Introduction
Over the last 20 years, minimally invasive mitral valve surgery (MIMVS) has established itself as an alternative to the conventional sternotomy approach and has increasingly been used in patients with mitral valve pathology. Rather than a single approach, MIMVS refers to a collection of techniques and operation-specific technologies such as modified perfusion and visualization techniques that are aimed at minimizing surgical access and trauma. Several studies have reported promising outcomes with MIMVS as compared to conventional surgery with less pain, shorter hospital stays, faster return to normal activities, potential cost savings and superior cosmesis [1–4]. Yet, because the extent and location of mini-thoracotomies employed vary from center to center, the conclusions regarding superior esthetics with minimally invasive surgery are not generalizable [5–7].
Recently, some totally thoracoscopic techniques have been introduced, with excellent results [8–10]. The totally thoracoscopic periareolar (TTP) access employs an incision through the natural scarring in the body around the right nipple. This new approach seems to be comparable or even superior to the minithoracotomy access with regards to preferable esthetic results [11].
Aim
The aim of the current paper was to report early surgical data as well as mid-term outcomes in patients operated on because of MV disease with totally thoracoscopic periareolar access.
Material and methods
Study population
The study involved 48 consecutive patients undergoing heart surgery between 2015 and 2017 due to mitral valve disease. Data were collected prospectively in a single-center- and all-participant registry. After completion, these were retrospectively analyzed. A primary anatomical exclusion criterion for the TTP approach was the female gender, since this technique required an incision around the right nipple and the preparation of the tissue around the mammary gland; in fact, that was revised shortly after and women with a small mammary gland were also considered for the TTP approach. Patients with endocarditis and re-do procedures were excluded.
Periareolar approach. Surgical technique
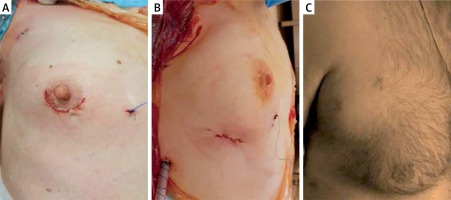
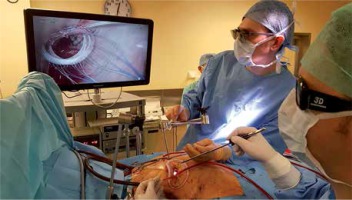
A standard peripheral cannulation for extracorporeal circulation (ECC) in MIMVS was advocated. Cannulation of the femoral vessels was performed through surgical access (via a 3–4 cm groin incision) using Seldinger’s technique. For venous cannulation, a 22–25 Fr and 60 cm long, perforated soft cannula (VFEM Femoral Venous Cannula, Edwards Lifesciences, USA or RAP cannula, LivaNova, United Kingdom) was used. The positioning of the venous cannula with its tip in the superior vena cava was achieved under transesophageal echocardiography (TEE) guidance. In patients scheduled for elective tricuspid valve intervention, an additional venous cannula (EOPA Medtronic, USA or OptiSite, Edwards Lifesciences, USA) was inserted into the internal jugular vein. The femoral artery was cannulated with a 16–18 Fr OptiSite (Edwards Lifesciences, USA) cannula. Choice of the cannulas was left to the surgeons’ preference. A right lateral chest wall incision of 3.5–4.0 cm around the bottom of the nipple was performed (Photos 1 A–C); two additional 1.0 cm incisions were made for the thoracoscope and the aortic clamp in the 2nd and 3rd intercostal spaces. The chest cavity was inflated with carbon dioxide at a flow rate of 1.5–2 l/min. In all patients, a Chitwood clamp was used for cross clamping of the aorta. The heart was arrested with either Bretschneider or 4 : 1 cold blood cardioplegia infused into the aortic root under direct vision. Mitral annuloplasty was performed using semirigid mitral rings (Carpentier-Edwards Physio or Physio II Annuloplasty Ring, Edwards Lifesciences, USA or Memo 3D LivaNova, United Kingdom) (Photo 2). In patients with mitral valve leaflet prolapse, a correction was performed using the polytetrafluoroethylene (PTFE) loop technique. In the case of concomitant tricuspid valve surgery, an annuloplasty was performed on the arrested heart with a dedicated ring (MC3 Tricuspid Annuloplasty Ring, Edwards Lifesciences, USA). The acute result of the mitral and tricuspid valves’ repair was confirmed in TEE. Remnant regurgitation less than or equal to 1+ was accepted.
Statistical analysis
A statistical analysis was performed using the Statistica program (StatSoft, Inc. 2012). The Shapiro-Wilk W-test was used to test for normality. When the W statistical value proved significant, the hypothesis that the corresponding distribution is normal was rejected. Normally distributed continuous variables were expressed as a mean ± standard deviation or median and interquartile range (IQR). Nonparametric and parametric data correlations were evaluated using either the Spearman rank test or the Pearson test. Logistic regression was fitted to identify the mortality and reoperation risk factors. The Kaplan-Meier curves were used for presentation of overall survival and freedom from reoperation.
Results
Preoperative characteristics
During the study period 2015–2017, 98 minimally invasive mitral/tricuspid procedures were performed; of those 48 were conducted via the TTP approach. Study subjects were predominantly male (83.3%); p < 0.001 aged 65.4 ±10 years. The principal indications were MV regurgitation in 44 (91.7%) and MV stenosis in 4 (8.3%); additional TV regurgitation was present in 11 (22.9%). Mean EuroSCORE II was 5.1 ±4.0 and ranged from 0.2 to 14.4, primary MV regurgitation was diagnosed in 38 (86.4%), secondary MV regurgitation in 6 (13.6%) patients. Predominant posterior leaflet prolapse was the most frequent regurgitation cause 31/48 (81.6%), followed by anterior leaflet prolapse. Detailed characteristics are presented in Table I.
Table I
Preoperative characteristics and indications for surgery
Surgical characteristics
Mitral valve repair was the preferred approach in 44 (91.7%) cases, followed by MV replacement in 4 (8.3%); the PTFE loops and annuloplasty rings were used in all MV repair cases. Replaced valves were bioprostheses in all 4 cases. No procedural complications occurred. A conversion to full sternotomy was not required in any patient. No conversion from a totally thoracoscopic approach to the video-assisted minithoracotomy approach was needed either. Mean aortic cross-clamp time was 103 ±39 min while total ECC time was 166 ±53 min (Table II); average blood loss was 445 ±308 ml with a median (IQR) number of transfused blood units per patient of 1.0 (0–2.0). Two patients required chest revision due to increased postoperative bleeding. Mechanical circulatory support (i.e. by means of extracorporeal membrane oxygenation, an intra-aortic balloon pump or a left ventricular assist device) was not necessary in any patient. The average postoperative mechanical ventilation time was 7.6 ±8.0 h; extended mechanical ventilation (over 24 h) was needed in 1 (2.1%) patient. Length of ICU stay was 1.2 (1.0–2.0) days. Wound infection requiring surgical intervention did not occur in any patients operated on using the totally endoscopic technique (Table II). One patient died on post-op day 14, due to multiorgan failure; he underwent an MV and TV combined procedure and his EuroSCORE II was > 14. No strokes occurred.
Table II
Clinical outcomes
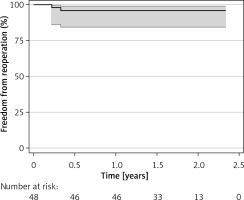
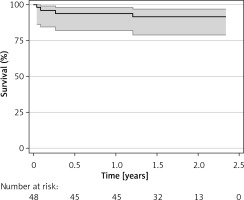
Freedom from reoperation was estimated at 96.4% within the investigated follow-up (Figure 1). The overall estimated survival was 96.9% (Figure 2). Risk factors for mortality and reoperation are presented in Table III. Among them, length of ICU stay (p = 0.003) and number of transfusions (p = 0.05) were associated with increased mortality. Surgical access, operation type, baseline patients’ and procedural characteristics were not associated with either risk of mortality or reoperation.
Table III
Mortality and reoperation risk factors
Discussion
In the last two decades, mitral valve surgery has significantly evolved [1–9]. Key progress has been made due to the introduction of the combination of minimally invasive techniques and standardized valve repair techniques [12–16]. The benefits of minimally invasive surgery in the mitral valve setting have been summarized in the consensus statement of the International Society of Minimally Invasive Cardiothoracic Surgery (ISMICS) [17]; these include the reduction of blood loss, the reduced need of transfusion, a lower risk of postoperative atrial fibrillation and a shortened length of hospital stay. The downsides observed were longer aortic cross-clamp and ECC time, which however were shown not to have any significant effect on postoperative complications. Moreover, several reports showed improved results of the minimally invasive approach in comparison to the standard sternotomy access in patients with increased operative risk, such as chronic kidney disease or undergoing redo surgery [18, 19]. Numerous studies from leading cardiac surgery clinics have shown the effectiveness and durability of mitral repair performed using minimally invasive techniques [20], suggesting however volume effect. Regardless, a minimally invasive program for mitral valve surgery is being successfully introduced in a growing number of hospitals [21], even despite an evident necessity of additional training and financial resources.
The most widely performed minimally invasive setting is the video-assisted approach through right minithoracotomy and femoral vessels’ cannulation [22]. The periareolar totally thoracoscopic access gained some popularity recently as potentially being an even less invasive method [11]. The current pilot study of the TTP approach for minimally invasive surgery has shown promising results as well. Our findings therefore endorse the endoscopic approach as the next step in the evolution of minimally invasive mitral valve surgery.
Several issues, however, need to be addressed concerning our results. Firstly, the totally endoscopic technique has an anatomic limitation; we found that this approach can safely be used in male patients; however, in our opinion it should carefully be considered in females since it requires mammary gland preparation. Exceptions may be made for selected females with small mammary glands. Moreover, in males, the position of the thoracotomy incision was determined by the position of the nipple and was typically located more anteriorly and, at times, also superiorly when compared to video-assisted access. Consequently, safe introduction of the totally endoscopic mitral valve surgery program requires of the surgeon to have a combined experience in ‘direct vision’ minimally invasive valve surgery, mitral valve repair and thoracoscopic procedures. It should be stressed that the main technical challenge for the surgeon while learning the endoscopic technique is the change from having direct vision to total video surgery in crucial steps of the procedure.
Several previous studies reported increased risk of cerebral complications during retrograde arterial perfusion in minimally invasive mitral valve procedures [23]; others pointed to complications during aortic occlusion [24]; however, the use of cerebral oximetry guided extracorporeal circulation along with TEE as a routine practice in our clinic resulted in a significant reduction of stroke risk in the abovementioned circumstances. The proposed strategy is based on zero tolerance for significant changes in brain oxygenation during the cardiopulmonary bypass and the adjustment of immediate perfusion parameters and cannulas’ position. Cerebral desaturation during operations is a proven predictor of cognitive decline and should, therefore, be avoided at all costs [25]. In our series, no in-hospital stroke occurred.
Worth mentioning is the standardized perfusion protocol of moderate hypothermia (between 32°C and 34°C) and oxygen delivery (goal directed perfusion protocol) [26, 27]. Another practice is defining patients at high risk of stroke in preoperative CT scans, which could help in peripheral cannulation planning and aorta clamping (Endoclamp vs transthoracic clamp) [24, 28, 29].
The future place of the totally endoscopic approach in cardiac surgery needs to be discussed with respect to robotic techniques [30]. It may seem that the totally thoracoscopic technique presents several advantages over robotic surgery. Firstly, the number of surgical incisions is vital in both thoracoscopic and the robotic approaches. In our technique, the following surgical cuts are performed: access to femoral vessels (this can be performed percutaneously), one working port, one camera port and one port for the aortic clamp placement (if needed). According to the latest studies from Musumeci et al. [12], the periareolar robotic approach results in a similar total [mm] length of the incision. Secondly, when compared to the robotic access, the thoracoscopic approach was associated with shorter aortic cross clamp time, lower costs and a shorter learning curve for the surgeon. Last but not least, the introduction of the totally endoscopic mitral valve surgery might be easier than that of the robotic surgery program even for experienced surgeons. The economic aspect is also not to be overlooked [31].
The use of stereoscopic vision systems is a clear advantage of robotic surgery; however, the latest 3D thoracoscopic systems in clinical observations have already greatly facilitated the totally thoracoscopic surgery. In our experience, the current 3D optics provide the surgeon with a better sense of the working space, especially with regard to the subvalvular apparatus. The 3D optics facilitate the performance of complex and precise movements, which necessitate both eyeballing and actual distance assessment, especially when it comes to examining the structures positioned parallel to the surgeon’s axis of vision, e.g. measuring the proper length of PTFE loops or chordae. Using 3D optics may also, in fact, provide better comfort of reoperation through minimally invasive access. The downside of the use of 3D optics in cardiac surgery is the inability to change the camera’s viewing angle without the surgeon making a movement of the entire 3D camera arm, while other camera devices require only the touching of a small switch to change the viewing angle.
Several limitations to the current study need to be acknowledged. Firstly, the study was designed as an analysis of results from a single center and is thus based on the case study of a limited group of patients operated on using the new technique, and thus includes the team’s learning curve, which may not be a factor present in other circumstances. Secondly, the number of patients included in the totally endoscopic approach group is limited and has a gender preference due to the very nature of the incision necessary in this approach.