Introduction
Biliary diseases are common and frequently encountered in abdominal surgery. With the development of endoscopic [1], laparoscopic and robot technology, surgeons have more and more standardized principles and appropriate procedures for the diagnosis and treatment of biliary stones, biliary tumors and biliary malformations. With the expansion of the number of operations and the diversification of surgical methods, complications and iatrogenic biliary tract injuries also increase. Biliary stricture is one of the common complications after biliary surgery. According to previous literature, biliary stricture can occur in 0.1–0.5% of open biliary procedures and 0.25–1.0% of laparoscopic biliary procedures [2]. The probability of biliary injury and biliary stricture after laparoscopic cholecystectomy can be as high as 0.2–1.1% [3, 4].
Biliary stricture can be divided into benign stricture and malignant stricture according to its origin. Pancreatic ductal adenocarcinoma is the most common cause of distal malignant stricture, followed by cholangiocarcinoma, and less common ampullary cancer or metastatic cancer. Proximal malignant stricture is caused by cholangiocarcinoma, hepatocellular carcinoma, gallbladder carcinoma or lymphoproliferative diseases and metastatic lesions. The most common causes of benign stenosis include iatrogenic injury, chronic pancreatitis, primary sclerosing cholangitis, autoimmune diseases, etc. [5]. Although biliary stricture has a wide range of sources and diverse causes, its common characteristic is the manifestation of bile duct thinning caused by inflammation, scar or mass occupation, accompanied by upstream bile duct dilatation or not.
Because of bile duct obstruction, patients often suffer obstructive jaundice [6]. If bile duct inflammation is combined, they can also have symptoms of right upper abdominal pain, chills and high fever. In severe cases, the patients can also present with Reynolds’ sign. Right upper abdominal tenderness radiating to the back and right shoulder often occurs during physical examination [7]. In terms of imaging manifestations, magnetic resonance cholangiopancreatography (MRCP), computed tomography (CT), B-ultrasound, T-tube radiography, choledochoscopy, duodenoscopy and other imaging examinations and endoscopy have played an important role in the diagnosis and differential diagnosis of postoperative biliary stenosis [8–10]. Due to the persistence of stenosis, secondary surgery under endoscopy, direct vision or endoscopy is often required, which not only increases the pain of patients, but also wastes medical resources [11, 12].
Aim
In this regard, we reviewed all kinds of case data related to bile duct surgery in our hospital in the past 12 years, aiming to analyze and determine the relevant risk factors before and during the operation, determine the influencing factors of different operation methods or different operation sites for postoperative biliary stricture (POBS), explore the internal relationship of POBS from different sources, and provide ideas for clinical diagnosis and treatment.
Material and methods
Retrospective analysis was performed on the clinical data of 9011 patients who underwent biliary surgery in our hospital in the period from July 2010 to June 2022 due to various diseases. In combination with the inclusion and exclusion criteria, 6783 patients who only underwent gallbladder surgery were excluded, 2228 patients who underwent biliary surgery were collected, and follow-up evaluation of biliary stenosis was carried out from 6 weeks to 3 months after surgery.
Inclusion and exclusion criteria are as follows: Inclusion criteria: (1) age 18–90 years, gender unlimited, with complete case data; (2) manual treatment based on biliary tract diseases, including but not limited to bile duct stones, various acute and chronic cholangitis, benign and malignant bile duct tumors, bile duct cysts, bile duct metastatic cancer, etc.; surgical procedures including but not limited to bile duct incision, exploration, stone removal, resection, suture, shaping, repairing and anastomosis, etc.; (3) cases were confirmed as POBS by, among other things, symptoms, signs, various laboratory and imaging examinations (ultrasound, CT, MRCP, enhanced MR [13], ERCP, T-tube angiography, etc.) or secondary surgery. Exclusion criteria: (1) The patients’ disease requires surgery after various examinations, such as bile duct stones and tumors mentioned in the inclusion criteria, but no surgical treatment is performed, or the surgical method does not involve the bile duct. For example, in patients with advanced biliary tract tumors who have peritoneal metastasis, only local resection and biopsy are performed on the metastatic focus, but the primary focus isn’t treated; (2) accompanied by serious basic diseases, poor general assessment, and may have a greater impact on the prognosis; (3) incomplete case data.
The judgment criteria for biliary stricture include but are not limited to postoperative ultrasound, CT, MR, T-tube angiography, ERCP or secondary surgery findings to confirm that the common bile duct, common hepatic duct and left and right hepatic ducts are locally thinner and narrower, with or without expansion of the upstream bile duct [14]. The clinical risk factors of POBS to be analyzed are classified according to preoperative and intraoperative risk factors, including: sex, age, body mass index (BMI), peripheral blood leukocyte level, C-reactive protein level, conjugated bilirubin (CB) level, albumin level, alkaline phosphatase (ALP) level, γ-glutamyl transferase (γ-GTT) and whether complicated by hypertension, diabetes and other basic diseases. Intraoperative risk factors include: American Society of Anesthesiologists (ASA) score, common bile duct diameter, with or without cholecystectomy, operation time, operation mode, operation position, length of common bile duct incision, and seniority of surgeons. The whole data were grouped according to the above clinical risk factors, and the incidence of POBS in each group was counted.
Statistical analysis
SPSS 25.0 statistical software was used to collate and analyze the data. The χ2 test was used to count the difference in the incidence of POBS in each group under different clinical risk factors. The χ2 test of pairwise comparison was used for biliary stricture in different surgical methods or different parts, and continuity correction of the χ2 test was used when the predicted value was less than 5; Fisher’s exact probability method was used when the predicted value was less than 1; logistic multivariate regression analysis was used to analyze the risk of clinical factors, and the difference was considered statistically significant with p < 0.05.
Results
Due to factors such as patient loss and death due to other complications, a total of 2014 patients were included in this study, with a loss rate of 9.6%. There were 1039 male patients and 975 female patients undergoing biliary surgery, with an average age of 64.5 ±11.7 years. In terms of classification of surgical purposes, 1847 cases were benign biliary surgery, with bile duct stones being the most common (1816 cases, 98.6%). The rest were choledochal cysts, bile plugs, foreign bodies in the biliary tract, chronic sclerosing cholangitis, etc. 108 cases had POBS. 167 cases were malignant biliary operations, most of which were biliary tumors (cholangiocarcinoma and gallbladder cancer, including precancerous lesions of the biliary system) (98 cases, 58.7%), followed by liver cancer operations involving bile ducts (41 cases, 24.6%), and the rest included pancreatic cancer, gastric cancer, colorectal cancer and other bile duct invasion. Forty-one cases had POBS. There is a statistically significant difference in the stenosis rate of benign and malignant diseases (χ2 = 78.2, p < 0.001).
Next, we analyzed the risk factors of benign and malignant POBS cases, and the sorting results are shown in Tables I and II. In the diagnosis and treatment of benign biliary tract diseases, patients aged ≥ 60 years (p = 0.034), complicated with diabetes (p = 0.001), common bile duct diameter < 0.8 cm (p = 0.034), complicated with Mirizzi syndrome (p = 0.001), years of experience of the chief surgeon < 25 years (p = 0.001), and years of operation for the first 6 years (p = 0.015) were more likely to have POBS. But CB ≥ 6.8 μmol/l (p = 0.042), ALP ≥ 125 U/l (p = 0.042), γ-GTT ≥ 50 U/l (p = 0.047), combined with diabetes (p = 0.038), and the experience of the chief surgeon < 25 years (p = 0.008) also play a role in POBS of malignant diseases. Through logistic regression analysis of multiple factors, age, combined with diabetes, diameter of common bile duct, combined with Mirizzi syndrome, and seniority of the chief surgeon will affect the incidence of POBS after benign biliary diseases (Table III). But CB > 6.8 μmol/l and the seniority of the surgeon in charge will affect the stenosis rate of malignant biliary diseases (Table IV).
Table I
Clinicopathological data of postoperative biliary stricture in patients with benign biliary diseases
| Clinicopathological factors | No POBS (1739 cases) | POBS (108 cases) | χ2 | P-value |
|---|---|---|---|---|
| Age [years]: | ||||
| < 60 | 449 | 18 | ||
| ≥ 60 | 1290 | 90 | 4.509 | 0.034 |
| Sex: | ||||
| Male | 898 | 54 | ||
| Female | 841 | 54 | 0.109 | 0.741 |
| BMI [kg/m2]: | ||||
| < 21.7 | 527 | 35 | ||
| ≥ 21.7 | 1212 | 73 | 0.212 | 0.645 |
| Time of onset [months]: | ||||
| < 1 | 1130 | 61 | ||
| > 1 | 609 | 47 | 3.207 | 0.073 |
| Peripheral blood leukocytes [×109/l] | ||||
| < 12 | 1554 | 95 | ||
| ≥ 12 | 185 | 13 | 0.208 | 0.648 |
| C-reactive protein [mg/l]: | ||||
| < 10 | 610 | 32 | ||
| ≥ 10 | 1129 | 76 | 1.331 | 0.249 |
| Conjugated bilirubin [μmol/l]: | ||||
| < 6.8 | 668 | 43 | ||
| ≥ 6.8 | 1071 | 65 | 0.084 | 0.771 |
| Albumin [g/l]: | ||||
| < 35 | 1023 | 67 | ||
| ≥ 35 | 716 | 41 | 0.433 | 0.510 |
| Alkaline phosphatase [U/l]: | ||||
| < 125 | 647 | 34 | ||
| ≥ 125 | 1092 | 74 | 1.431 | 0.232 |
| γ-glutamyl transferase [U/l]: | ||||
| < 50 | 477 | 25 | ||
| ≥ 50 | 1262 | 83 | 0.942 | 0.332 |
| Hypertension: | ||||
| Yes | 524 | 31 | ||
| No | 1215 | 77 | 0.099 | 0.753 |
| Diabetes: | ||||
| Yes | 478 | 46 | ||
| No | 1261 | 62 | 11.418 | 0.001 |
| ASA score: | ||||
| ≤ 2 | 1341 | 77 | ||
| ≥ 3 | 398 | 31 | 1.930 | 0.165 |
| Common bile duct diameter [cm]: | ||||
| ≤ 0.8 | 496 | 42 | ||
| > 0.8 | 1243 | 66 | 5.294 | 0.021 |
| Cholecystectomy: | ||||
| Yes | 1582 | 93 | ||
| No | 157 | 15 | 2.845 | 0.092 |
| Mirizzi syndrome*: | ||||
| Yes | 173 | 23 | ||
| No | 1566 | 85 | 13.805 | 0.001 |
| Severity grading: | ||||
| 0–I | 1304 | 73 | ||
| II–III | 435 | 35 | 2.930 | 0.087 |
| Operation time: | ||||
| ≤ 150 min | 1009 | 55 | ||
| > 150 min | 730 | 53 | 2.097 | 0.148 |
| Operation mode: | ||||
| LCBDE + T-tube drainage | 1077 | 53 | ||
| LCBDE + primary suture | 52 | 9 | ||
| Laparoscopic transcystic biliary exploration | 43 | 3 | ||
| OCBDE + T-tube drainage | 536 | 41 | ||
| Choledochojejunostomy | 31 | 2 | 11.958 | 0.013 |
| Length of bile duct incision [cm]: | ||||
| ≤ 2 | 1061 | 63 | ||
| > 2 | 678 | 45 | 0.306 | 0.580 |
| Seniority of surgeon [years]: | ||||
| < 25 | 574 | 57 | ||
| ≥ 25 | 1165 | 51 | 17.671 | 0.001 |
| Year of operation: | ||||
| First 6 years | 696 | 56 | ||
| Last 6 years | 1043 | 52 | 5.894 | 0.015 |
Table II
Clinicopathological data of postoperative biliary stricture in patients with malignant biliary diseases
| Clinicopathological factors | No POBS (126 cases) | POBS (41 cases) | χ2 | P-value |
|---|---|---|---|---|
| Age [years]: | ||||
| < 60 | 48 | 11 | ||
| ≥ 60 | 119 | 30 | 0.059 | 0.808 |
| Sex: | ||||
| Male | 61 | 21 | ||
| Female | 68 | 20 | 0.193 | 0.668 |
| BMI [kg/m2]: | ||||
| < 21.7 | 66 | 19 | ||
| ≥ 21.7 | 63 | 22 | 0.289 | 0.591 |
| Conjugated bilirubin [μmol/l]: | ||||
| < 6.8 | 31 | 4 | ||
| ≥ 6.8 | 95 | 37 | 4.116 | 0.042 |
| Albumin [g/l]: | ||||
| < 35 | 107 | 33 | ||
| ≥ 35 | 21 | 8 | 0.211 | 0.646 |
| Alkaline phosphatase [U/l]: | ||||
| < 125 | 35 | 5 | ||
| ≥ 125 | 91 | 36 | 4.124 | 0.042 |
| γ-glutamyl transferase [U/l]: | ||||
| < 50 | 42 | 7 | ||
| ≥ 50 | 84 | 34 | 3.945 | 0.047 |
| Time of onset [months]: | ||||
| < 1 | 57 | 17 | ||
| > 1 | 69 | 24 | 0.179 | 0.673 |
| Hypertension: | ||||
| Yes | 27 | 9 | ||
| No | 99 | 32 | 0.005 | 0.944 |
| Diabetes: | ||||
| Yes | 21 | 13 | ||
| No | 105 | 28 | 4.316 | 0.038 |
| ASA score: | ||||
| ≤ 2 | 44 | 14 | ||
| ≥ 3 | 82 | 27 | 0.008 | 0.928 |
| Common bile duct diameter [cm]: | ||||
| ≤ 0.8 | 34 | 16 | ||
| > 0.8 | 92 | 25 | 2.138 | 0.144 |
| Cholecystectomy: | ||||
| Yes | 112 | 35 | ||
| No | 14 | 6 | 0.107 | 0.744 |
| Operation time [min]: | ||||
| ≤ 240 | 81 | 19 | ||
| > 240 | 86 | 22 | 0.062 | 0.804 |
| Operation position: | ||||
| Left/right hepatic duct | 41 | 15 | ||
| Junction and common hepatic duct | 45 | 4 | ||
| Middle upper segment of common bile duct* | 28 | 11 | ||
| Lower segment of common bile duct* | 53 | 11 | 7.935 | 0.041 |
| Seniority of surgeon [years]: | ||||
| < 25 | 31 | 19 | ||
| ≥ 25 | 95 | 22 | 6.969 | |
| Year of operation: | 0.008 | |||
| First 6 years | 54 | 21 | ||
| Last 6 years | 72 | 20 | 0.874 | 0.350 |
Table III
Multivariate logistic regression analysis of benign biliary stricture
Table IV
Multivariate logistic regression analysis of malignant biliary stricture
Different surgical methods will affect the incidence of POBS (χ2 = 9.717, p = 0.034). Through the χ2 test, we found that laparoscopic common bile duct exploration (LCBDE) + T tube drainage had a lower probability of stenosis than LCBDE + primary suture (p = 0.002). Setting the detection probability of the pairwise χ2 test α’ = 0.005, endoscopic surgery could not reduce the stenosis rate compared with open surgery (p = 0.031). The choice of surgical site has an impact on the incidence of POBS of malignant biliary diseases (χ2 = 7.935, p = 0.041), in the pairwise χ2 test, the detection probability α’= 0.008, the incidence of stenosis at the confluence site after surgery was not statistically related to the left/right hepatic duct (p = 0.013) or the upper middle segment of the common bile duct (p = 0.013).
Discussion
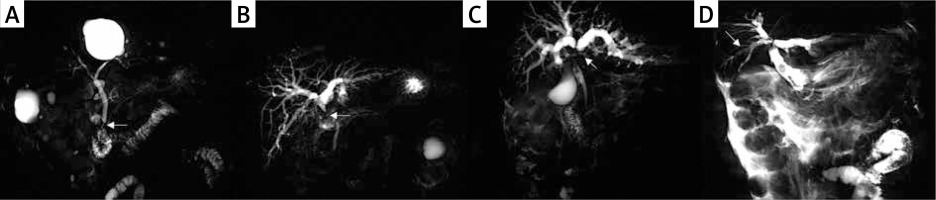
It is necessary and feasible to study the risk factors of POBS. First of all, POBS is universal. Biliary diseases are a large class of surgical diseases with a high incidence rate and a variety of types. Biliary stones are the most common benign diseases. According to the National Institute for Health and Care Excellence (NICE) consensus in 2016, 10–15% of British adults suffer from gallbladder stones and/or bile duct stones, 50% of symptomatic patients will have recurrent abdominal pain, fever, jaundice and other symptoms, and 1–2% of patients will have serious complications [15]. Cholangiocarcinoma is the most common malignant disease, originating from biliary epithelial cells. Its incidence rate is rising steadily worldwide, and it is currently listed as the second most common hepatobiliary tumor [16]. Because the hepatoduodenal ligament and its adjacent structures have an important position as a “crossroad”, the selection of surgical methods and the treatment of postoperative complications have become one of the key challenges faced by general surgeons. Postoperative biliary fistula, fluid accumulation in the operation area, bile duct infection, biliary bleeding, biliary stricture, etc. are common complications, which are related to the hospital stay, survival period and long-term prognosis of patients [17]. Secondly, the body damage of patients with biliary stricture is long-term and persistent. As one of the most common postoperative complications of the three major bile ducts, long-term retrohepatic bile duct obstruction of patients with biliary stricture can lead to liver fibrosis at different levels, thus affecting liver function for a long time, inducing liver failure and cirrhosis. Removing the obstruction can alleviate the process of liver fibrosis, but it cannot reverse fibrous hepatocytes [18]. In addition, hepatic duct stricture after surgery for intrahepatic calculi is an important risk factor for patients to have long-term complications, such as recurrent cholangitis, cirrhosis and cholangiocarcinoma [19], and bile duct stricture in different parts will also affect the occurrence of postoperative cholangitis. The incidence of postoperative stenosis cholangitis in the common hepatic duct or bile duct is higher than that in the single intrahepatic bile duct stricture [20]. Thirdly, similar with bile duct bleeding and infection, laboratory tests and imaging examinations can be confirmed in patients with POBS before secondary surgery. Although the current literature does not quantitatively determine the specific value of bile duct stricture in different parts, considering that the bile duct is a hollow organ, the recognized biliary stricture in the imaging field is still a qualitative diagnostic standard: namely, the thinning and narrowing of the downstream bile duct and the expansion of the upstream bile duct, and Bismuth classification is still considered as the gold standard for biliary stenosis classification [21]. Surgeons can evaluate the position and severity of stenosis of patients according to the changes of clinical, laboratory and imaging indicators, so as to timely intervene to avoid damage of liver function (Photo 1).
Photo 1
T-tube angiographic image of bile duct stricture after operation of benign biliary tract disease, with arrows indicating the position of stricture. A – Bismuth I, B – Bismuth II, C – Bismuth III, D – Bismuth IV
By analyzing the risk factors of biliary stricture, we found that age > 60 years, bile duct diameter < 0.8 cm, and Mirizzi syndrome were unique risk factors for benign biliary stricture, while CB > 6.8 μmol/l, ALP ≥ 125 U/l and γ-GTT ≥ 50 U/l is unique to malignant biliary stricture. The reason might be related to the difference and relationship between inflammation and cancer. Recent studies tend to suggest that chronic inflammatory conditions, such as certain infections, toxic substances and even immune disorders, may ultimately be related to the development of cancer [22]. However, there are many differences between the two in the fields of histopathology, molecular biology, etc., which make different risk factors have different diagnostic and predictive values. Older people often have multiple biliary tract inflammation, with close tissue adhesion and difficult anatomy. The diameter of the bile duct determines the tolerance rate for incision and suture. Biliary tract variation, especially when Mirizzi syndrome occurs, affects the surgeon’s interpretation of the correct anatomical structure. In malignant biliary diseases, the degree of POBS is related to the type and malignancy of the tumor itself [23, 24], and the level of blood CB and ALP is one of the important laboratory indicators reflecting cholestasis [25]. Since it is difficult to judge the aggressiveness and malignancy of different cancer foci, we choose CB as the evaluation index of tumor bile duct invasion before surgery, which also becomes a risk factor for malignant POBS. ALP and γ-GTT as specific indicators reflecting biliary obstruction and liver function damage are of great significance in the identification of POBS [26]. However, preoperative CB and ALP cannot well reflect the incidence of benign POBS, which is inconsistent with the study of Lokesh et al. [27]. It is considered that this difference might be because of the time of laboratory examination. The blood indexes reflecting bile duct obstruction before the operation cannot well reflect the progress of benign stenosis after the operation. However, the influence of the combination of inflammation, bile duct variation, and age has no significance in the diagnosis of malignant POBS. On the other hand, the combination of diabetes and the low seniority of the chief surgeon will affect the incidence of benign and malignant POBS. It is considered that they either reduce the healing ability of the body or have a higher possibility of iatrogenic biliary tract injury.
The incidence of biliary stricture varies in different surgical methods. Although many previous reports have shown that LCBDE + primary suture is more and more widely used in patients with biliary stones with less inflammation, similar to the study of Tang et al. [28], the traditional LCBDE + T-tube drainage has a lower incidence of POBS. An earlier study also pointed out that if it is suspected that POBS occurs, long-term retention of T-tubes is a necessary measure for treatment [29]. Indwelling T tube can improve the prognosis of patients with POBS [30]. Although it has been reported in the past that laparoscopic biliary surgery has more biliary complications than open surgery [31], with the progress of endoscopic technology, the incidence of complications has been on a par with open surgery. On the other hand, for malignant biliary diseases, the surgical site has a correlation with the incidence of POBS. However, due to the limited number of included samples, the specific differences are not clear, and the sample size still needs to be further expanded for classification research.
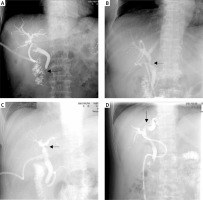
In addition, this study still has some limitations. Because the judgment of POBS is mainly based on the imaging examination in the follow-up period, some patients will be misjudged and missed. In clinical practice, it was also found that some patients only had biliary stricture on imaging (Photo 2), but the clinical symptoms, signs or endoscopy did not show corresponding characteristic changes. Conversely, some imaging examinations could not display the abnormalities of biliary stricture [32]. Thus, invasive surgery such as endoscopic techniques can assist surgeons in the diagnosis and treatment of biliary stricture under direct vision. Various endoscopic techniques can have different effects on the prognosis of patients with benign and malignant biliary stricture, and their surgical success rate, bile duct restenosis and functional recovery vary [1, 33]. However, as in non-invasive diagnostic methods, “stenosis” in the imaging report can indicate the possible pathophysiological changes of the surgical patient, facilitate the formulation of a diagnosis and treatment plan that is beneficial to the patient, and as a transcendental examination method, facilitate the early treatment of biliary complications, actively intervene, and reduce organ function damage.
Conclusions
POBS is one of the common complications after biliary surgery, and its probability of occurrence is related to many risk factors. Compared with biliary bleeding and biliary infection, its incidence is relatively hidden, which also puts forward new requirements for biliary surgeons. However, it is still the most basic diagnostic and treatment processes to evaluate patients before an operation, standardize surgery during an operation, avoid structural damage, and regularly review and follow up after the operation. By analyzing the risk factors of POBS, this article aims to improve the basic understanding of such diseases, deal with the risk factors accordingly, strengthen the study and training of diagnostic and therapeutic norms, reduce the occurrence of iatrogenic biliary injury, and benefit patients.