculated based on the following formula:
IgA(G) – X = (OD X-BSA – OD BSA)/OD BSA
where X is Es or Pg, ODX-BSA is the absorbance of the binding to hapten-BSA conjugate, ODBSA is the absorbance of the binding to BSA.
Steroid hormones determination
The concentrations of Es and Pg were determined using the commercial kits ImmunoFA-Estradiol and ImmunoFA-PG (Immunotech, Russia) according to the instructions for use.
Statistics
All statistical analyses were conducted using STATISTICA version 8.0 (StatSoft Inc., USA). Normality was evaluated by Shapiro-Wilk’s W-test. The non-parametric χ2 test with Yates’ correction was used for comparison of data. The relationship between the Abs levels and the steroids hormones was assessed using Spearman’s rank correlation analysis. All statistical analyses were two-sided, and p < 0.05 was considered statistically significant. ROC analysis was performed to determine the threshold values (cut-off) of Abs and steroid hormones [13]. The odds ratio (OR) was determined with the 95% confidence interval (95% CI).
Results
Serum levels of estradiol and progesterone and corresponding specific antibodies in postmenopausal healthy women and breast cancer patients
The blood serum concentrations of Es and Pg and levels of specific IgA (IgA-Es1 and IgA-Pg1) and corresponding anti-idiotypic IgG (IgG-Es2 and IgG-Pg2) were studied in postmenopausal HW and BCP. The individual ratios Pg/Es, IgA-Pg1/IgA-Es1 and IgG-Pg2/IgG-Es2 were calculated. ROC analysis was performed to determine the threshold values of studied Abs and steroid hormones (cut-off) between BCP and HW.
The frequencies of cases with low (≤) and high (>) hormone concentrations and ratios in compared groups are shown in Table 1. High levels of Es (> 200 pmol/l) were revealed in BCP more frequently (68.2%) than in HW (50.0%, p = 0.002, OR = 2.1). High levels of Pg (> 800 pmol/l) in BCP were found more rarely (41.1%) than in HW (67.1%, p < 0.0001, OR = 0.3). High Pg/Es ratios (> 4) were revealed in BCP more rarely (31.6%) than in HW (51.2%, p < 0.0001, OR = 0.4). It means that high Es concentration was associated with high BC risk, but Pg concentration was associated with low BC risk, as well as high personal Pg/Es ratio.
Table 1
Case numbers (n) and frequencies (%) with low (≤) and high (>) estradiol and progesterone (Es and Pg) concentrations (pmol/l) and Pg/Es ratios in the blood serum of postmenopausal breast cancer patients and healthy women
So the disruption of normal balance between Pg and Es levels was a critical event for the breast carcinogenesis.
The frequencies of cases with low (≤) and high (>) studied Abs levels and ratios in compared groups are shown in Table 2. No associations of IgA-Es1 and IgA-Pg1 with BC were found. High individual IgA-Pg1/IgA-Es1 ratios (> 1) were found in BCP more rarely (17.8%) than in HW (31.7%, p = 0.006, OR = 0.5). High IgG-Es2 levels (> 3) were found more frequently in BCP (71.6%) than in HW (59.8%, p = 0.04, OR = 1.7). High personal IgG-Pg2/IgG-Es2 ratios (> 1) were revealed more rarely in BCP (19.6%) than in HW (37.8%, p = 0.0005, OR = 0.4). It means that the predominance of IgA-Pg1 and IgG-Pg2 levels over IgA-Es1 and IgG-Es2 was associated with low BC risk.
Table 2
Case numbers (n) and frequencies (%) with low (≤) and high (>) antibody levels and ratios in postmenopausal breast cancer patients and healthy women
However, it could be true only when high individual IgA-Pg1/IgA-Es1 ratio was combined with high IgG-Pg2/IgG-Es2 ratio. This combination (3.4 in Table 2) was revealed in BCP more rarely (4.1%) than in HW (23.2%, p < 0.0001, OR = 0.1). The BC risk increased (OR = 1.7) when the levels of IgA-Pg1 and IgG2 were lower than IgA-Es1 and IgG-Es2 (combination of ratios 3.1). There were no significant differences between BCP and HW in the other possible combinations (3.1-3.2 in Table 2).
So the disruption of normal balance between IgA-Pg1 and IgA-Es1 as well as between IgG-Pg2 and IgA-Es2 was a critical event for the breast carcinogenesis.
Relationships between hormone concentrations and specific antibody levels in postmenopausal healthy women and breast cancer patients
We studied the frequencies of low and high levels of Es and Pg and Pg/Es ratios depending on the low and high levels of the specific Abs and Abs ratios in postmenopausal HW and BCP.
There was no interconnection between hormone concentrations and ratio on the one hand and IgA-Es1 and IgA-Pg1 levels on the other hand in HW (Table 3). The high individual IgA-Pg1/IgA-Es1(> 1) ratio was associated with a low concentration of Es (< 200 pmol/l, p = 0.008) and with a high concentration of Pg (≥ 800 pmol/l, p = 0.005) and with a high personal Pg/Es ratio (> 4, p = 0.001) in HW.
Table 3
Case numbers (n) and frequencies (%) of low (<) and high (≥) levels of estradiol and progesterone (Es and Pg) (pmol/l) and Pg/Es ratios depending on low (<) and high (≥) levels of the studied antibodies and antibody ratios in postmenopausal healthy women
Hormone concentrations and Pg/Es ratio were not associated with IgG-Es2 levels in HW. High levels of IgG-Pg2 (> 2) were associated with high Pg concentration (p = 0.012) and high Pg/Es ratio (p = 0.001) in HW. High individual IgG-Pg2/IgG-Es2 ratios (> 1) were associated with low Es concentration and with high Pg concentration and high personal Pg/Es ratio in HW.
The corresponding relationships in BCP had some differences (Table 4). There were no associations between IgA-Pg1/IgA-Es1(>1) ratio and hormone concentrations and ratio, unlike in HW. High levels of IgG-Pg2 (> 2) were associated with high Es concentration (p = 0.016) and low Pg concentration (p = 0.001) and low personal Pg/Es ratio (p = 0.012). High individual IgA-Pg2/IgA-Es2 ratio was associated with low Pg concentration (p = 0.045).
Table 4
Case numbers (n) and frequencies (%) of low (<) and high (≥) levels of estradiol and progesterone (Es and Pg) (pmol/l) and Pg/Es ratios depending on low (<) and high (≥) levels of the studied antibodies and antibody ratios in postmenopausal breast cancer patients
So the disruptions of normal hormonal balance and normal immune balance were interconnected critical events in the breast carcinogenesis.
Interactions of antibodies to benzo[a]pyrene, estradiol and progesterone in postmenopausal healthy women and breast cancer patients
Interactions between levels of antibodies to Bp, Es and Pg were studied (Table 5). Positive linear correlations were found between levels of IgA-Bp1 and IgA-Es1 and IgA-Pg1 in BCP and HW (rs = 0.65-0.78, p < 0.0001). IgA-Es1 correlated with IgA-Pg1 in both compared groups (rs = 0.71-0.75, p < 0.0001). IgA-Pg1/IgA-Es1 ratios in turn correlated with IgG-Pg2/IgG-Es2 in HW (rs = 0.42, p < 0.0001). Interaction of these ratio in BCP was weaker (rs = 0.11, p = 0.02).
Table 5
Correlations between levels of studied antibodies in postmenopausal breast cancer patients and healthy women
It means that IgA-Bp1 could influence IgA-Es1 and IgA-Pg1 formation and IgG-Pg2/IgG-Es2 ratio in HW. The influence of IgA-Bp1 on IgG-Pg2/IgG-Es2 ratio was disrupted in BCP.
Discussion
It is well known that proliferation of the normal and malignant mammary cells depends on cooperative genomic and non-genomic actions of Es and Pg on the corresponding cytoplasmic and membrane receptors [14-16]. It was shown that Pg could inhibit the proliferative effect of Es on the breast line cells and xenografts of mammary tumors [17]. Evidently that resulting action of Es and Pg in women depends on their balance in the blood serum. This proposition was confirmed by our first result: a high individual ratio Pg/Es (> 4) was observed in BCP (31.4%) significantly more rarely than in HW (51.2%, p < 0.0001). So we categorized the high Pg/Es ratio in HW as “hormonal balance”, and the low Pg/Es ratio in BCP as “hormonal disbalance”.
Previously the associations of Abs to Es and Pg with breast cancer were described [11]. It was assumed that Abs to Es could stimulate but Abs to Pg could inhibit the carcinogenesis. This proposition was confirmed by our second result: high personal IgA-Pg1/IgA-Es1 ratios (> 1) were observed in BCP significantly more rarely (17.8%) than in HW (31.7%, p = 0.006, OR = 0.5). Immunoanalysis of the corresponding anti-idiotypic Abs had shown that high individual IgG-Pg2/IgG-Es2 ratios (> 1) were found in BCP significantly more rarely (19.6%) than in HW (37.8%, p = 0.005, OR = 0.4). It means that the predominance of IgA-Pg1 and IgG-Pg2 levels over the IgA-Es1 and IgG-Es2 levels was associated with breast carcinogenesis. The high IgA-Pg1/IgA-Es1 and IgG-Pg2/IgG-Es2 ratios in HW could be considered as “immunological balance”. The low ones in BCP could be considered as “immunological disbalance”.
The model Abs against Es and Pg modulated their concentrations in the blood serum and biological effects in immunized animals [7, 8]. For example, the immunization against Es was the primary cause of anti-Es Ab formation and extended the interval between breast cancer cell implantation and tumor growth in rats [18]. The non-genomic effects of an anti-idiotypic mAb as an Es mimetic in female cells were described [19]. The model Abs against Es receptor (analog of anti-idiotypic Abs to Es) blocked the rapidly nongenomic effects of Es in breast cancer cells MCF7 associated with tumorigenicity [20]. Abs to Es receptor purified from BCP sera was able to induce MCF7 cell proliferation [12].
So it would be logical to assume that Es and Pg concentrations in the women’s blood serum depended on specific Abs and corresponding anti-idiotypic Abs. Our third result confirmed this proposition. It was found that high individual Pg/Es ratios (> 4) were revealed in HW with high IgA-Pg1/IgA-Es1 ratios being more frequent (83.3%) than low Pg/Es ratios (16.7%, p = 0.001). The same frequencies of the high and low Pg/Es ratios in HW with the high IgG-Pg2/IgG-Es2 ratios were 83.9% and 16.1% respectively (p < 0.001). It means that hormonal balance depends on immunological balance in HW.
In contrast to HW there were no associations between Pg/Es ratios and IgA-Pg1/IgA-Es1 and IgG-Pg2/IgG-Es2 ratios in BCP. It means that immunological regulation of hormonal balance was disrupted. Evidently it was due to inversion of the studied Ab actions at the hormone level. Indeed, high IgA-Pg1/IgA-Es1 ratios were associated with low Es concentration (< 200 pmol/l; 75% of cases) and with high Pg concentration (≥ 800 pmol/l; 91.7%) in HW in contrast to high Es concentration (≥ 200 pmol/l; 73.4%) and low Pg concentration (< 800 pmol/l; 54.4%) in BCP. High IgG-Pg2/IgG-Es2 ratios were associated with low Es levels (71.0%) and with high Pg levels (90.3%) in HW in contrast to high Es levels (66.7%) and low Pg levels (69.0%) in BCP.
The interactions of Bp with Es in carcinogenesis were described: most of the Bp derivatives bound to Es receptors and several of them had estrogenic or antiestrogenic activity [21, 22], while Es promoted Bp-induced effects [23, 24]. So it was interesting to study the interaction of Abs to Bp with Abs to Es and Pg. Here the strong linear positive correlations of IgA-Bp1 levels with IgA-Es1 and IgA-Pg1 levels were revealed in HW and in BCP (rs = 0.65-0.78, p < 0.0001). IgA-Pg1/IgA-Es1 ratios correlated with IgG-Pg2/IgG-Es2 ratios in a medium manner in HW (rs = 0.42, p < 0.0001). The same correlation was low but significant in BCP (rs = 0.11, p < 0.0021). That means that formation of Abs to environmental Bp and to endogenous steroids are real interconnecting processes. The formation of idiotypic Abs1 and anti-idiotypic Abs2 to steroid hormones are interconnected only in HW, but this relationship is disrupted in BCP. This disruption is perhaps the cause of disturbance of immune-hormonal relationships in breast carcinogenesis.
The hypothetical immuno-hormonal network is schematically shown in Figure 1.
Fig. 1
Immuno-hormonal network induced by environmental chemical carcinogens (Bp – benzo[a]pyrene, Es – estradiol, Pg – progesterone, AhR – aryl hydrocarbon receptor, ER – estrogen receptor, PR – progesterone receptor, Ab-1 – antibodies, Ab-2 – anti-idiotypic antibodies, CYP – cytochrome P450 enzymes)
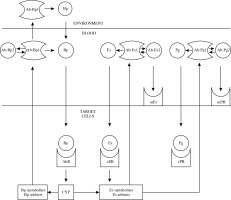
Environmental chemical carcinogens (Bp) penetrate into the bloodstream through the surface epithelium, reach the target cells, react with cytoplasmic receptors (AhR) and stimulate the activity of cytochrome P450 enzymes (CYP). The genotoxic Bp metabolites form the Bp adducts, inducing Abs-Bp1 synthesis. The serum Abs-Bp1 stimulate the penetration of environmental Bp into the blood serum and thus stimulate the malignization of the target cells. Anti-idiotypic Abs-Bp2 induced by Abs-Bp1 modulate the interactions between Bp and Ab-Bp1. The mucosal Ab-Bp1 bind the Bp and thus inhibit Bp transport into the target cells and the following malignization.
The stimulation of CYP by Bp leads to formation of Es metabolites and Es adducts. Es adducts induce the synthesis of Abs-Es1 and/or Abs-Pg1 depending on individual characteristics of the immune system. Abs-Es1 and Abs-Pg1 modulate the serum concentrations of Es and Pg depending on the Ab1 levels and affinities. The cooperative biological action of Abs-Es1 and Abs-Pg1 (stimulation or inhibition of carcinogenesis promotion) depends on their personal ratio.
Anti-idiotypic antibodies Abs-Es2 and Abs-Pg2 induced by Abs-Es1 and Abs-Pg1 modulate the interactions between steroid hormones and specific Abs1. At the same time Abs-Es2 and Abs-Pg2 react with membrane receptors (mER and mPR). The following non-genomic effects mimicking the Es and Pg actions depend on Ab-Pg2/Ab-Es2 ratio.
Therefore, the immune-hormonal network consists of idiotypic and anti-idiotypic Abs specific to the environmental chemical carcinogens and to the endogenous steroid hormones, on the one hand, and steroid hormones with their cellular receptors, on the other hand. The normal immune-hormonal balance supports the real adaptation of the organism to the environmental carcinogens and inhibits the initiation and promotion of carcinogenesis. The disturbance between certain elements of this network (immune-hormonal disbalance) could stimulate carcinogenesis.
Further studies of immune-hormonal interaction could be helpful for understanding the pathogenesis of other carcinogen-induced steroid-dependent diseases in humans.


