Introduction
Both acute thrombotic and chronic critical lower limb ischaemia are associated with high morbidity and mortality, and also with a high risk of unsuccessful revascularisation of the limb, requiring its amputation. Routine management of both types of limb ischaemia consists of anticoagulation followed by open surgical repair of occluded arteries (usually bypass grafting) [1–4]. Yet, such a treatment in patients with acute thrombotic ischaemia is associated with an amputation rate at the level of 10–70% and in-hospital mortality even as high as 15% [5]. In patients with critical limb ischaemia the amputation rate after surgical revascularization is at the level of 20–25% and the 2-year mortality rate in these patients is about 50% [2, 5, 6]. Although local thrombolysis is associated with better clinical outcomes and currently it is preferred to surgical revascularization [7–10], not all occlusions can be opened by thrombolytic agents. Those primarily atherosclerotic poorly respond to thrombolysis. Moreover, even if thrombolysis is successful, these arteries usually re-occlude and stent implantation is often required, which carries another problem ‒ in-stent stenosis or occlusion, primarily associated with intimal hyperplasia within the stent. Mechanical thrombectomy seems to be an alternative treatment modality [5, 11–14], but stent implantation with similar late problems is usually required after this endovascular procedure. The use of drug-eluting balloons (DEB) instead of stents may theoretically overcome clinical problems associated with stents, but it remains unclear how often such a treatment is technically feasible in these challenging patients and how efficient mechanical thrombectomy not augmented with stent implantations is in the long run.
Aim
This post hoc single-centre study was aimed at assessment of the feasibility, safety and efficacy of mechanical thrombectomy followed by application of DEBs for acute thrombotic or chronic critical ischaemia of the lower limbs in the femoropopliteal segment. We also analysed how often the use of rotational thrombectomy enabled an endovascular procedure not accompanied by stent implantation, and whether the utilisation of DEBs instead of stents was associated with a better clinical outcome.
Material and methods
We reviewed our register of endovascular interventions and identified patients with acute thrombotic ischaemia or chronic critical lower limb ischaemia due to occlusions in the femoropopliteal segment, who were managed using mechanical rotational thrombectomy (Rotarex®s device; Straub Medical AG, Wangs, Switzerland). Technical success of mechanical rotational thrombectomy was defined in terms of absence of relevant post-procedural residual stenosis, with cutoff at the level of 50%. Primary-assisted patency rate was defined as exempt from significant stenosis (cutoff at the level of 30%) in the target artery following rotational thrombectomy and additional endovascular interventions during the primary procedure, such as balloon angioplasty and stenting.
Potential risks and benefits associated with such a procedure were discussed with the patients, and all patients gave their written informed consent. Clinical indications for mechanical rotational thrombectomy in our centre included:
occlusions and/or critical stenoses of the distal femoral artery (distally from the profunda femoris artery) or the popliteal artery (with or without involvement of its branches);
atherosclerotic, atherothrombotic and atheroaneurysmatic lesions;
primary lesions and secondary lesions after previous balloon angioplasty or stent implantations.
Exclusion criteria comprised: highly calcified lesions, no adequate vascular access, contraindications for antiplatelet therapy, and lack of the patient’s consent.
In this study we did not include patients presenting with arterial emboli. From June 2014 to November 2016 there were 51 eligible patients, 26 men and 25 women, with a mean age of 69.1 ±11.6 years. Thirteen (25.5%) patients were managed for acute non-embolic occlusions of the distal femoral artery and/or popliteal artery and its branches. Out of these patients, 6 (46.2%) presented with an acutely occluded stent. Thirty-eight (74.5%) patients were admitted to the hospital because of critical limb ischaemia resulting from atherothrombotic lesions at the same level as patients with acute ischaemia. In this group there were 5 (13.2%) patients with thrombotic occlusions after balloon angioplasty and 18 (47.4%) patients with chronically occluded stents. A majority of patients presented with grade 4 and 5 (21 and 25 patients, accordingly) of the Rutherford classification, and 5 patients presented with severe ischaemic ulcers (grade 6 in this classification). The demographic profile of both groups of patients and their co-morbidities are presented in Table I, while localisations and characteristics of arterial lesions are described in Table II.
Table I
Clinical characteristics of patients
Table II
Localisations and characteristics of arterial lesions
Endovascular procedures were performed through ipsi- or contralateral femoral access. Before intervention all patients received 300 mg of clopidogrel and 75 mg of aspirin. During endovascular intervention patients were administered intravenously unfractionated heparin. Dosing of heparin depended on the duration of the procedure. We used 110 cm or 135 cm long 6 Fr Rotarex®s catheters. Firstly we navigated through the occluded segment with a 0.018” guidewire and then performed 2–6 passages of the Rotarex system. After at least 2 passages of the rotational catheter, control catheter angiography was performed. If there was still over 50% stenosis, balloon angioplasty was performed. Afterwards, if there was no major residual stenosis (over 40%) in the target artery and no significant dissection, this area was managed with paclitaxel-coated DEBs, such as Elutax SV (Aachen Resonance, Aachen, Germany) or Luminor (iVascular, Barcelona, Spain). This was a desired strategy, which was possible in 24 patients (49.0%). In 25 (51.0%) patients arteries revealed significant stenoses despite balloon angioplasty, or there were severe (grade C or higher) dissections. These patients underwent stent implantations, which was regarded as a bailout treatment. In addition, 6 (11.8%) patients presenting with over 60% residual stenosis following balloon angioplasty and/or significant peripheral embolisation received alteplase intra-arterially (5 mg as a bolus, and then 15 mg during 12 h). Details regarding results of rotational mechanical thrombectomy with the Rotarex system are given in Tables III and IV.
Table III
Results of rotational mechanical thrombectomy with Rotarex system in patients with acute vs. critical leg ischaemia
Table IV
Results of rotational mechanical thrombectomy with Rotarex system in patients finally managed with drug-eluting balloons vs. those managed with stents.
All patients were assessed before discharge from the hospital. They were discharged with the recommendation of dual antiplatelet therapy with aspirin (75–150 mg daily) and clopidogrel (75 mg daily). Their follow-ups were scheduled 30 days, 6 and 12 months after the procedure. Since there was 1 death and 2 amputations during the hospital stay, only 48 patients were followed up (24 patients managed with DEBs and 22 patients who underwent stent implantation). At each visit patients underwent physical examination, evaluation of degree of limb ischaemia according to the Rutherford classification and duplex sonography of the recanalised arteries. Patients were also evaluated in a case of clinical worsening or delayed wound healing. Clinical worsening, restenosis revealed by sonographic examination and delayed healing of an arterial ulcer were the indications for control angiography and reintervention.
Results
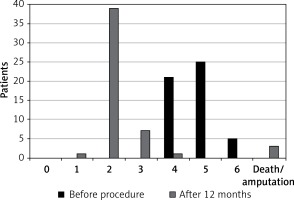
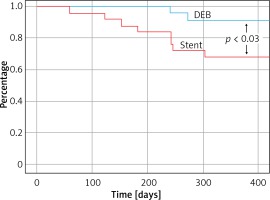
Technical success of mechanical rotational thrombectomy alone was achieved in 20 (19.6%) patientsand there was a 97.1% primary-assisted patency rate (49 patients) after additional balloon angioplasty and stenting. In 2 (4.1%) patients despite recanalisation of the target artery and stenting this procedure clinically failed and in both of them amputations of ischaemic limbs were performed during the hospital stay. Such an unfavourable outcome occurred in 1 patient presenting with acute thrombotic limb ischaemia and in 1 with chronic critical ischaemia. In both patients, in addition to occlusions of the distal femoral artery and popliteal artery, there were occlusions of the branches of the popliteal artery. There was one in-hospital death (mortality rate: 2.0%). This patient died because of intracranial bleeding, which probably was associated with infusion of alteplase. There were local complications associated with mechanical thrombectomy in 5 (9.8%) patients – distal embolisation in 4 patients, which was successfully managed with aspiration and local infusion of alteplase, and perforation of the artery in 1 case, which required implantation of a covered stent. There were neither mortalities nor major adverse events, such as myocardial infarction, stroke or limb amputation in all 48 remaining patients during 12 months of follow-up. At 12 month follow-up the clinical status of the majority of ischaemic limbs had improved. Only 1 (2.1%) patient suffered from rest pain and 7 (14.6%) patients from severe claudication. There were no patients presenting with ischaemic ulcers. Details are given in Figure 1. In 13 patients (27.1%, excluding deceased and amputated patients) duplex sonography revealed occlusions or severe stenoses in the target arteries. These lesions primarily occurred in patients managed for secondary lesions (12 limbs). There was only 1 patient with restenosis after primary intervention. Also, restenoses and occlusions at follow-up were significantly more frequent in patients who underwent stent implantation (10 patients; 45.5%) than in those managed with DEBs (3 patients; 12.5%) – Figure 2. The risk of recurrent lesions was higher in patients with chronic critical lower limb ischaemia (11 patients; 30.6%) than those managed for acute thrombotic occlusions (2 patients; 16.7%). Details are described in Table V. The logistic regression analysis revealed that peripheral embolisation during the procedure and more than 4 passages of the Rotarex system were significantly associated with a higher risk of restenosis/occlusion (hazard ratio: 5.6 and 5.0; p = 0.018 and 0.025 respectively).
Table V
Number of patients presenting with severe restenoses and occlusions at 12-month follow-up (patients who died or had their limbs amputated during first hospitalization were excluded)
Discussion
In this post-hoc analysis we have demonstrated that the majority of severe atherothrombotic lesions in the femoropopliteal segment that result in acute or chronic critical limb ischaemia, and are not highly calcified, can be reopened using mechanical rotational thrombectomy. In our patient series the primary-assisted patency rate after thrombectomy augmented by balloon angioplasty and stenting was as high as 97.1%. in the in-hospital amputation rate was 4.1%. Such management was also safe. In-hospital mortality was 2.0%, which was significantly lower than after an open surgical revascularization. Moreover, in 49% of patients it was possible to avoid stent implantation and instead to manage the area of occlusion with DEB.
Analysis of the clinical outcome of our patients at 12-month follow-up demonstrated that mechanical rotational thrombectomy with the Rotarex system followed by DEB was not inferior to such a thrombectomy assisted by stent implantation. Actually, the results after DEB were better; there were fewer restenoses and no amputations. Yet, stents were implanted in patients with more advanced pathology and therefore these differences should be interpreted with caution. Similarly, although we identified peripheral embolisation during the procedure and more than 4 passages of the Rotarex system as risk factor of reocclusion, these events were probably predictors of more advanced arterial disease, and thus the risk of reocclusion in these patients was higher. Similarly worse late results in patients who required local fibrinolysis in addition to mechanical thrombectomy have already been reported by Kronlage [5].
Large epidemiological studies have revealed a significant risk of major amputation and/or mortality associated with open surgical revascularization for acute and critical leg ischaemia [1–3, 15]. Consequently, local fibrinolysis or endovascular thrombectomy is currently suggested to be a preferred treatment modality [16–20]. In the large study by Freitas et al., who managed with Rotarex 525 patients presenting with acute and subacute ischaemia, with an average length of occluding lesions of 159 mm, there was 1.1% mortality and a 2.3% major amputation rate during 30-day follow-up. Adverse events associated with the treatment occurred in 6.9% of patients and mortality after 1 year was 8% [19]. Similar outcomes were reported by Kronlage et al. They managed 202 patients and in this group amputation-free survival was 94.3% [5].
Although mechanical thrombectomy with the Rotarex system has been demonstrated to be both relatively safe and efficient [5, 7–9, 14, 16–19], it remains to be established how to optimize such treatment. Even if short-term results are encouraging, long-term patency rates in the femoropopliteal segment after standard balloon angioplasty or stent implantation are relatively low. The 1-year reocclusion rate after balloon angioplasty is at the level of 60% [21–28]. Stents do not seem to be a proper solution either. When implanted in this part of the arterial system, especially in the distal part of the popliteal artery or in its branches, a significant proportion of currently available stents occlude in the long run, either because of a fracture, or due to thrombosis and intimal hyperplasia [21–29]. Although novel wire-interwoven Nitinol or helically shaped stents, exhibiting a swirling flow, try to overcome these problems, they are not yet routinely used and their actual long-term advantage remain to be proven [30–35]. On the other hand, long-term patency rates in the femoropopliteal segment after DEBs are higher than after standard balloon angioplasty [36–47], while the problems associated with stents are avoided.
The results of our study suggest that the use of DEB after mechanical thrombectomy for thrombotic acute or critical leg ischaemia resulting from arterial occlusion in the femoropopliteal segment could be a desired treatment strategy. However, it should be emphasized that it was a retrospective analysis and the groups of patients were not fully comparable. A larger prospective study should be designed and performed in order to fully compare the clinical value of DEBs with stents in these challenging patients. Also, probably some novel area-dedicated stents (such as the aforementioned helically shaped ones) should be applied in such a trial.
Conclusions
The short and intermediate term results from this nonrandomised study indicate that the combination of mechanical thrombectomy with DEB is safe and feasible for the treatment of intermediate to long superficial femoral artery/popliteal artery lesions in selected patients with severe limb ischaemia. The DEB group had higher rates of primary patency and freedom from restenosis than the group of patients with stent implantation.