Coronary artery bypass graft (CABG) is the most commonly performed cardiac surgery procedure [1]. Approximately 62 procedures are performed per 100,000 inhabitants in Europe each year [2]. However, the complication rate is still relatively high, even after scheduled CABG [3].
Among the many problems related to CABG, the most important aspect, which might be affected by anesthetic care, is postoperative pain [4]. Many patients still suffer from severe pain after CABG procedures [5]. Regional anesthetic techniques are effective in pain alleviation and are associated with decreased mechanical ventilation time [6]. Still, most cardiac surgery patients are mechanically ventilated under sedation during the postoperative period [4]. Prolonged mechanical ventilation is associated with postoperative respiratory complications [7].
Effective pain treatment is one of the mainstays of the Enhanced Recovery After Surgery (ERAS) protocol [8]. However, this strategy involves a simultaneous modification of many aspects of perioperative care.
Our previous study showed that the implementation of the ERAS protocol was possible in patients undergoing mitral/tricuspid valve repair via the right mini-thoracotomy [9]. For the mini-thoracotomy, the institutional ERAS protocol consisted of patient education, avoidance of prolonged pre-op fasting, and pharmacological premedication. Moreover, in this study, we used a unilateral erector-spinae-plane (ESP) block before the operation and continuous infusion of remifentanil during the surgery and for two hours during the early postoperative period, followed by patient-controlled analgesia with intravenous oxycodone. The ESP block provided efficient pain control, enabling a significant reduction in the perioperative opioid dosage, early extubation, and a shorter patient stay in the ICU. In the conclusion of our previous study, we stated that the usefulness of ESP block in other types of procedures needs to be studied more extensively.
Our current study aimed to investigate whether a preemptively performed, bilateral ESP block combined with intraoperative remifentanil infusion could be useful and safe in patients undergoing off-pump CABG (OP-CABG). These patients were compared with a retrospective cohort that was anesthetized with fentanyl, but without any regional analgesia techniques.
The main goal of the study was to compare mechanical ventilation time in the two groups of patients. The other outcomes included ICU and hospital stay, postoperative drainage time, postoperative troponin T level, pain severity, total opioid consumption, and perioperative complications in both patient groups.
METHODS
This was a prospective cohort feasibility study conducted in a tertiary cardiac surgery department. Before patient recruitment, the study protocol was approved by the Bioethics Committee of the Medical University of Lublin in Lublin, Poland (permit number KE-0254/219/2018). Informed consent was obtained from each patient, and the study was conducted in accordance with the tenets of the Declaration of Helsinki for medical research involving human subjects.
Participants
The patients subsequently undergoing OP-CABG had perioperative treatment according to the institutional ERAS cardio protocol (ERAS group). All patients signed the written informed consent form. Their perioperative course was compared with the retrospective data from patients who had the same surgery performed by the same surgeon, but before the ERAS protocol implementation (standard care group).
Intervention
In the ERAS group prior to the induction of general anesthesia, a single-shot ESP block was performed bilaterally as described in our previous study [9], and 0.2 mL kg-1 of 0.375% ropivacaine (Ropimol, Molteni, Italy) was administered on each side. The total volume of local anesthetic solution did not exceed 40 mL.
Induction of general anesthesia was provided with 0.2–0.4 mg kg-1 of etomidate (Hypnomidate, Janssen-Cilag International NV, Belgium), 0.4–0.6 µg kg-1 of remifentanil (Ultiva, GlaxoSmithKline, UK), and 0.6 mg kg-1 of rocuronium (Esmeron, NV Organon, The Netherlands). Sevoflurane 0.5 MAC (age-adjusted, Sevorane, Abbvie, USA), remifentanil infusion according to the target control protocol (4–8 ng mL-1), and incremental doses of rocuronium were used for maintenance.
Patients received an intravenous bolus of oxycodone (0.1 mg kg-1) about 30 minutes before the end of the surgery. Remifentanil infusion was decreased to the target plasma concentration of 0.5–2 ng mL-1 and continued for 60–120 minutes after the patient’s transfer to the ICU. During this period, respiratory support was continued, and patients were observed for excessive postoperative bleeding, hemodynamic instability, tachypnea, and/or excessive respiratory muscle efforts. Sixty to 120 minutes after the end of the surgery, remifentanil infusion was discontinued. The ventilator mode was changed to continuous positive airway pressure (set to 5 cm H<sub>2</sub>O [0.5 kPa] and fraction of inspired oxygen 0.25). The accepted rate of breaths was more than 10 and less than 20 per minute, and the accepted tidal volume was more than 6 mL kg-1. After 30 minutes of spontaneous breathing, an arterial blood gas sample was collected. The accepted pO2 was at least 60 mm Hg, and the accepted CO2 was less than 50 mm Hg. If all elements were fulfilled, the patient’s trachea was extubated. Patient-controlled analgesia (PCA) was started via a pump that supplied incremental bolus doses of oxycodone (1 mg per bolus dose, lock-out time at seven-minute intervals, no basal infusion) during the first postoperative day. Patients were ambulated within the first 24 postoperative hours and transferred to the surgery ward by the end of the first postoperative day. The chest drain was removed approximately 10–12 hours after the patient’s extubation, according to the surgeon’s discretion.
In the standard care group, none of the regional anesthesia techniques were done before the surgery. Induction of general anesthesia was performed in a manner similar to the ERAS group; however, fentanyl (1–3 µg kg-1; Fentanyl WZF, Polfa Warszawa, Poland) was used instead of remifentanil. Additional doses of fentanyl were administered according to the anesthesiologist’s discretion. Patients in the standard care group were transferred to the ICU and sedated with continuous infusion of propofol and fentanyl until the other day. Before discontinuation of mechanical ventilation, 0.1 mg kg-1 of morphine (Morphini Sulfas WZF, Polfa Warszawa, Poland) was given and the PCA pump was started (1 mg per bolus dose, lock-out time at seven-minute intervals, no basal infusion).
In both groups, pain management also included one gram of intravenous paracetamol every six hours. The nurses used the numerical rating scale (NRS) at regular intervals to evaluate postoperative pain.
Outcomes
The primary outcome of this study was the postoperative mechanical ventilation time in both groups of patients. The secondary outcomes included ICU and hospital stay, postoperative drainage time, postoperative troponin T level, pain severity evaluated via the NRS, the total consumption of opioids, and perioperative complications in both groups of patients.
Pain severity was measured five times. It was first measured after the extubation, when the PCA pump was started, and then it was measured in regular six-hour intervals. The pain was measured on the NRS (0–10) by nurses who were not involved in the study.
Two different opioids, oxycodone and morphine, were used in the postoperative period; thus direct comparison of these analgesics was not possible. However, because 1 mg of morphine is comparable to 1 mg of oxycodone and the same PCA pump lock-out was set in both groups, we decided to compare the positive demands of each patient (i.e., the number of boluses of each opioid received by patients in the standard care group and the ERAS group in 24 hours).
The current trial was planned as a feasibility study. The data obtained here will help determine the sample size for further studies.
Statistical analysis
Student’s t-test was used to analyze parametric data. These data were presented as means and 95% confidence intervals. Statistics derived from nonparametric data were calculated using the Mann-Whitney U test and are presented as medians and interquartile ranges. All measurements were performed using Statistica 13.1 software (Stat Soft. Inc., Tulsa, United States).
RESULTS
The study was conducted between November 2018 and July 2019. Overall, 57 patients were analyzed, 29 in the ERAS group and 28 in the standard care group. Only one patient declined to participate in the study. Patient demographics are presented in Table 1. No difference was found between the ERAS and the standard care groups.
TABLE 1
Patient demographics
[i] The table presents patient demographics and preoperative results of ASA, CCS, and NYHA scoring systems. Patient age, body mass, and height are presented as means and confidence intervals. Statistics for these parameters were calculated with Student’s t-test. Results of ASA, CCS, and NYHA were presented as medians and interquartile ranges and calculated with the Mann-Whitney U test
Anesthesia and surgery time did not differ between the ERAS and the standard care groups. Anesthesia and surgery time was 4 (3.0–4.4) h and 3 (2.2–3.5) h in the standard care group and 4 (3.0–4.0) h and 3 (2.1–3.1) h in the ERAS group, respectively.
Primary outcome
The mechanical ventilation time was significantly shorter in the ERAS group (1 [1–3] h) than in the standard care group (10.5 [8–13.3] h; P = 0.00001).
Secondary outcomes
Patients in the ERAS group spent less time in the ICU (20 [16–24] h) compared to the standard care group (48 [36–48] h; P = 0.00001). Moreover, the ERAS patients were discharged from the hospital before the standard care patients. The mean hospital stay was 7 [6–8] days in the ERAS group and 10 [8–12] days (P = 0.0004) in the standard care group.
The pain intensity was significantly lower at each evaluation. The detailed pain severity description is presented in Table 2. Moreover, the PCA demand was lower in the ERAS group than in the standard care group. Patients in the ERAS group and the standard care group received 3 (3–6) and 25 (20–25) opioid boluses, P = 0.000001, respectively.
TABLE 2
Pain severity
[i] The table presents pain intensity evaluated using the numerical rating scale (NRS) where 0 = no pain, 10 = worst imaginable pain. The first assessment was done immediately after the patients’ extubation when the PCA was started.
The postoperative drainage time was significantly shorter in the ERAS group. Thoracic drains were removed after 12 (11–17) h in the ERAS group and 23 (19–27) h (P = 0.00001) in the standard care group. Postoperative drainage volume was comparable between the groups and was 480 (400–550) mL in the standard care group and 400 (300–520) mL in the ERAS group.
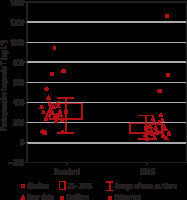
Postoperative troponin T concentration was significantly lower in the ERAS group (135 [94–197] ng L-1) than in the standard care group (305 [240–389] ng L-1; P = 0.000061). No difference was found between groups in the preoperative troponin level (Figure 1).
Perioperative complications
Several serious adverse events occurred in our patients, all in the standard care group. Two patients required reoperation due to severe bleeding, two had postoperative pneumonia, likely ventilatory-associated, and three developed symptoms of hyperactive delirium. No difference was noted between groups regarding the number of transfused blood products.
DISCUSSION
The results presented in the study showed the beneficial effects of ERAS protocol implementation in patients undergoing OP-CABG surgery. The time of mechanical ventilation, thoracic drainage, and ICU and hospital stay were shorter in the ERAS group than in the standard care group. The pain was less intense in the ERAS patients, and the postoperative opioid demand was reduced. Moreover, the increase of the troponin T concentration in plasma was significantly smaller in the ERAS group than in the standard care group. This suggests less myocardial damage due to ischemia-reperfusion, which inevitably occurs during the procedure. Finally, more serious complications occurred in the standard care group in the postoperative period.
We believe that these significant differences in patient outcomes were possible due to ERAS protocol implementation. Among the many elements contained in the protocol, the ESP block and remifentanil were the most important. The feasibility of this regional blockade was illustrated in our previous study [9].
To our knowledge, the current study can be compared with two others regarding ESP block utilization in cardiac surgical patients. In the study by Krishna et al., the ESP block was compared prospectively to a group without any regional analgesia technique [10]. The authors of this paper presented some beneficial effects of the ESP block, including shorter mechanical ventilation time and lower pain intensity – similar to our study’s findings. However, in contrast to the current study, fentanyl was used in both groups as intraoperative and rescue analgesia in the postoperative period. Moreover, PCA with morphine or oxycodone was not a part of the study protocol.
Continuous epidural analgesia was compared with a continuous bilateral ESP block in the study by Nagaraja et al. [11]. In this article, patients in the ESP group had lower pain intensity at 24, 36, and 48 hours at rest and during coughing. Like the previously mentioned study, fentanyl was used perioperatively in both groups and PCA was not implemented in the postoperative period.
Importantly, both studies showed that the ESP block could be performed easily and did not cause any relevant complications. However, more studies are necessary to establish the role of the ESP block in cardiac surgery and compare it to other regional anesthesia techniques.
Although fentanyl is still the opioid of choice for most cardiac anesthetists, the results of numerous trials showed a positive impact of remifentanil on postoperative troponin level and perioperative complications in cardiac surgery [12]. It seems that continuous remifentanil infusion alleviates sympathetic input, which is caused by surgical stress [13]. Thus, fluctuations in heart rate and blood pressure are reduced in patients on remifentanil during the surgery. The present data confirm our previous results, suggesting possible cardioprotection provided by the combined regional/remifentanil anesthesia [14].
It appears that most of the relevant postoperative problems in the standard care group might be entailed by prolonged mechanical ventilation and sedation. Many studies have shown that both delirium and pneumonia are associated with longer mechanical ventilation under anesthesia/sedation [15, 16]. It might be reasonable to shorten sedation and mechanical ventilation time to reduce some of the perioperative complications.
The one-year experience after implementing the ERAS protocol in cardiac surgery is described in the study by Williams et al. [17]. The authors of this article presented a significant improvement in patient outcome after inclusion of several elements, including preoperative patient education, carbohydrate loading before general anesthesia, opioid-sparing analgesia, goal-directed perioperative insulin infusion, and bowel regimen. Most perioperative process elements that should be considered by multi-specialist cardiac surgery teams are summarized in the guidelines by Engelman et al. and in the recommendations by Gregory et al. [18, 19]. Interestingly, in both articles, regional anesthesia techniques are mentioned only as a potential part of a multimodal pain management strategy, not as routinely implemented methods.
Our study has some limitations. First, it was not a randomized controlled trial; thus, patients were recruited in the series. This increased the risk of selection bias. Moreover, the standard care group was analyzed retrospectively. More than one intervention/modification differed in both groups, including the bilateral ESP block in the ERAS group, different opioids during anesthesia and surgery, and prolonged mechanical ventilation under sedation in the standard care group. Pain intensity was evaluated in the same time interval, but the first pain assessment in the standard care group occurred much later than in the ERAS group due to the prolonged mechanical ventilation period.
CONCLUSIONS
The ERAS protocol implementation in OP-CABG surgery can improve patient outcomes. This rationale is supported by the results presented in the current study, including the shorter time of mechanical ventilation, thoracic drainage, and ICU and hospital stay. Moreover, pain management was improved in patients who were treated according to the ERAS protocol. Finally, the new strategy might decrease postoperative troponins and reduce complication rates. Our study confirmed that introduction of the ERAS protocol was feasible and would allow for a fully-powered study.