Introduction
Takayasu arteritis (TA) is a large vessel vasculitis of granulomatous nature and unknown aetiology affecting predominantly the aorta and its major branches. Being diagnosed all over the world, featured with huge differences in incidence and prevalence between geographical regions and genders, it poses a challenge in its successful treatment with good long-term outcomes. The treatment options include pharmacotherapy as well as both open surgical and endovascular procedures, which have to be carefully chosen and adjusted to the patient’s state in order to obtain clinical success [1]. In patients with Takayasu arteritis, multiple supra-aortic branch involvement may lead to ischaemic symptoms of the central nervous system and upper limbs. To decrease the risk of neurological complications, including ischaemic stroke, and to improve the quality of life, an arterial revascularisation is necessary. There is an ongoing debate on the advantages, possibilities, and indications for implementing endovascular and open surgical methods of the treatment [2–4]. In TA patients an open surgical approach is considered superior to endovascular methods in the treatment of long and irregular lesions; however, it might be technically difficult because of inflammation in the vicinity of the target arteries. In Takayasu arteritis, a large percentage of the lesions are relatively short and focal, and therefore feasible for endovascular treatment. However, in our clinical experience, vast majority of impairments in extracranial vessels were longer than usually reported in the literature. What is more, the endovascular approach allows multiple changes to be treated at the same time [5]. Long-term results of both open and minimally invasive methods are satisfactory. They reach almost up to 70% patency in a five-year follow-up in bypass surgery. In endovascular methods, long-term results vary from 0 to 70% of restenosis rate, with a follow-up period of 0.5 to 5 years – improving with the advances of endoluminal techniques and equipment [6–9]. Wrotniak et al. showed that endovascular treatment is safe and effective in symptomatic patients and is featured with a relatively low complication rate of 7.1% in a 30-day period [9]. Despite better long-term outcomes, bypass surgery is related with greater perioperative complication rates. Patients with cerebral ischaemia symptoms are also more prone to acquiring hyperperfusion syndrome, and consequently may develop stroke or brain haemorrhage up to 2 weeks after open procedure [10–12]. Restenosis remains one of the most serious problems in patients with Takayasu arteritis. Contemporarily, it can be quickly diagnosed with non-invasive imaging methods including Doppler ultrasonography, cpmputed tomography (CT), and magnetic resonance imaging (MRI) angiography. Furthermore, it may be treated successfully with both open surgical and endovascular methods with the use of different devices, including cutting, drug-coated balloons, as well as drug-eluting stents [9, 12]. Nowadays, imaging modalities also allow for the diagnosis of early prestenotic disease and active inflammatory processes in the vessel wall [13, 14].
Aim
The aim was to present our own experience in the treatment of an unusually complex and high-risk patient with multiple occlusion of supra-aortic branches, focusing on the technical aspects of the procedures and the decision-making process, as well as to confront it with contemperary medical knowledge.
Is minimally invasive treatment still possible in multiple extracranial artery occlusion? Technical aspects of the treatment
The treatment of patients with multiple aortic arch branch involvement, including their occlusion, has been widely described [7, 8, 15–25]. In most cases of patients with the active inflammatory phase of TA, the immunosuppressive treatment alone may be effective in reducing the cerebral ischaemic symptoms [19–22]. Sometimes surgical treatment is needed [15, 17, 18]. Both methods – endovascular and surgical – are featured with good long- and short-term results, provided that the patients undergo proper treatment [7, 8, 17, 18].
Photo 1 presents a patient with type IIA Takayasu arteritis with the involvement of all aortic arch branches. Imaging examinations including computed tomography, angiography, and Duplex-Doppler revealed occlusions of the innominate artery (IA), both common carotid arteries (CCA), both internal carotid arteries (ICA), and stenosis in the ostium of the left subclavian artery (LSA). The dilatation of the ascending aorta to 45 mm, the aortic arch to 41 mm, both with underlying atherosclerotic lesions, and thrombosed aneurismal dilation, with underlying atherosclerotic plaques resulting in occlusion of the innominate artery, was shown. The right vertebral artery (RVA) presented retrograde flow – right subclavian steal syndrome was diagnosed. The 8 mm wide, patent left vertebral artery (LVA) with antegrade flow was the only vessel providing blood supply to the brain structures which is shown in the Photo 2. The patient was scheduled for elective recanalisation of the innominate artery as the first step of treatment. The occlusion of common and internal carotid arteries initially excluded both the endovascular attempt of carotid recanalisation or surgical bypass to ICA.
Photo 1
A 53-year-old woman with medical history positive for headaches, syncope, vertigo, dizziness, fatigue, and malaise. Intermittent claudication of the right upper extremity, with the unpalpable pulse on the arteries of the extremity in question and the systolic blood pressure difference of about 30 points compared to the left upper limb. Occlusion of the innominate artery (IA), both common carotid arteries (CCA), both internal carotid arteries (ICA), and stenosis in the ostium of the left subclavian artery (LSA). Right-sided Subclavian Steal Syndrome. 8 mm wide patent left vertebral artery (LVA) with antegrade flow is the only vessel providing blood supply to the brain structures
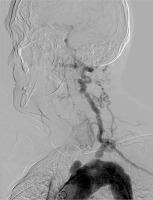
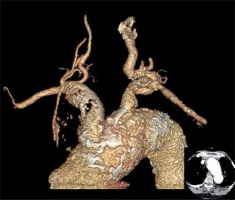
Photo 2
Three-dimensional reconstruction: occlusion of the innominate artery (IA), both common carotid arteries (CCA), both internal carotid arteries (ICA), and stenosis in the ostium of the left subclavian artery (LSA). Right-sided subclavian steal syndrome
Conventional surgical options such as bypass grafting or arterial transposition are related with much higher blood loss than endovascular procedures and more perioperative complications [7, 26]. General anaesthesia is linked with perioperative blood pressure drop, which in patients with multiple aortic arch syndrome might additionally lower the cerebral perfusion [27]. Endovascular procedures do not require general anaesthesia, so its impact on blood pressure is reduced. Therefore, in cases with high perioperative risk assessed using the V-POSSUM scale or possible cerebral ischaemic complications, endovascular methods should be preferred. In our department, the femoral approach is usually chosen because it allows us to examine the whole aortic arch during angiography.
Photo 3 presents the initial recanalisation of the occluded innominate artery with the implantation of a 10 × 60 mm ZILVER stent and subsequent stenting of the vertebral artery with an XPERT 5 × 30 mm stent due to vessel dissection and acute occlusion.
Photo 3
From the right axillar artery and right femoral artery approaches the totally occluded segment of the IA was passed with a guidewire and predilated. Subsequently the 10 × 60 mm ZILVER stent (Cook Medical), which covered the ostium of the RVA, was positioned and post-dilation was performed with the help of a 10 mm balloon catheter. The flow in the IA was achieved but the dissection of the proximal part of the RVA resulting in the occlusion of the vessel in question was shown in the control angiography. Subsequently the guidewire was navigated to the RVA from the femoral approach via the stent mesh, and an XPERT 5 × 30 mm stent (Abbot) was successfully implanted with the use of a neuroprotection system. Final angiography from the right femoral approach demonstrated good position of both stents with good wall apposition, and the antegrade flow in both subclavian and vertebral arteries was confirmed
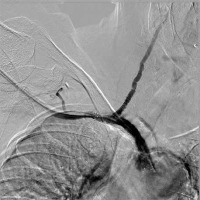
Photo 4 presents endovascular intervention of the left subclavian artery stenosis with the placement of Protege GPS stents – 14 × 20 and 14 × 30 mm – with post dilatation using a Wanda 9 × 40 mm balloon. The results of the treatment are shown in the Photos 5 and 6.
Photo 4
From the right femoral approach, the LSA stenosis was passed with the guide wire, and after predilatation with the help of an 8 × 40 mm balloon catheter two self-expendable Protege GPS stents 14 × 20 mm and 14 × 30 mm (Medtronic) were implanted. Postdilatation using the Wanda 9 × 40 mm balloon (Boston Scientific Corporation) was performed with good haemodynamic and morphological effect in the control angiography
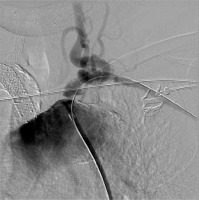
Photo 5
Angiography, lower image – CT scan 3-D reconstruction. Good position of the stents in innominate artery, right vertebral artery, and left subclavian arteries with the antegrade flow in both subclavian and vertebral arteries
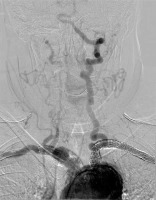
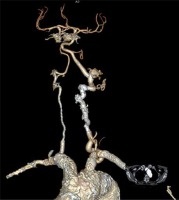
Photo 6
3D CT scan reconstruction after successful recanalisation of both subclavian, right vertebral, and innominate artery
Various authors have discussed the long-term results of endovascular treatment. The surgical treatment is considered to be safe and effective regardless of disease activity [17]. Some authors stress that conventional bypasses are featured with better long-term patency than endovascular options; however, adequate immunosuppression and qualification of patients to surgical treatment may give satisfactory results in both kinds of methods [7, 8]. We prefer the endovascular options first. We introduce conventional surgical treatment after a second endovascular treatment (with stent implantation) failure, if anatomical conditions allow that. In our opinion, the minimally invasive treatment is possible in occlusion of multiple extracranial arteries. Due to the potentially high number of complications of open procedures, it should the first choice in the treatment of high-risk patients.
Discussion and review of literature
Takayasu arteritis, also known as pulseless disease, occlusive thromboaortopathy, Martorell syndrome, or aortoarteritis, is a large vessel vasculitis of granulomatous nature and unknown aetiology affecting predominantly the aorta and its major branches, as well as proximal portions of the pulmonary, coronary, and renal arteries. Rare manifestations of Takayasu arteritis involving the superficial femoral artery have also been described [28]. Cell-mediated inflammatory processes in the arterial wall result in its thickening, fibrosis, stenosis, and thrombus formation, while more acute inflammation can destroy the arterial media, causing vessel dilation and aneurysm formation [29]. It is believed that the first case of TA was described in 1761 by Italian pathologist Giovanni Baptista Morgagni [30]. TA cases are diagnosed all over the world, but huge differences in incidence and prevalence between geographical regions and between genders are observed: in Europe and North America the disease is rather uncommon (prevalence rate of 4.7/million in the United Kingdom), while the highest incidence is observed in East Asia countries – especially in Japan – where about 40 people in 1 million are affected [30–33]. TA is more common in women of childbearing age (the female to male ratio is 8–9 : 1) and Asians [29, 32]. The onset of the disease is mostly observed before the age of 50 years, but TA was also reported in children and in people over 60 years old [32, 34, 35]. Genetic factors in TA have been extensively studied, and several connections have been discovered, for example: HLA B-52 and DR-2 in the Japanese population, HLA B-52 and B-5 in Korea and India, and HLA B-39 in Mexican patients. IL12B has been also described, suggesting an immunogenetic predisposition [36–40].
The American College of Rheumatology 1990 criteria for TA diagnosis include six points:
Onset before 40 years of age.
Claudication of an extremity.
Decreased brachial artery pulse in one or both arms.
Difference of 10 mm Hg or more in blood pressure measured in both arms.
Audible bruit on auscultation over the aorta or subclavian arteries.
Arteriographic evidence of narrowing or occlusion of the entire aorta, its primary branches, or large arteries in the proximal upper or lower extremities.
The presence of at least three of the six criteria is necessary for the TA diagnosis and demonstrates high specificity and sensitivity [41].
Various patterns of large vessel involvement are observed in TA, which vary between geographical regions, mainly due to ethnical differences; in Japanese and Korean patients the aortic arch with its branches are most commonly affected, while in the Indian population the disease affects abdominal aorta and renal arteries [4, 7, 42, 43]. It is reported that in 10–20% of cases with TA a transient ischaemic attack (TIA) or ischaemic stroke is observed [43, 44]. The clinical course of TA can be divided into two phases: an early active inflammatory and late chronic, occlusive period. An early active inflammatory phase may not affect and may not be observed in some patients, and the symptoms in this phase are more often seen in children with TA. It is characterised by duration ranging from weeks to months and possible remitting-relapsing course. Non-specific systemic inflammation symptoms such as low-grade fever, fatigue, malaise, arthralgia, weight loss, loss of appetite, headaches, dizziness, skin rashes, and night sweats may be noticed in this period of the disease. It is worth mentioning that TA is rarely diagnosed in this phase before the symptoms connected with organ ischaemia occur but symptoms such as tenderness along arteries, audible bruits, and aneurysm may suggest the diagnosis of TA [40]. The symptoms connected with vascular insufficiency and impaired blood supply to specific organs are a result of vascular stenosis or occlusion found in the late chronic phase of TA, which follows the inflammatory period [45, 46]. The involvement of the main aortic arch branches may result in decreased or absent pulse, claudication and impaired function of the upper extremities, malaise, vision and orientation problems, and subclavian steal syndromes symptoms: vertigo, presyncope, syncope, neurologic defects, and memory problems [46, 47]. In TA ischaemic heart disease and myocardial infraction may be observed as a result of coronary artery involvement [46, 48]. Chest pain, dyspnoea, coughing, haemoptysis, and congestive heart failure are the result of a disease affecting the pulmonary arteries [46, 49]. Involvement of the arteries supplying blood to abdominal organs may manifest as symptoms of intestine ischaemia, abdominal pains, as well as arterial hypertension or even chronic renal disease in the case of involvement of renal arteries [6, 46, 50]. Despite the significant progress in pharmacological treatment, surgical therapy plays an important role in Takayasu arteritis management [32, 51]. Both open surgical revascularisation including endarterectomy, patch angioplasty, and bypass grafting and endovascular procedures: percutaneous transluminal angioplasty (PTA) and stent-graft or stent implantation should be taken into consideration in TA patients with symptomatic stenotic or occlusive lesions. Those procedures can decrease morbidity and improve long-term results by lowering the number of complications that are the consequences of impaired blood supply to various important organs. A fact worth highlighting is that the preoperative assessment of the activity of the disease plays a key role in the surgical treatment. Optimal immunomodulatory therapy should be administered to achieve the best possible long-term results. PTA is considered a safe and minimally invasive method featured with good short-term results ranging from 81% to 100%, but unfortunately there is a huge risk of restenosis, which occurs in more than two-thirds of patients in late follow-up. A restenosis rate of 17% after a period of 23.7 ±18.4 months in the case of stent placement has been noted [6, 49, 50]. Recently it has been shown that both open surgical and endovascular methods are featured with long-term durability. Dao et al. reported that the results of open surgical and endovascular procedures in follow-up period of up to 10 years are satisfactory, with primary patency rates as follows: in open repair and endovascular groups at 1, 3, 5, and 10 years of follow-up, respectively: 97.3%, 86.2%, 70.5, and 48.8% and 93.3%, 73.1%, 57.5%, and 31.8% [25]. In the literature several indications for surgical intervention with good risk-to-benefit ratio in patients with TA are listed: (1) severe renovascular stenosis causing hypertension; (2) severe coronary artery stenosis leading to myocardial ischemia; (3) extremity claudication induced by routine activity; (4) critical cerebral ischemia and/or critical stenosis of 3 or more cerebral vessels; (5) severe aortic regurgitation; (6) thoracic or abdominal progressive aneurysms > 5 cm in diameter with a tendency for dissection or rupture; (7) severe coarctation of the aorta [32]. The adequate diagnosis of the TA phase is mandatory in proper treatment method selection. Unfortunately, there is no single test to verify this issue, and the coexistence of acute inflammatory with chronic inactive stage may be observed in different vascular beds in the same patient. Therefore, the combination of various diagnostic modalities including contrast-enhanced MRI, CT-scans, duplex Doppler examination, as well as preoperative angiography is recommended. Gadolinium-DTPA-enhanced MRI is thought to detect inflammatory lesions in the arterial wall, providing adequate information concerning the disease. Duplex Doppler examination is useful in identifying arterial wall thickening and provides important haemodynamic data [16, 49, 52]. A PET-CT (positron emission tomography) with 18-fluorodeoxyglucose might be also used to determine the increased metabolic activity suggesting an active inflammatory process in the vessel wall. This method, combined with coregistered CT scans, might be useful in detecting early prestenotic disease [13, 14]. Fields et al. published a work in which they determined the influence of the disease activity on short- and long-term results in patients with TA requiring surgical intervention. The authors concluded that patients with active disease are more prone to reintervention or development of symptomatic disease in another vascular bed. Contemporarily, contrary to previous opinions, the surgical treatment is thought to be featured with safety regardless of disease activity with good long-term survival [17]. Other researchers underlined the fact that surgical procedures improve outcomes in patients with severe disease, in whom two or more complications occur [18]. Kim et al. compared the patency of supra-aortic bypasses with endovascular treatment results, demonstrating higher patency of surgical bypasses in almost 4 years of follow-up (53.3% of restenosis in the endovascular vs. 12.5% in the bypass group). It is worth stressing that conventional surgical treatment was featured with higher risk of early complications [7]. Chen et al. showed that endovascular treatment combined with immunosuppressive therapy improved treatment results [8].









