Introduction
Epidemiological studies suggest that obesity is a risk factor for osteoarthritis (OA), particularly knee osteoarthritis (KOA) [1-6]. Obesity results in high mechanical joint loading and elevated risk of KOA [3, 7]. Interestingly, studies have also suggested that altered metabolic conditions associated with obesity increase the production of growth factors and cytokines that are associated with KOA pathology [8, 9]. Moreover, the presence of OA in the hand joints of obese subjects [10, 11] suggests that mechanical factors are not the only type of factor in KOA progression. To date, however, the mechanisms of this effect have not been well established.
Synovial cells mainly consist of two populations, macrophages (Mφ) and fibroblasts [12-14]. In particular, Mφ contribute to synovial inflammation via inflammatory cytokine production in the synovial membrane (SM) [15-17]. CD5L has been identified as a Mφ-secreted protein [18] and plays a role in modulation of the recruitment of Mφ and inflammation [19]. CD5L is detected in both mouse and human serum [20-23] and is increased in the serum of obese mice [23]. CD5L protein in the synovial fluid (SF) of KOA patients was 3.3-fold higher than that in rheumatoid arthritis patients [24]. However, it is unclear whether cells in the SM express CD5L or whether CD5L expression is increased in the SM of KOA patients with obesity.
Here, we investigated CD5L expression in the SM of obese KOA patients.
Material and methods
Subjects
Ninety subjects who underwent primary total knee replacement (TKR) at our hospital participated in this study. SM was extracted during TKR from subjects with radiographically defined KOA. The protocol was approved by the Ethics Review Board of Kitasato University (approval number: B13-113), and participants provided written informed consent to participate in this study. The patient demographic profile is summarized in Table 1. All subjects were allocated to three groups based on body mass index (BMI): normal weight (NW, < 25 kg/m<sup>2</sup>), overweight (OW, 25-29.99 kg/m<sup>2</sup>) and obese (OB, ≥ 30 kg/m<sup>2</sup>), according to the World Health Organization (WHO) BMI classification.
CD5L expression in synovial membrane
RNA extraction, cDNA synthesis and quantitative RT-PCR (qRT-PCR) were conducted using previously reported methods [17]. CD5L primer pair sequences (product length: 152 bp) used in qRT-PCR analysis were: CD5L-Sense (5'-CTT GCC ATT TGC ACC AGA CC-3') and CD5L-antisense (5'-GCC ACG TCC TTA ATG TCC CA-3'). CD14 and GAPDH primers were synthesized based on previous studies [17]. mRNA expression levels of CD5L in the SM were determined by normalization to that of GAPDH using the ΔΔCT method. We compared the expression of CD5L in the SM among the three groups. To evaluate the effect of aging on CD5L expression, patients were also grouped into two age groups, < 65 and ≥ 65 years, and CD5L expression in the two groups was compared.
CD5L-expressiong cells in synovial membrane
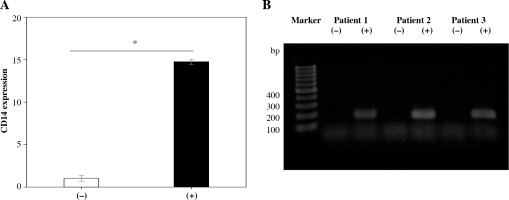
To evaluate CD5L-expressing cells, the CD14-positive (Mφ) rich fraction was isolated from 3 SM samples. Following collagenase digestion, SM-derived cells were reacted with biotinylated anti-CD14 antibody, washed twice with PBS, streptavidin-labelled with magnetic particles (BD Biosciences, Tokyo, Japan), and reacted for 45 minutes at 4°C in a magnetic separation device (IMag, BD Biosciences). RPMI medium was added to the cell suspension to collect CD14– fractions, and an additional 3 ml of RPMI was added to collect the CD14+ fraction by removing the tube from the magnetic field. The CD14+ and CD14– fractions were obtained by centrifugation at 270 × γ for 5 minutes, and used to evaluate CD14 and CD5L expression using qRT-PCR. To confirm the CD5L expression, qRT-PCR products were subjected to electrophoresis and stained with ethidium bromide.
Statistics
All statistical analyses were done using SPSS 25.0. Categorical variables were analyzed using the χ2 test (two groups) and Fisher’s exact test (three groups). Continuous variables were compared using the t-test (two groups) and Bonferroni post hoc test (three groups). Statistical significance was defined by p < 0.05.
Results
Patient demographic profiles in NW, OW, and OB groups
Following patient allocation to the three WHO BMI classification groups, those in the OB group were significantly younger than those in the NW group (p = 0.016, Table 1). Kellgren/Lawrence (K/L) grade 2/3/4 ratio and male/female ratio and were both similar among the groups (Table 1).
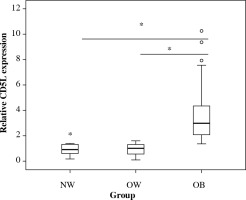
CD5L expression in SM of NW, OW, and OB groups
To determine whether CD5L expression is elevated in obese KOA patients, we analyzed expression levels in the SM of KOA patients. CD5L expression was significantly higher in the OB group than in the NW and OW groups (p < 0.001 and p < 0.001, respectively), but comparable between the NW and OW groups (p = 0.999) (Fig. 1).
Fig. 1
Effect of obesity on synovial CD5L expression. Asterisks (*) indicate a significant difference of p < 0.05 between the groups. Small circles indicate mild outliers (greater than 1.5 interquartile ranges from an edge of the box). Stars indicate extreme outliers (greater than 3.0 interquartile ranges from an edge of the box)
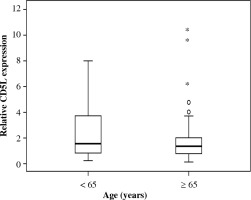
Patient demographic profiles in the < 65 and ≥ 65 years groups
Kellgren/Lawrence (K/L) grade 2/3/4 ratio and male/female ratio and were both similar between the groups (Table 2).
CD5L expression in SM of < 65 and ≥ 65 years groups
Age significantly differed between the OB and other groups. To investigate the effect of aging, we compared CD5L expression between the < 65 and ≥ 65 years groups. There was no difference between the two groups (p = 0.629) (Fig. 2).
Expression of CD5L in synovial Mφ
qRT-PCR analysis also revealed that CD14 mRNA levels in CD14+ fractions from the SM of KOA patients were higher than in CD14– fractions (p = 0.037, Fig. 3A). CD5L expression was observed in CD14+ cells in all patients. However, there was no detection in CD14– fractions in any patients (Fig. 3B).
Discussion
Epidemic research has suggested a link between obesity and KOA progression [1, 25, 26]. KOA patients with obesity are approximately 7 times more likely to experience KOA progression than those of normal weight [1]. A meta-analysis revealed that the odds ratio for obese subjects with KOA development was 2.6 times that of normal weight subjects [25]. In the present study, KOA patients with obesity who underwent total knee arthroplasty (TKA) were significantly younger than OW and NW subjects. Together with previous reports, our observations suggest that obesity is associated with the pathology of KOA.
Inflammation plays a role in the progression and pain of KOA. Among findings to date, obesity was associated with synovial inflammation in human and animal studies [27, 28]; a high-fat diet led to synovial inflammation and promoted OA in rats [28]; and interleukin 6 (IL-6) levels were higher in the SF of obese hip OA patients than in hip OA patients of normal weight [27]. Regarding CD5L, levels were elevated in the course of metabolic and cardiovascular pathologies, such as in adipose tissue in obese mice [19] and in regions of atherosclerosis in mice [29]. CD5L contributes to Mφ infiltration and inflammation in mice [19]. In the present study, CD5L expression in KOA patients with obesity was significantly higher than in normal and overweight patients. Obese groups contained a younger population than the normal weight group; however, CD5L expression did not differ between them. We speculate that CD5L contributes to synovial inflammation in OA patients with obesity, and that the elevation of CD5L in SM may partly explain the relationship between KOA pathology and obesity.
Synovial Mφ contributes to synovial inflammation via the production of inflammatory cytokines and thereby contributes to OA pathology [15, 16, 30]. CD5L supports the survival of infiltrating macrophages, and overexpression of CD5L in CD5L transgenic mice remarkably increased the number of Mφ in tissues [31]. CD5L deficiency also results in a reduction in macrophage infiltration in adipose tissue in obese mice [19]. In the present study, CD5L was specifically expressed in synovial Mφ. CD5L may play an important role in synovial inflammation through the Mφ in KOA.
In this study, we found that CD5L expression was elevated in SM of KOA patients with obesity. Further investigation of the precise role of CD5L in the relationship between KOA pathology and obesity is required.