Introduction
IgG4-related disease (IgG4-RD) is an immune-mediated fibroinflammatory condition characterized by a myriad of clinical presentations with the potential to lead to multi-organ involvement [1].
Despite an increasing number of cases being reported globally, it is still considered to be an uncommon aetiology, and awareness among physicians is low. The involved organs share several core pathologic features and striking clinical and serologic similarities, including tumour-like swelling of involved organs, a lymphoplasmacytic infiltrate enriched with IgG4-positive plasma cells, and a variable degree of fibrosis that has a characteristic “storiform” pattern [2].
The disorder was initially described in relation to the pancreas, but improved recognition has led to the identification of extra-pancreatic manifestations as well. The most common presentation is that of a subacute development of a mass in the affected organ with signs and symptoms attributable to local site compression [3, 4].
Commonly involved organs include the pancreas, salivary glands, kidneys, orbits and lacrimal glands, aorta and the retroperitoneum, meninges, and the thyroid gland [5, 6].
Most patients are diagnosed late owning to the slowly progressive nature of the disease, and classification criteria have been published to improve this situation [1, 7].
The entry criteria for these require typical clinical, radiological, or pathological involvement of organs/systems while excluding mimics. This exclusion involves specific serological, radiological, and pathological criteria while important clinical exclusions include prominent fever or a lack of response to a trial of glucocorticosteroids [1].
Treatment is largely based upon observational data including case reports and case series, and while the optimal therapy remains to be determined. A good initial response to glucocorticosteroids is characteristic, particularly if excessive tissue fibrosis has not supervened, thereby making diagnostic delays especially problematic [1]. Additional immunosuppressive agents or steroid-sparing strategies are also sometimes required as per clinical response and disease burden [8].
As literature from the Indian subcontinent is scarce [9–11], we conducted this study to determine the spectrum of extra-pancreatic manifestations of IgG4-RD at our institute, highlighting the role of heightened suspicion and prompt therapy for this otherwise overlooked disease entity.
Material and methods
This retrospective study was conducted at a tertiary care centre of north India from January 2017 to January 2021 after obtaining ethical clearance from the Institute Ethics Committee. The primary objective of the study was to establish the clinical presentation, and laboratory and radiological findings of extra-pancreatic IgG4-RD cases presenting to our institute.
Patients were screened from the outpatient and inpatient facilities of the Department of Medicine, where patients came via self-referral or after a referral from other specialities of the hospital or other hospitals.
All patients over 18 years of age were included if they had a diagnosis of IgG4-RD established by typical imaging and elevated serum IgG4 levels and/or histopathology. Their treatment charts and follow-up data were reviewed and recorded.
Previously published diagnostic criteria by Umehara et al. [12] were used to categorize IgG4-RD as definite, probable, or possible. Patients lacking either radiological corroboration or serum IgG4 levels, or with an alternate diagnosis during workup were excluded from the analysis. Patients with radiological/clinical evidence suggestive of pancreatic involvement were also excluded from the study.
Cases were reviewed for the clinical presentation, the diagnostic workup, and the final histopathological basis for diagnosis along with data on symptom response during follow-up after the institution of treatment.
Clinical and laboratory data collected for the evaluation of IgG4-RD in included patients consisted of complete blood count, renal and liver function tests, and tests for erythrocyte sedimentation rate, C-reactive protein, and serum IgG4 levels.
Radiological imaging included computed tomography (CT), magnetic resonance imaging (MRI) scans of specific sites involved with or without contrast, as well as F-18 fluorodeoxyglucose (FDG) positron emission tomography and computed tomography (PET/CT) scans.
The most accessible tissue and least invasive methods were used to establish histological diagnoses guided by radiological findings. The duration and dose of corticosteroids and steroid-sparing agents with the response to treatment were recorded.
Statistical analysis
The data were analysed using Stata/SE v12.1 software. Continuous variables were summarized by median (range: min–max) while categorical data were described as frequency or proportion.
Correlation between quantitative values was tested using Spearman’s rank-order correlation. Group comparisons for continuous data were done either with Mann-Whitney U test (independent observations) or Wilcoxon signed-rank test (for repeated measurements), while the χ2 test or Fischer’s exact test was used for the analysis of categorical data. A p-value of < 0.05 was considered statistically significant.
Results
Patient characteristics
A total of 56 patients with a provisional diagnosis of IgG4-RD at the time of recruitment were screened for this study. Of these, 6 did not have radiological evidence, 4 did not have serum IgG4 levels, and 32 had an alternate diagnosis on workup and were therefore excluded from the final analysis.
A total of n = 14 patients with a diagnosis of extra-pancreatic IgG4-related disease were subsequently included in the analysis. Baseline characteristics are summarized in Table I, while baseline investigations are shown in Table II.
Table I
Baseline characteristics of the patients with IgG4-related disease (n = 14)
Table II
Investigations at baseline of the recruited patients with IgG4-related disease (n = 14)
The median age of presentation was 39 years (range 19–56 years); 71% (n = 10) of patients were males. The median delay in diagnosis from the time of symptom onset was 9.5 months (range 2–72 months). Serum IgG4 levels were elevated above the upper range (200 mg/dl) in 10 of 14 cases (71.43%). A total of 8 patients were definite IgG4-RD while 6 were possible IgG4-RD.
Presentation
All patients presented due to gradually enlarging swellings in the affected organs or mass effects related to the same. Constitutional symptoms like fever were seen in 3 of 14 (21.4%) while weight loss was a presenting complain in 4 of 14 (28.6%). Pain in the involved areas was present in 8 of 14 (57.1%) cases. Patients had a long duration of symptoms with a median time to diagnosis of 9.5 months.
Imaging
Reports from cross-sectional imaging of involved areas consisted of CT scans for 8 patients and MRI for 6 patients. All patients had a soft tissue mass on imaging (14 of 14).
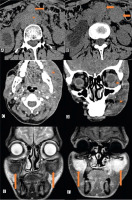
Figure 1 below depicts the changes as seen on cross-sectional imaging. Details of contrast characteristics were available for 7 patients, and all of these showed contrast enhancement. In patients with retroperitoneal involvement, the kidneys were involved in 3 of 5 (60%) cases.
Fig. 1
Non-contrast computed tomography of the abdomen showing homogenous, sheet-like retroperitoneal soft tissue (*) around the abdominal aorta, with bilateral hydroureteronephrosis (A, B). Two separate peritoneal nodules are also seen (arrows). Unilateral left maxillary and periorbital soft tissue, which is uniformly hypodense on contrast-enhanced computed tomography images (*) (C, D). Another patient with bilateral maxillary involvement on T2W (C) and contrast-enhanced magnetic resonance imaging (D) images (arrow) (E, F).
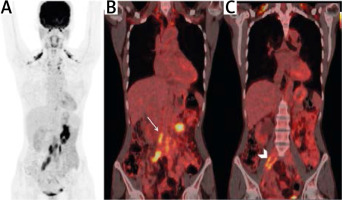
F-18 fluorodeoxyglucose PET/CT reports with uptake characteristics were available for 9 of the 14 patients and showed uptake in 8 of the 9 cases (88.9%). All patients with retroperitoneal involvement (n = 5) underwent PET/CT, with FDG uptake seen in all the cases. Only one case with skull base and cavernous sinus involvement had a report that did not comment on FDG avidity. Representative images from the FDG-PET/CT scan are shown in Figure 2 below.
Histopathology
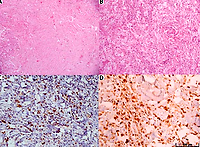
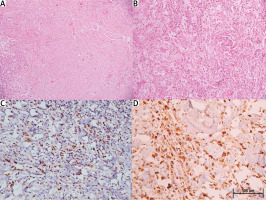
Out of the 14 cases, 12 underwent biopsy with detailed histopathological evaluation. Overall, the most common findings were lymphoplasmacytic infiltrates that were present in 100% (12 of 12) of the cases, followed by IgG4 positivity in 66.7% (8 of 12 cases), storiform fibrosis in 58.3% (7 of 12), and obliterative phlebitis in 8.3% (1/12) of cases with available biopsies. Representative slides are shown in Figure 3 below.
Fig. 3
Photomicrographs showing spindle shaped fibroblastic stroma arranged in a storiform pattern alternating with inflammatory areas comprising plasma cells, lymphocytes, and foamy macrophages (A, B, H&E, 100× and 200×, respectively) (A, B). Immunostaining for CD138 and IgG4 highlighting plasma cells (200× and 400×, respectively) (C, D).
Statistical analysis
There was no statistically significant correlation between erythrocyte sedimentation rate (ESR), C-reactive protein (CRP), and IgG4 levels at baseline or follow-up. Patients with fever had a trend towards elevated ESR (p = 0.05) but not CRP (p = 0.40), while weight loss was not significantly associated with either (p = 0.07 and p = 0.30, respectively).
F-18 fluorodeoxyglucose uptake was not associated with the presence of fever or weight loss (p = 0.24 and p = 0.7, respectively).
Treatment response
All patients were treated with oral prednisolone at a dose of 0.5–1 mg/kg/day, and the median duration of follow-up was 10 months (range 0–18 months). Clinical improvement with regards to initial presenting symptoms was observed in all patients.
Follow-up showed a statistically significant reduction in ESR levels from median 53.5 to 24 (p = 0.002), and CRP levels from median 9.5 to 1.8 (p = 0.003). Follow-up serum IgG4 levels were only available in 2 patients, and they were below the upper range of normal.
Corticosteroids were successfully tapered in all the 12 (100%) patients who remained in follow-up; 5 of 14 (35.71%) of these patients were maintained only on azathioprine without the need for corticosteroids. These data are summarized below in Table III.
Table III
Follow-up of cases
Discussion
We herein provide a retrospective analysis of 14 patients; to our knowledge, this is the largest study of its kind from India [9, 10].
Our study provides pertinent information regarding the expression of IgG4-RD in the cohort of patients from the Indian subcontinent. All patients presented with symptoms related to local effects arising from IgG4-RD, and most of them had been afflicted for several months (median 9.5 months) before a diagnosis was made. This is consistent with the usual clinical manifestations noted with IgG4-RD [3, 4].
The most common sites of extra-pancreatic involvement included the paranasal sinuses (n = 7, 50%) followed by the retroperitoneum (n = 5, 35.7%). This may, however, not necessarily be reflective of the overall incidence of IgG4-RD presentation because this could have been related to a referral bias, with more patients having unexplained abdominal pain or headache being referred to our tertiary care centre for evaluation.
The presence of localized symptoms and the long time to diagnosis highlights the need for physicians to have a lower threshold to initiate diagnostic testing in the presence of such symptoms.
Unlike prior publications [3] and recently published classification criteria by the American College of Rheumatology/European League Against Rheumatism (ACR/EULAR) suggesting that clinicians rule out IgG4-RD in patients with significant fever [1], 3 out of 14 patients in our study had fever at presentation (2 of 3 with definite and 1 with possible IgG4-RD).
This may warrant further characterization, especially in the Indian population, with due cognizance of this facet while evaluating suspected cases using existing classification criteria. IgG4-related disease can mimic several disorders with the presence of constitutional symptoms including fever in India (where an important differential is tuberculosis).
Therefore, a detailed diagnostic workup must exclude infections before instituting a careful trial of corticosteroids. The long median time to diagnosis of 9.5 months after symptom onset reflects the need for the necessary work-up for excluding mimics before a diagnosis is made.
Inflammatory markers such as ESR and CRP although elevated in the majority of our patients (100% and 57.1%) are not sufficiently specific to identify IgG4-RD, as has been found in existing literature [13].
However, at a laboratory-specific cut-off of 200 mg/dl, serum IgG4 was elevated in 10 of 14 cases (71.4%), which improved to 92.8% using a cut-off of 135 mg/dl.
This high sensitivity is similar to data from other studies [14–16] including a meta-analysis by Hao et al. [17] that showed a sensitivity of 87.2% (95% CI: 85.2–89.0) and a specificity of 82.6% (95% CI: 81.6–83.6) for cut-offs ranging from 135 to 144 mg/dl. It also highlights the need for standardization of assays across various laboratories to ensure the optimal performance of this test.
The radiological assessment helped to confirm the presence of soft tissue masses and enabled guidance for biopsies. Cross-sectional imaging must therefore be amongst the first-line investigations in patients with unexplained symptoms within local sites [18].
F-18 fluorodeoxyglucose PET/CT is proposed as a diagnostic modality in the newly proposed criteria for the diagnosis of IgG4-RD because it reflects the glucose metabolism and therefore disease activity, in comparison to conventional contrast studies. It can serve as a sensitive tool for defining the extent of organ involvement and aids in the selection of adequate biopsy sites [19].
As is expected in an inflammatory process, contrast enhancement was present in all cases with available reports for contrast-enhanced studies, and FDG-PET showed uptake in close to 90% of the cases. However, being a fibrotic process, contrast enhancement will be present once fibrosis occurs and may not reflect disease activity as much as the extent of fibrosis [19].
Histopathology showed that all the patients who underwent histopathological examination had a lymphoplasmacytic infiltrate, and as such this finding should alert pathologists and clinicians to the possibility of IgG4-RD after common causes are ruled out with further characterization including IgG4 subclass staining (present in 66.7% of the cases) and closer examination for the presence of storiform fibrosis (58.3% of cases) and obliterative phlebitis (8.3% of cases) for diagnosis.
This is in agreement with the 2019 ACR/EULAR classification criteria [1] using lymphocytic infiltrate as one of the entry criteria for diagnosis. This also underscores the need for core biopsies in contrast to fine-needle aspirate samples.
Like other studies [20], while using standard treatment regimens including corticosteroids, there was a robust response both clinically as well as in inflammatory markers. Keeping in mind the median delay of 9.5 months in diagnosis along with the excellent response to treatment and overall favourable prognosis, it becomes imperative to be aware of the possibility of IgG4-RD while evaluating patients presenting with mass lesions or with local symptoms from mass effect.
Furthermore, our experience shows that steroids can be safely tapered in the majority of cases, and attempts to transition exclusively to steroid-sparing agents such as azathioprine can be successful in a significant proportion, as well.
Study limitations
Our study has limitations owing to the retrospective design. The number of cases in the study for individual organs precludes discernment of organ-specific behaviour of this disease.
While the median duration of follow-up was 10 months, longer follow-ups with larger cohorts will be important to better understand the waxing-waning course of the disease where long-term immunosuppression may have important consequences, especially in a country like ours.
Despite the fact that the final number of analysed patients with confirmed IgG4-RD is relatively small (depending on presentation during the analyzed period at our center), it is nonetheless reflective of real world experience.
Future case-control studies between confirmed IgG4-RD and IgG4-RD mimics in similar settings can also provide important clinical and diagnostic information that cannot be inferred from the data obtained for the current study.
Conclusions
IgG4-related disease is a slowly progressive disease that presents with soft tissue masses causing local symptoms, which may be non-specific to begin with. Hence, it is pertinent to consider it as a part of the differential diagnoses early during the evaluation.
The Indian subcontinent has a high infectious disease burden and 3 of our 14 patients had a prominent fever, which is an exclusion criterion as per the ACR/EULAR 2019 classification criteria for IgG4-RD, and this problem warrants further discussion.
Cross-sectional imaging followed by guided tissue sampling can help establish the diagnosis more rapidly. Clinicians must familiarize themselves with the various clinical manifestations of this disease to reduce delays, especially in view of the remarkable therapeutic response and excellent prognosis.