Introduction
Ovarian cancer is the most frequent cause of gynecological cancer related mortality in women [1]. Most ovarian cancers are sporadic and only up to 10% of ovarian cases are familial [2]. The 5-year survival rate for localized ovarian cancer is 93% but the survival rate is as low as 3% for metastatic ovarian tumors [3]. The late diagnosis of the disease and the unavailability of potent therapeutic targets form an obstacle in the treatment of ovarian cancer [4]. MicroRNAs (miRNAs) are 23 nucleotide-long RNA molecules that regulate the expression of a number of genes in humans and other organisms by binding to the messenger RNA and enforcing their post-transcriptional repression [5]. As miRNAs regulate the expression of about thirty percent of the human protein coding genes, they are implicated in different cellular and physiological processes such as cell proliferation and apoptosis [6]. A strong body of evidence suggests that several microRNAs are aberrantly expressed in cancer cells and are considered to be prospective therapeutic targets/agents for the treatment of cancer [7]. Amongst all, miRNA-101 is involved in the proliferation and metastasis of a number of malignancies including endometrial cancer [8]. Moreover, miRNA-101 exerts potent inhibitory effects on the proliferation of hepatocellular cancer cells [9]. Similarly, miRNA-101 suppresses cell progression and enhances the drug sensitivity in patients with breast cancer by targeting MCL-1 [10]. However, the therapeutic potential of miRNA-101 in ovarian cancer has not been investigated. To the best of our knowledge, this is the first report to investigate the role of miRNA-101 as the tumor suppressor in ovarian cancer. It has been observed that miRNA-101 is aberrantly downregulated in ovarian cancer and ectopic expression of miRNA-101 suppresses the proliferation, migration and invasion of the ovarian cancer cells by inhibiting the expression of the phosphatase and tensin homolog (PTEN).
Material and methods
Cell lines and culture conditions
Ovarian cancer cell lines (SW-626, SKOV-3, OVACAR-3, PA-1) and a normal ovarian cell line (SV40) were purchased from American Type Culture Collection (Manassas, VA, USA). The cells were cultured in RPMI 1640 medium (Gibco, Carlsbad, CA, USA) containing penicillin (100 U/ml), streptomycin (100 U/ml) (Sigma-Aldrich, St. Louis, MO, USA), and 10% fetal bovine serum (FBS; Gibco) at 37°C in 5% CO2.
The qRT-PCR analysis
The total RNA from the cervical cancer cell lines was isolated by TRIzol Reagent (Invitrogen) following the manufacturer’s instructions. The cDNA was synthesized using M-MLV reverse transcriptase (Promega, Madison, WI, USA) and amplified with Platinum SYBR Green qPCR Super Mix-UDG reagents (Invitrogen) using the CFX96 sequence detection system (Bio-Rad, Hercules, CA, USA).
MTT cell viability assay
The OVACAR-3 cells were transfected with different constructs and seeded in 96-well plates and treated and incubated for 24 h at 37°C. Following incubation, the cells were incubated with MTT for 4 h. After this the medium was removed and the colored formazan product was solubilized with 200 μl of dimethyl sulfoxide. The viability of the transfected OVACAR-3 cells was then determined by taking the absorbance at 570 nm.
Apoptosis assays
The transfected ovarian cancer OVACAR-3 cells (0.6 × 106) were seeded in 6-well plates and subjected to incubation for 12 h. Following incubation, the OVACAR-3 cells were subjected to incubation for 24 h at 37°C. As the cells sloughed off, 10 μl cell cultures were put onto glass slides and subjected to staining with a 0.5 μl solution of acridine orange (AO) and ethidium bromide (EB) solution. The slides were covered with cover slips and examined with a fluorescent microscope. Annexin V/PI staining of the OVACAR-3 cells was performed as described previously [11].
Cell cycle analysis
The OVACAR-3 ovarian cancer cells were transfected with appropriate constructs and then incubated at 37°C for 24 h. The cells were then subjected to washing with phosphate buffered saline (PBS). Afterwards, the AGS cells were stained with propidium iodide (PI) and the distribution of the cells in cell cycle phases was assessed by a FACS flow cytometer.
Target identification and dual luciferase assay
The miR-143 target was identified by TargetScan online software (http://www.targetscan.org).
Western blotting
The OVACAR-3 cells were firstly subjected to washing with ice-cold PBS and then suspended in a lysis buffer at 4°C and then shifted to 95°C. Thereafter, the protein content of each cell extract was checked by Bradford assay. About 40 μg of protein was loaded from each sample and separated by SDS-PAGE before being shifted to polyvinylidene fluoride membrane. The membranes were then subjected to treatment with TBS and then exposed to primary antibodies at 4°C. Thereafter, the cells were treated with appropriate secondary antibodies and the proteins of interest were visualized by enhanced chemiluminescence reagent.
Results
miRNA-101 is downregulated in ovarian cancer cell lines
The miRNA-101 expression was examined in normal and ovarian cancer cell lines by qRT-PCR (Figure 1 A). The results showed that miRNA-101 was significantly down-regulated (p < 0.05) in all the ovarian cancer cell lines. Furthermore, miRNA-101 was found to be downregulated in ovarian cancer lines by up to 7-fold relative to the normal cell SV40 line. The lowest expression was observed in the case of the OVACAR-3 cell line.
Figure 1
Overexpression of miRNA-101 inhibits the proliferation of ovarian cancer cells. A – Expression of miRNA-101 in normal and four different ovarian cancer cell lines. B – Expression of miRNA-101 in NC or miRNA-101 mimic transfected OVACAR-3 ovarian cancer cells. C – MTT assay showing the % viability of NC or miRNA-101 mimic transfected OVACAR-3 ovarian cancer cells at indicated time intervals. The experiments were performed in triplicate and results are expressed as mean ± SD (*p < 0.05)
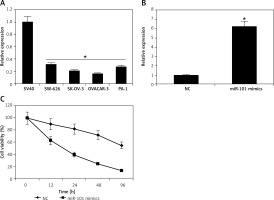
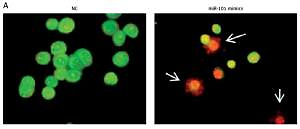
miRNA-101 induces apoptosis and G2/M cell cycle arrest in ovarian cancer cells
Next, we investigated the role of miRNA-101 in ovarian cancer. Therefore, the OVACAR-3 cells were transfected with either the miRNA-101 mimics or NC (negative control). The ectopic expression of miRNA-101 in OVACAR-3 cells was validated by qRT-PCR, which showed that transfection of the OVACAR-3 cells with miRNA-101 mimics caused around 6-fold enhancement of the expression of miRNA-101 relative to NC transfected cells (Figure 1 B). The proliferation rate of the NC and miRNA-101 mimics OVACAR-3 transfected cells was monitored at different time intervals by MTT assay. It was found that transfection of miRNA-101 mimics in the OVACAR-3 cells resulted in a significant decline in the viability of OVACAR-3 cells (Figure 1 C). AO/EB staining of the NC and miRNA-101 transfected OVACAR-3 cells was performed to reveal the underlying mechanism and it was observed that miRNA-101 mimics’ overexpression led to activation of apoptotic cell death of the OVACAR-3 cells (Figure 2 A). In addition, annexin V/PI staining showed that the apoptotic cell percentage increased from 5% in NC transfected cells to about 28% in the miRNA-101 mimic transfected OVACAR-3 cells (Figure 2 B). These results unequivocally indicate that miRNA-101 suppresses the OVACAR-3 cell proliferation by prompting apoptosis. We also determined the cell cycle phase distribution of NC and miRNA-101 transfected OVACAR-3 cells and it was found that the miRNA-101 overexpression caused the arrest of the OVACAR-3 cells in the sub-G1 phase of the cell cycle. The sub-G1 cells increased from 2.11% in NC transfected cells to around to 21.55% in miRNA-101 mimic transfected cells (Figures 2 B, 3).
Figure 2
Overexpression of miRNA-101 triggers apoptosis in ovarian cancer cells. A – AO/EB staining showing induction of apoptosis in NC or miRNA-101 mimic transfected OVACAR-3 ovarian cancer cells. B – Annexin V/PI staining showing percentage of apoptosis in NC or miRNA-101 mimic transfected OVACAR-3 ovarian cancer cells. C – Flow cytometry showing cell cycle phase distribution of the NC or miRNA-101 mimic transfected OVACAR-3 ovarian cancer cells. The experiments were performed in triplicate
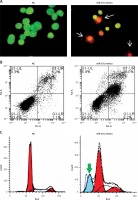
Figure 3
miRNA-101 exerts its effects by targeting PTEN. A – TargetScan analysis showing PTEN as the target of miRNA-101. B – Expression of PTEN in normal SV40 and four different ovarian cancer cell lines. C – Western blot analysis showing the expression of PTEN in NC or miRNA-101 transfected ovarian cancer cells. D – Expression of PTEN in the NC or miRNA-101 transfected ovarian cancer cells. E – MTT assay showing the % viability of the NC or Si-PTEN transfected ovarian cancer cells. The experiments were performed in triplicate and results are expressed as mean ± SD (*p < 0.05)
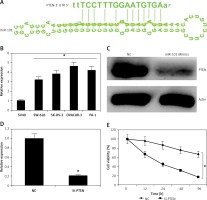
miRNA-101 targets PTEN in ovarian cancer cells
The target of miRNA-101 in ovarian cancer cells was identified by online TargetScan analysis. PTEN was identified as the potential target of miRNA-101 in OVACAR-3 ovarian cancer cells (Figure 4 A) and therefore the expression levels of PTEN were investigated in all the ovarian cancer cell line as well as the normal cell line. It was found that relative to the NC the expression of PTEN was significantly upregulated by 4.5-fold in the ovarian cancer cell lines (Figure 4 B). However, as the OVACAR-3 cells were transfected with the miRNA-101 mimics, the expression of PTEN was considerably downregulated as depicted by the western blot analysis (Figure 4 C). The effects of the PTEN silencing on the proliferation rate of the ovarian cancer OVACAR-3 cells was also investigated. It was found that the silencing of PTEN expression (Figure 4 D) caused a significant (p < 0.05) decline in the viability of the OVACAR-3 ovarian cancer cells (Figure 4 E).
Figure 4
Silencing of miRNA-101 triggers apoptosis and sub-G1 arrest of ovarian cancer cells. A – AO/EB staining showing induction of apoptosis in NC or Si-PTEN transfected OVACAR-3 ovarian cancer cells. B – Annexin V/PI staining showing percentage of apoptosis in NC or Si-PTEN transfected OVACAR-3 ovarian cancer cells. C – Flow cytometry showing cell cycle phase distribution of the NC or Si-PTEN mimic transfected OVACAR-3 ovarian cancer cells. The experiments were performed in triplicate
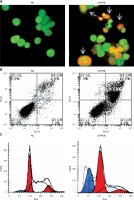
miRNA-101 modulates the PI3K/AKT pathway in OVACAR-3 cells
As miRNA-101 was found to target PTEN and PTEN was a strong regulator of the PI3K/AKT signaling pathway, the expression of AKT and PI3K was determined in NC or miRNA-101 mimic transfected OVACAR-3 cells. It was found that the overexpression of miRNA-101 caused a significant decline in the expression of AKT and PI3K (Figure 5 A).
Figure 5
A – Effect of miRNA-101 overexpression on the expression of PI3K and AKT. B – Effect of PTEN overexpression on the inhibitory effects of miRNA-101 overexpression on the proliferation of the OVACAR-3 cells. The experiments were performed in triplicate and results are expressed as mean ± SD (*p < 0.05)
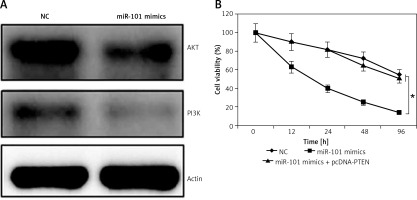
PTEN rescues growth inhibitory effects of miRNA-101 on OVACAR-3 cells
Since the overexpression of miRNA-101 and silencing of PTEN exhibited similar effects on the proliferation of OVACAR-3 cells, it was assessed whether the overexpression of PTEN could rescue the effects of miRNA-101 overexpression in OVACAR-3 cells. Interestingly, it was found that PTEN overexpression in the miRNA-101 mimic transfected OVACAR-3 cells promoted the proliferation of the OVACAR-3 cells. Thus, it was suggested that the inhibitory effects of the miRNA-101 overexpression are directly due to the PTEN suppression (Figure 5 B).
Discussion
Ovarian cancer is a lethal type of malignancy which accounts for approximately 0.3 million new cases and 0.152 million deaths worldwide each year [12]. The clinical outcome is unsatisfactory due its relatively late diagnosis at an advanced stage and the emergence of chemoresistance in cancer cells [3]. The miRNAs control the expression of the majority of the human genes and are involved in a wide array of cellular processes [13]. Because of the importance of the miRNAs in cellular and physiological processes, several studies have revealed the potential of miRNAs as therapeutic targets [14]. Herein, the role and therapeutic potential of miRNA-101 was investigated in ovarian cancer. It was found that miRNA-101 was aberrantly downregulated in the ovarian cancer cells. Previous studies have indicated that downregulated expression of miRNA-101 is associated with poor prognosis and may act as a biomarker of bladder cancer [15]. In addition, miRNA-101 has been shown to be significantly downregulated in salivary gland adenoid cystic carcinoma [16]. Overexpression of miRNA-101 in OVACAR-3 ovarian cancer cells caused a significant reduction in the proliferation rate of OVACAR-3 ovarian cancer cells via induction of apoptotic cell death and sub-G1 cell cycle arrest. Studies carried out previously have shown that miRNA-101 suppresses the proliferation and metastasis of lung cancer cells by targeting ITGA3 [17]. Moreover, miRNA-101 enhances sensitivity of hepatocellular carcinoma cells to doxorubicin-triggered apoptosis by targeting Mcl-1 [18]. In silico analysis together with dual luciferase indicated PTEN to be the potential target of miRNA-101. Herein, we observed that PTEN is highly upregulated in ovarian cancer and miRNA-101 overexpression could suppress the expression of PTEN. Additionally, PTEN silencing could inhibit the growth of OVACAR-3 ovarian cancer cells similar to that of miRNA-101 overexpression. Further, PTEN inhibition was found to be essential for the tumor suppressive effects of miRNA-101 on ovarian cancer cells. Studies have shown PTEN is a negative regulator of PI3K/AKT [19] and therefore we examined the expression in NC as well as miRNA-101 mimic transfected OVACAR-3 cells, and it was found that miRNA-101 overexpression downregulates both PI3K and AKT expression. Finally miRNA therapy is a reliable, safe and cost-effective treatment strategy that would largely be beneficial for the management of different cancer types.
In conclusion, the evidence shows that miRNA-101 is downregulated in human ovarian cancer cells. It inhibits proliferation by inducing apoptosis and sub-G1 cell cycle arrest. miRNA-101 acts a tumor suppressor in ovarian cancer and may prove to be an essential therapeutic target for ovarian cancer.