Introduction
Heart failure (HF) is a clinical syndrome associated with physical capacity impairment manifested by fatigue, shortness of breath and dyspnea, which affects the everyday quality of life (QoL) [1–4]. Among patients with cardiovascular diseases patients with HF experience the lowest QoL in all (physical, emotional, and social) areas [5]. Available data have shown that QoL might predict prognosis [6]. Moreover, poor QoL scores are associated with higher mortality in HF patients [6]. For this very reason multidisciplinary interventions enhancing health status and QoL might be an important aspect of care in HF treatment [1, 2, 7, 8]. Therefore, the European Society of Cardiology (ESC), American Heart Association (AHA), and American College of Cardiology (ACC) guidelines strongly recommend holistic management for HF patients which encompasses the appropriate treatment, cardiac rehabilitation and scheduled follow-up visits [1, 2, 7, 8].
Regular exercise training improves functional capacity, autonomic nervous system function, peripheral hemodynamics, QoL and prognosis in HF patients [7–9]. Nevertheless, there is a risk that those advantages might decrease after patients are discharged home and return to their previous health behaviors and self-management habits [3, 5, 6]. The low level of adherence to treatment and recommendations influences QoL in HF patients, making this area an important target for the holistic approach in HF management [10]. According to current knowledge, patients with psychological difficulties tend to engage less in physical activity, which, in turn, could lower their physical capacity and, as an effect, health-related QoL [11]. Moreover, people experiencing both somatic and mental illness manifest the lowest QoL, in comparison to healthy subjects or those with only somatic or mental illness [12, 13]. Therefore, psychological interventions aimed at emotional difficulties and adjustment to treatment could be beneficial not only for the patients’ psychological well-being but also for the engagement in self-care behaviors, such as physical activity.
Another issue is the unsatisfactory participation of HF patients in cardiac rehabilitation programs [14]. It results from the existence of many potential factors hindering the implementation of rehabilitation in everyday clinical practice [15]. The most common are patients’ condition related factors such as the level of disability, the impact of comorbidities, cognitive problems, the lack of motivation as well as logistic problems resulting from the need to organize transport and to be assisted by relatives or partners [14, 15]. One possible solution to overcome some of these problems is the implementation of novel telemedical technology (such as home-based, remote supervised cardiac rehabilitation), which is a valuable alternative to a standard form of rehabilitation for HF patients [16–21].
The novel hybrid comprehensive telerehabilitation (HCTR) consists of telecare including remote psychological support, telerehabilitation and remote monitoring of cardiovascular implantable electronic devices (CIEDs) and might be a valid solution aimed at improving QoL and patients’ emotional condition [22, 23]. The assessment of the impact of the new therapeutic procedure on QoL is an important indicator of its effectiveness in terms of daily functioning of HF patients. In fact, there is some evidence regarding QoL improvement of cardiac patients who were referred for home-based cardiac telerehabilitation but the results of studies including HF patients are still scarce and inconsistent [15, 24–27]. Little is known about the impact of HCTR on QoL changes in the physical and mental domain as well as in detailed specific areas of QoL. Therefore, we aimed to investigate the influence of an HCTR program on various facets of QoL in HF patients in comparison to usual care (UC) alone based on data from the Telerehabilitation in Heart Failure Patients TELEREH-HF study [22, 23].
Material and methods
Patient population
The present study formed part of a randomized, prospective, multicenter, open-label, parallel group-controlled TELEREH-HF trial comparing HCTR and UC in HF patients (Clinical Trials.gov NCT 02523560). The study was approved by the local ethics committee (IK-NP-0021-85/1402/13). Each patient provided written informed consent.
The design and main results of the TELEREH-HF study have been published previously [22, 23]. In brief, 850 HF patients with New York Heart Association (NYHA) class I, II, or III and left ventricular ejection fraction (LVEF) of 40% or less were enrolled in the TELEREH-HF trial between June 8, 2015, and June 28, 2017, in 5 centers in Poland. The inclusion and exclusion criteria are presented in Table I [22, 23]. Eligible patients were randomized 1 : 1 to HCTR plus UC or UC only and followed up for 14 to 26 months after randomization. During the first 9 weeks, patients underwent either an HCTR program (1 week in the hospital [initial stage] and 8 weeks at home; exercise training 5 times per week) or UC with observation. The goals of the initial stage were: a baseline clinical examination, optimization of treatment, education, planning of exercise training and performing five monitored educational training sessions [22, 23]. The home-based HCTR consisted of two parts: consent to access each training and the training session. Details are shown in Table II [22, 23].
Table I
[i] ESC – European Society of Cardiology, LVEF – left ventricular ejection fraction, NYHA – New York Heart Association, CRT-P – cardiac resynchronization therapy, CRT-D – cardiac resynchronization therapy and implantable cardioverter-defibrillator, ICD – imp lan- table cardioverter-defibrillator, PM – pacemaker
Table II
| Telerehabilitation set |
| The EHO mini device was able to record ECG and transmit them via a mobile phone network to the monitoring center. An EHO mini device has training sessions preprogrammed for each patient (defined exercise duration, breaks, timing of ECG recording). The moments of automatic ECG registration were preset and coordinated with the exercise training. The planned training sessions were executed with the device indicating what needed to be done with sound (bleeps) and light signals (colors emitting diodes). The timing of automatic ECG recordings corresponded to peak exercise [22, 23] |
| Telesupervised exercise training |
| Before beginning a training session, patients answer a series of questions regarding their present condition: fatigue, dyspnea, blood pressure, body mass, and medication taken. Patients then transmitted resting ECG data to the monitoring center. Before giving permission to start the training session, the medical staff also analyzed data sent from the remote monitoring of CIEDs. If no contraindications were identified, patients were given permission to start the training session (the consent procedure) [17, 18]. The system was used to monitor and control the training in any place where the patient elected to exercise. If the training session was completed uneventfully, the patient would transmit the ECG recording to the monitoring center immediately after the end of every training session. The ECGs were analyzed at the monitoring center, and the safety, efficacy, and accuracy of a tailored patient’s rehabilitation program were assessed. Telephone contact was also used for psychological support [22, 23] |
| Education |
| Patients were taught how to self-evaluate, how to measure HR, blood pressure, body mass, how to performed exercise training, how to evaluate the level of perceived exertion according to the Borg scale and how to operate a TR set. Education also encompassed smoking cessation, lipid management, nutritional counselling, vocational and psychosocial support [22, 23] |
The HCTR intervention encompassed telecare, tailored telerehabilitation and remote monitoring of CIEDs. HCTR was conducted by a medical team (physicians, physiotherapists, nurses, and psychologist). The telemonitoring system was composed of a remote device for tele-ECG monitoring and supervised exercise training (the telerehabilitation set), mobile phone, and a monitoring center. The telerehabilitation set included: a special remote ECG transmission device with a personalized training session (EHO-MINI device, Pro-Plus Company), blood pressure meter and weighing scale [22, 23]. Exercise training was planned in line with the guidelines [9]. The HCTR consisted of 3 training modalities: endurance aerobic Nordic walking training, respiratory muscle training, and light resistance and strength exercises. The details are presented in Table III [22, 23].
Table III
Assessment of health-related QoL
During both parts of the assessment (initial and 9-week follow-up), all patients were asked to fill in the Medical Outcome Survey Short Form 36 Questionnaire (SF-36) regarding their health-related QoL and Beck Depression Inventory II (BDI) [23, 28]. The SF-36 is a worldwide known tool for evaluating overall health-related QoL, which consists of two major domains (physical and mental QoL) and various subscales, such as physical functioning, mental health, bodily pain, general assessment of one’s health and reported health transition, vitality, social functioning and functioning in social roles related to physical and emotional wellbeing [28]. It was proven to help recognize and understand the facets of QoL in many populations of patients.
Initial psychological assessment and consultation, along with clinical evaluation, allowed identification of patients who could benefit from psychological intervention. All patients were assigned to the usual psychological care, which consisted of feedback after a clinical interview and questionnaire assessment and 1–2 sessions of psychological counseling during 3–5 days of the initial hospitalization regarding adjusting to HF treatment and current issues important for the patient. During the sessions, a psychologist discussed with the patients their issues related to the disease, its course, treatment, and rehabilitation approval. Additionally, patients from the HCTR group were offered psychological support via mobile phone during a 9-week telerehabilitation program. The selection of patients referred to phone counseling was based on clinical evaluation and the level of psychopathology assessed with psychological questionnaires (SF-36, BDI > 13 points). The number of phone calls and its length were discussed with the patient during initial psychological assessment and qualification for psychological counseling sessions
Patients from the UC group received standard care appropriate for each center. Some of them could participate in a rehabilitation program which (apart from exercise training) encompassed psychosocial assessment focused on cardiovascular risk factors, psychological support, and counselling regarding adjustment to treatment and enhancing health-related QoL.
Psychological teleintervention during HCTR
The psychological telecare intervention (telepsychology) took the form of supportive one-to-one psychological counseling sessions via mobile phone according to the schedule established with patients. Phone support was aimed at adjustment to the treatment, engagement in telerehabilitation and other important psychological issues which the patient wanted to discuss with the psychologist (e.g. anxiety management, strategies to cope with depression).
Patients in the HCTR group were equipped with a mobile phone, which they used to contact the telerehabilitation team, and psychologists made phone calls according to patients’ training schedule. Telepsychology sessions were performed by an experienced psychologist or a clinical psychologist in each of the five medical centers involved in the study. Psychological interventions were aimed at following patients’ experience with HCTR training, enhancing patients’ motivation to perform exercise and coping with emotional problems that were reported by patients during the first assessment or occurred during the telerehabilitation process. Interventions were based on a cognitive-behavioral paradigm, and used motivational interviewing strategies, as they have been proven to be an effective form of support for HF patients [29].
Statistical analysis
The sample size was calculated assuming 1 : 1 treatment allocation ratio, and an overall two-sided level of significance of 0.05. Assuming a difference of 21 days alive and out of hospital between study arms, with a standard deviation of 100, a sample size of 400 evaluable subjects per study arm (a total of 800) would be required to achieve 80% power. Accounting for a 5–6% loss to follow-up, the total number was increased to 850.
Descriptive statistic for quantitative variables are expressed as mean ± SD and for qualitative variables as frequencies and percentage. Characteristics of patients who underwent HCTR vs. UC were compared using the χ2 test of independence or Fisher’s exact test for categorical variables and the two independent samples t-test for continuous variables. Ordinal variables were compared using Cochran Mantel-Haenszel modified ridit scores (row mean scores statistic). Continuous tertiary outcomes describing QoL were compared using the two independent samples t-test (analysis between groups) and paired t-test (analysis within groups). The p-value of 0.05 was the threshold used in determining the level of significance. All statistical tests were two-sided.
Subgroup analyses focused on the difference between study arms in QoL measures by heart failure etiology, age and depression. Covariance analysis was used, adjusting for the baseline level of the measurement. Heterogeneity of treatment effect was assessed using an interaction term between subgroup and treatment arm. All analyses were performed using SAS software version 9.4 (SAS Institute Inc., Cary, NC, USA).
Results
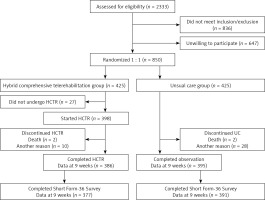
A total of 2333 patients were screened, and 836 patients did not meet inclusion/exclusion criteria and 647 refused to participate. Of the 850 patients randomized, 425 were assigned to HCTR and 425 to UC. No patient withdrew consent from the study or was lost during the 9-week intervention (HCTR group)/observation (UC group) period. Twenty-seven patients did not undergo telerehabilitation due to: technical difficulties with operating the telerehabilitation set (21), new onset of comorbidities (4), return to work (2) [23]. Eleven patients from the HCTR group and 4 patients from UC group did not agree to fill out the SF-36 questionnaire during the 9-week follow-up. Eventually, 377 patients from HCTR and 391 from the UC group were analyzed (Figure 1). In the UC arm 51 (12%) patients participated in cardiac rehabilitation programs.
Figure 1
Flow of patients through the TELEREH-HF study – subanalysis of QoL
HCTR – hybrid comprehensive telerehabilitation, UC – usual care.
The baseline clinical characteristics of both groups are presented in Table IV.
Table IV
Baseline characteristics
[i] HCTR – hybrid comprehensive telerehabilitation, UC – usual care. SD – standard deviation, BDI – Beck Depression Inventory, NYHA – New York Heart Association class, ACEI – angiotensin converting enzyme inhibitors, ARB – angiotensin receptor blockers, CIEDs – cardiovascular implantable electronic devices, NOAC – non-vitamin K antagonist oral anticoagulants, CRT-P – cardiac resynchronization therapy, CRT-D – cardiac resynchronization therapy and cardioverter-defibrillator.
During the initial psychological evaluation and clinical assessment, 153 patients indicated that they required psychological support – 73 patients from the HCTR group (19.4%) and 80 patients from the UC group (20.5%) (p = 0.704). Among patients referred for psychological intervention, 45 patients from the HCTR group (61.6%) and 37 patients from the UC group (46.2%) agreed to follow the recommendations within the telerehabilitation program or outside the clinic (p = 0.056). Of patients from the HCTR group who consented to psychological care during the initial evaluation, 34 (75.5%) participated in psychological tele-support via mobile phone during the 9-week HCTR program. Eleven patients withdrew from a telesupportive psychological session during the program or could not participate due to technical difficulties or severe cognitive impairment. Between 1 and 9 teleconsultations per patient during home telerehabilitation were performed and they lasted for 14.03 min on average (standard deviation = 5.58). Most of the patients preferred to receive psychological tele-support at least once every 2 weeks; therefore 24 participants (70.59% of patients who received phone calls) underwent 5 to 9 teleconsultations during the 9-week program and 10 patients preferred less frequent teleconsultations (1 to 4 phone calls from the psychologist during the telerehabilitation program).
At the baseline assessment, the two groups did not differ significantly in any of the evaluated (medical and psychological) variables (Tables IV and V).
Table V
Comparison of QoL outcomes
[i] HCTR – hybrid comprehensive telerehabilitation, UC – usual care, SD – standard deviation, p-value1 level of significance (comparison of QoL in HCTR group before rehabilitation and after its completion – within HCTR group analysis), p-value2 level of significance (comparison of QoL in UC group before and after observation – within UC group analysis), p-value3 level of significance (comparison of QoL in groups studied before intervention/observation – between groups analysis), p-value4 level of significance (comparison of QoL changes [Δ] – between groups analysis).
After intervention, the HCTR group in comparison to baseline values showed significant improvement in overall QoL (Δ = 1.87 ±9.95, p = 0.003), physical domain of QoL (Δ = 1.25 ±5.7, p < 0.0001), and 4 areas of QoL (physical functioning: Δ = 1.08 ±5.9, p = 0.0005; role functioning related to physical state: Δ = 1.50 ±7.60, p = 0.0002; general health: Δ = 0.86 ±6.78, p = 0.014; and vitality: Δ= 1.51 ±7.49, p = 0.0001).
A significant positive change in QoL in the UC group was observed in vitality (Δ = 0.83 ±6.98, p = 0.019) and social functioning (Δ = 0.94 ±9.3, p = 0.48) in comparison to baseline data.
QoL in the UC group decreased in the physical domain and in four specific areas (physical functioning, role functioning related to physical factors, bodily pain and role functioning related to emotional factors), but these results were not statistically significant. Changes in the level of QoL in both compared groups are shown in Figure 2.
Figure 2
Change in the level of various facets of quality of life in HCTR and UC groups
HCTR – hybrid comprehensive telerehabilitation, UC – usual care, PF – Physical Function, RP – Role- Physical, BP – Bodily Pain, GH – General Health, VT – Vitality, SF – Social Functioning, RE – Role- Emotional, PC – Physical Component Score, MC – Mental Component Score, SF – Short Form.
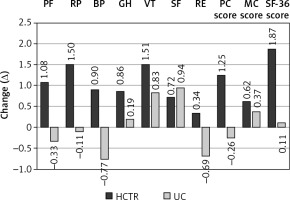
There were significant differences in QoL after a 9-week intervention/observation between two groups. Results of the study imply a greater improvement in HCTR in comparison to UC for overall QoL (p = 0.009), physical domain of QoL (p = 0.0003), and three specific areas of QoL (physical functioning, p = 0.001; role functioning related to physical state, p = 0.003; and bodily pain, p = 0.015). All obtained results are shown in Table V.
Subgroup by treatment interactions are presented in Tables VI–VIII. There were no interactions between health failure etiology or age category (at or above versus below 65) and treatment effects for any of the QoL measures (Tables VI and VII). We observed statistically significant interactions between depression status and treatment group for the following QoL parameters (Table VIII): vitality (p = 0.017), role-emotional (p = 0.008), mental component score (p < 0.001) and overall score of SF-36 (p = 0.025). In the UC group we observed a deterioration in QoL in patients with depression.
Table VI
Interaction of QoL in terms of heart failure etiology and treatment effects adjusted for baseline
Table VII
Interaction of QoL in terms of age and treatment effects adjusted for baseline
Table VIII
Interaction of QoL in terms of depression and treatment effects adjusted for baseline
Discussion
The main and novel finding of the TELEREH-HF trial subanalysis is that in comparison to UC, a 9-week HCTR program led to a significant improvement of overall QoL, physical domain of QoL and three specific areas of QoL: physical functioning, role functioning related to physical state and bodily pain.
These findings confirm the results of two single-center studies, which showed that telerehabilitation provided significant improvement in the overall QoL index [24, 25]. It is worth noting that in these studies, the results were limited only to overall QoL and the procedure was not as comprehensive as in the TELEREH-HF study. Our intervention combined telecare, structure telephone support, psychological telesupport, telemonitoring of electrocardiography (ECG), blood pressure and weight, remote monitoring of CIEDs and tele-supervised exercise training to create the HCTR program [22, 23].
The results of only one single-center trial have been published and assessed in detail the influence of telerehabilitation on QoL in all specific areas [26]. Similarly to our study, an improvement was observed in the area of physical function and physical domain after telerehabilitation. However, in contrast to our results, there was also an improvement in mental health and the mental domain of QoL. Such divergent data may be the result of the differences in the type of psychological support, age and gender of participants, different training modalities (type, intensity, duration) and patients’ adherence to and acceptance of indications.
Other authors reported that QoL improvement achieved during the outpatient program was maintained during the next 12 months only in telemonitored, adherent HF patients [27]. The results of our study allow us to draw some interesting conclusions. First of all, both assessed groups showed improvement in different areas of QoL after a 9-week intervention (HCTR group) or observation (UC group) depending on the study arm. Additionally, in both groups in comparison to baseline values, there were no significant differences in the mental domain of QoL. It is possible that it derives from a higher level of QoL in emotional and social aspects of functioning in the baseline assessment, even with the presence of physical limitations. There were also significant differences in between-group analysis. The HCTR group showed significant improvement compared to UC in overall QoL and physical domain as well as subscales related to physical functioning, bodily pain, and role functioning related to the physical state. It suggests that tele-management makes it possible to enhance physical functioning and wellbeing of HF patients. Although patients from the UC group improved significantly in vitality and social functioning, the HCTR group showed enhanced performance in more aspects of health-related QoL. This suggests that telecare for HF patients enables their physical functioning to be improved after discharge and supports their social functioning. Moreover, patients in the UC group showed a slight decline in various areas of QoL, e.g. physical domain of QoL (Figure 2). Even though it was a nonsignificant decrease, it could suggest that UC without further self-management support or rehabilitation is not sufficient to sustain patients’ QoL. The chronic and progressive character of HF could influence those results. Nevertheless, the same intervention time in the HCTR group led to an improvement in many areas of QoL, which allows them to be interpreted as a result of the comprehensive telemanagement including telesupervised exercise training. Therefore, it is possible to assume that HF patients can benefit from telerehabilitation in terms of QoL and slowing the natural course of HF. It is also worth mentioning that HCTR patients with depression referred for psychological counseling noted improvement in overall QoL and its mental subscales, whereas patients with depressive symptoms in the UC group experienced a decline in QoL in a 9-week observation. It suggests that this particular group of patients could benefit from psychological telecare in areas of their emotional wellbeing as well as in those related to HF burden.
Another important issue worth pointing out is that the cultural and religious factors could significantly interfere with the level of QoL. Because our study population was homogeneous in terms of religion and culture, we do not anticipate a significant impact of those factors on our results.
HCTR is a multicomponent care program, which allows incorporation of work experience and support from various team members. Interventions based on the human factor are known to significantly reduce the risk of rehospitalization. Psychological support based on cognitive behavioral therapy principles is effective in decreasing the burden of depressed mood in HF patients. The results from this study also support the results of previous papers that suggested that education in terms of coping skills can enhance QoL in HF patients along with the improvement in functional capacity [30]. Noteworthy is the fact that not every patient referred for psychological care during initial evaluation agreed to follow those recommendations. The main reasons for the absence of participating in psychological tele-counseling were technical problems or severe cognitive impairment, which made participation in the telerehabilitation process impossible. Another factor was patients’ preference for face-to-face counseling. The level of non-adherence with this recommendation is comparable with other studies and more profound than adherence to telemonitoring or an exercise program [31, 32]. The main reasons for the lack of concordance in this area could stem from objective obstacles, such as cost or availability of psychological services in the area or with more subjective reasons such as the lack of motivation, and fear of being stigmatized as a person with mental problems. All those hindrances are present in various studies regarding the use of psychological services in the general population or among patients treated for somatic illness [32].
Tailored cardiac rehabilitation leads to the improvement of QoL in most HF patients. It enhances their mental condition and makes them more self-reliant and independent in their daily activities. The emphasis on psychological care during telerehabilitation will make it possible to follow patients’ coping with illness and treatment. It would provide an opportunity to enhance their engagement in treatment and QoL through developing new coping strategies. The results of this study imply that the majority of patients referred for psychological telecare accept this form of cooperation. It means that it could be beneficial for patients who cannot participate in a face-to-face intervention, due to their somatic state or logistic reasons (e.g. living far from the hospital). The second practical issue is related to the efficacy of psychological support via phone. The results of this study showed that this intervention increased QoL, which means it could be useful in the treatment of HF patients as a supportive follow-up strategy, which enables patients to receive counseling regarding important areas of self-care and addressing their emotional needs. This, on the other hand, creates an impression that the disease or disability tends to affect patients less severely than before. HCTR group patients not only received assistance from the telerehabilitation staff (physician, nurse, and physiotherapist) but also benefited from the exercise training and psychological support. In this context, it is not clear whether the improvement in QoL was causally related to the exercise modalities or resulted in part from the psychological support of each intervention. Probably, both factors contributed to QoL improvement.
One of the most important aspects of this study is the conclusion that psychological telecare is an intervention accepted by most HF patients and it enables their health-related QoL to be enhanced. It is a valuable alternative to face-to-face counselling and worth considering by the medical teams, especially for patients who have difficulties in receiving standard psychological support during HF treatment.
This study has some limitations. Firstly, QoL was assessed on the basis of only one questionnaire, which may have had an impact on the results. Secondly, the analysis presented is limited to a 9-week intervention period. It does not allow us to make long-term conclusions about the impact of HCTR on HF course. Furthermore, not every patient in the HCTR group was able to continue with psychological care via phone. Also, what needs to be stressed is that most of the TELEREH-HF population was male (89%), and for this reason the results cannot be fully extrapolated to the female population. We did not gather data on the socioeconomic status of patients, which might have influenced the results.
In conclusion, this detailed analysis of the QoL in the population of the TELEREH-HF study demonstrated that in comparison to usual care, hybrid comprehensive telerehabilitation resulted in a significant improvement in overall QoL, physical domain of QoL and three specific areas of QoL: physical functioning, role functioning related to physical state and bodily pain.