Introduction
Since the first report of the association of absent nasal bone with Down syndrome by Cicero et al. [1] several studies have provided evidence supporting the association of absent or hypoplastic nasal bone with fetal aneuploidies [2–4]. The nasal bone is hypoplastic in about 50% to 60% of Down syndrome fetuses and 6% to 7% of euploid fetuses [5]. Using nasal bone hypoplasia as a marker increases both its sensitivity and false positivity. However, the detection rate depends upon the threshold used.
Although different assessment methods were developed to evaluate the nasal bone hypoplasia to increase specificity, sensitivity was reduced simultaneously. These evaluation methods include the ratio of biparietal diameter-nasal bone length (BPD/NB thresholds of > 9 to > 12 have been used to define abnormal) [6], ≤ 2.5 mm threshold for abnormal nasal bone length [7], a gestational age-based threshold (< 2.5th or 5th percentile) based on the distribution of nasal bone length in normal fetuses [8], and use of multiple of the median (MoM) of nasal bone length for gestational age (< 0.75 MoM) [9, 10].
Also, studies have shown that the normal ranges of the second trimester fetal nasal bone length vary significantly among different ethnic groups [11]. So, the use of nasal bone length percentiles in screening may be impractical for different ethnic groups. Yan Du et al. reported the hypoplastic nasal bone rate as 1.15% in the Chinese population by using 2.5% rates described in other studies [12]. As the evaluation of the fetal face may be difficult in the late second and third trimester, due to the position of the fetus, ultrasound assessment of nasal bone is more favorable in the early second trimester [13].
This study aimed to evaluate the efficiency of the nasal bone to nasal tip length (NB/NL) ratio as a practical marker for the prediction of Down syndrome in second trimester genetic sonography.
Material and methods
This double centered retrospective case-control study was conducted in the Department of Obstetrics and Gynecology, Maternal-Fetal Medicine Unit, School of Medicine, Izmir Katip Celebi University, Izmir, Turkey and in the Department of Obstetrics and Gynecology, Maternal-Fetal Medicine Unit, Sanliurfa Education and Research Hospital, Sanliurfa, Turkey. Both units are tertiary centers in Turkey which receive referral patients from their region. Ethical approval for this study was obtained by Harran University School of Medicine Ethical Board (registration number: 21/12/2018-10990). The study design was in accordance with the Helsinki Declaration [14], and conformed to the Committee on Publication Ethics Guidelines.
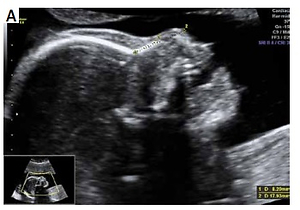
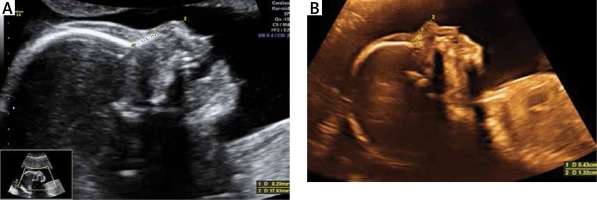
The collected data were provided from the patients’ recorded and stored 18-24 weeks anatomic survey ultrasonography files performed between January 2012 and November 2018. Files of patients who had a single euploid or trisomy 21 fetuses were selected. Multiple pregnancies were not included in the study. Files containing an adequate view for evaluating the nasal bone and nasal tip were selected for inclusion. Patients with absent fetal nasal bone were excluded from the study. Fetal nasal bone and nasal tip lengths were assessed with two-dimensional images of the fetal head in the sagittal plane enlarged to include the nose, as well as the lips, maxilla and mandibula, with an angle between the insonation beam and the nasal axis close to 45° or 135° to define the edges of the nasal bone and nasal tip more sharply. Nasal bone length was measured from the proximal edge of the bone to the end of the bone. Nasal tip length was measured from the proximal edge of the nasal bone to the distal part of the nasal tip (Figures 1 A, B). All sonographic evaluations were performed by two experienced physicians (Kelekci S and Ekmekci E). The ultrasound devices used were the Voluson E6 system (GE Healthcare, Milwaukee, WI) and the Sonoace X8 ultrasound machine (Medison, Korea) with RAB 4–8-MHz transabdominal probes. Other aneuploidies were excluded from the study. Fetal karyotypes were confirmed either by chromosomal analysis when available or by neonatal examination.
Figure 1
A – Nasal bone/nasal tip length ratio > 1/3 in euploid fetus, B – nasal bone/nasal tip length ratio ≤ 1/3 with Down syndrome fetus
The ratio of the nasal bone to the nasal tip length was calculated for all patients included in the study. The threshold ratio ≤ 1/3 was defined as nasal bone hypoplasia. Patients were divided into two groups as having euploid or trisomy 21 fetuses. The groups were compared for the presence of nasal bone hypoplasia according to the defined threshold for the nasal bone nasal tip ratio.
Statistical analysis
Statistical analysis was performed with the χ2 test for categorical variables and Student’s t-test for continuous variables. The primary outcome was to detect the efficacy of the NB/NL ratio for the prediction of trisomy 21. All analyses were performed using MedCalc Statistical Software version 17.9.7 (Med Calc Software, Ostend, Belgium).
Results
Among 9868 pregnant women who were evaluated over the 5-year period, an adequate record for sonographic evaluation of the NB/NL ratio was achieved in 2363 women. A total of 2363 records included 59 fetuses with trisomy 21 and 2304 fetuses with normal karyotype.
In the group of euploid fetuses, the 5th percentile for the NB/NL ratio was 0.37 while it was 0.27 in the trisomy 21 group. The median rate was 0.46 in euploid fetuses (0.26–0.64) and was 0.4 (0.26–0.61) in trisomy 21 fetuses. Mean gestational age at ultrasound assessment was 21.0 gestational weeks (range: 18–22 weeks) and the median maternal age was 33.0 years (range: 15–47 years).
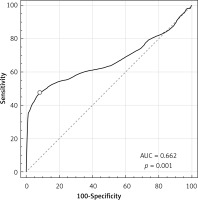
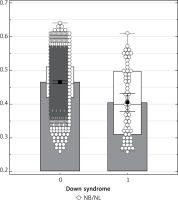
Patients were divided into two groups: 59 patients with trisomy 21 fetuses and 2304 with euploid fetuses were included in the study. Figure 2 is a box and whisker plot of the NB/NL ratio for each group and the regressed median rates in groups. The diagnostic performance of the NB/NL ratio for trisomy 21 is visualized in a receiver operating characteristics (ROC) curve (Figure 3). The NB/NL ratio was 1/3 and below in 35.5% (21/59) of fetuses with Down syndrome whereas this ratio was 1.3% (31/2304) in euploid fetuses. The difference was statistically significant (p < 0.001). Sensitivity, specificity, positive likelihood ratio and negative likelihood ratio of NB/NL ≤ 1/3 were 35.5%, 98.6%, 26.4 and 0.6 respectively for Down syndrome, with a false positive rate of 1.34% (Table I).
Figure 2
Box and whisker plot of NB/NL ratio in fetuses with and without Down syndrome
NB/NL – nasal bone nasal tip length ratio.
Discussion
This study showed that the NB/NL ratio could detect 35.5% of the fetuses with trisomy 21 with a higher specificity than other methods during the 18th and 22nd weeks of pregnancy. This practical method may be a good alternative to other difficult and time consuming methods for the evaluation of fetal nasal bone hypoplasia in the second trimester sonography.
Several studies have reported the absence of the nasal bone as the single most sensitive and specific second trimester marker for trisomy 21 in all populations [15]. It is therefore essential to evaluate the nasal bone hypoplasia during the second trimester.
Several methods have been described to define nasal bone hypoplasia with higher sensitivity and specificity. In a large review comparing the length of the nasal bone (percentiles or 2.5 or 3 mm cut-off value) in trisomy 21 and normal fetuses, according to the combined data, the nasal bone was short in 40 of 83 (48.2%) trisomy 21 fetuses and 136 of 5643 normal fetuses with a 2.4% false positive rate [8]. Odibo et al. reported 61% sensitivity by defining nasal bone hypoplasia if BPD/NB < 11 but with 84% specificity. Also they reported 49% sensitivity with 92% specificity by defining nasal bone hypoplasia according to nasal bone MOM levels < 0.75 MOM [10]. Has et al. found that a nasal bone length less than 5th percentile, < 0.72 MoM, or BPD : NBL > 12.16 can be used in the second trimester to define nasal bone hypoplasia as a means of predicting Down syndrome because their predictive values are similar at a fixed 5% false-positive rate [16].
In this study, sensitivity was 35.5% and specificity was 98.6% for the diagnosis of trisomy 21. Although the sensitivity of our method for defining nasal bone hypoplasia is not better than the other options, it is highly specific for the purpose compared to the other methods. None of the previous studies had a 1.34% false positive rate success with a similar sensitivity.
Prefumo et al. reported nasal bone length percentiles of 3992 fetuses with normal karyotype at 11–14 weeks of gestation in different ethnic populations and reported that distribution of fetal nasal bone length rates was associated with the ethnic origin of the mother [17]. Since normal limits for nasal bone length during the second trimester appear to vary according to race and ethnic background, a single fixed cutoff value cannot be used to predict trisomy 21 in different ethnic populations [11]. Thus, medical calculators are used in clinical practice to provide the nasal bone percentiles for different ethnic groups (available at http://perinatology.com/calculators/Nasal%20bone.htm). Although our study includes results from only one center and only the Turkish pregnant population, describing a ratio rather than using percentiles may be able to eliminate natural variations due to different ethnicities, in our opinion.
In this study we specified the NB/NL ratio threshold level ≤ 1/3 for defining hypoplasia. This threshold was chosen to achieve higher sensitivity and specificity. Sensitivity increases to 44% when the specificity is reduced to 95%, as evidenced by the ROC curve. The most important advantage of this method is its practicability in clinical use, although it does not have the advantage of previous studies in terms of diagnostic success. Our data demonstrate that the NB/NL ratio can be used to predict trisomy 21 in the second trimester as it is simple to measure, objective to interpret, and is therefore practical to use. We believe that the NB/NL ratio can be easily integrated in second trimester obstetric sonography.
To the best of our knowledge, this is the first study to evaluate the sensitivity and specificity of the NB/NL ratio within a relatively large population of fetuses with trisomy 21 and normal karyotype. The major strength of our study is that it evaluates only the NB/NL ratio in trisomy 21 and euploid fetuses regardless of other fetal anomalies and chromosomal aberrations. Its retrospective design is the major limitation of our study. The specificity of the obtained results to the Turkish population and pregnancies of 18–22 weeks is another limitation.
Certainly, with the introduction of noninvasive prenatal tests (NIPT), screening for aneuploidies is changing and sonographic markers are losing their significance. However, currently not all countries have an established first trimester screening program and the widespread introduction of expensive cell free fetal DNA takes time. Therefore, second trimester Down syndrome markers will remain invaluable for a while longer. Our study suggests that the evaluation of NB/NL ratio may be a new promising and practical sonographic marker for Down syndrome in the second trimester ultrasonography. Although its sensitivity is not high, it seems more specific and practical than the evaluation of fetal nasal bone length. Further prospective research is warranted to determine the predictive power of the NB/NL ratio in second trimester screening for trisomy 21, particularly in comparison to the other methods used for defining nasal bone hypoplasia.