The use of opioids to treat chronic non-cancer pain has increased dramatically in the last decade, particularly in North America [1]. Simultaneously, this increase in prescribing practice has been correlated with an almost four-fold increase in opioid- related deaths (i.e. overdosing) [2]. In Canada, apparent opioid-related overdose deaths have claimed the lives of over 11,500 people between 2016 and 2018 [3]. Moreover, postoperative chronic pain is common (experienced by as many as one third of surgical patients) and often dealt with by prescribing opioids inappropriately without long-term follow-up [4]. Despite recent efforts to control the prescription of opioids and the introduction of abuse-deterrent opioid formulations, the epidemic is still yet to be fully targeted with a multi-factorial approach [5]. This review will describe a brief history of the opioid epidemic, its current state, and provide a detailed description of the concept of a Transitional Pain Service (TPS). A TPS is a novel program which aims to effectively manage acute pain post-operatively, facilitate opioid weaning, reduce the development of chronic pain disability, and potentially help to flatten the worrisome trajectory of opioid-related deaths associated with excessive opioid prescribing.
HISTORY OF THE OPIOID EPIDEMIC
In order to give some context into how the opioid epidemic arose, it is imperative to examine patterns beginning in the 1980s. At that time, pharmaceutical companies marketed the use of opioids to treat pain and assured clinicians that the addiction profile of these analgesic agents was low [6]. Within a few years, the same companies promoted these drugs for use in long-term non-cancer pain, despite the lack of good evidence to ensure efficacy in the treatment of chronic pain in this population [7]. This misrepresentation on the part of the pharmaceutical companies sparked the first of three major waves of increased opioid prescribing. The number of opioid prescriptions increased among primary care clinics and hospitals [2], and with that an increased amount of opioids available for diversion – unlawful channeling of regulated pharmaceuticals from legal sources to the illicit marketplace [8] – took place [2]. Pharmaceutical companies responded to the public outcry by developing a sustained-release opioid formulation know as oxycodone (branded as OxyContin). This new formulation required fewer administrations daily, compared to taking pain medications every 2–4 hours as previously [9]. The Food and Drug Administration concluded that the sustained-release formulation of oxycodone would prevent abusive drug behavior due to its delay in reward which is necessary to reinforce addictive behavior [9]. This resulted in a false sense of security among prescribers, leading to an increase in OxyContin prescribing from 1997 to 2002 [2]. However, users circumvented the sustained release by crushing the tablets and delivering the medication intranasally or intravenously to deliver a potent dose of the drug. This led to increased rates of opioid oversedation [5] and eventually more opioid-related deaths. In light of these events, Purdue Pharma pleaded guilty to understating the risk of addiction with respect to OxyContin [2]. Effective March 1, 2012 Purdue Pharma pulled OxyContin from the market and introduced OxyNeo – an oxycodone formulation with a tamper-resistant tablet to reduce abuse [10].
The second wave began around 2010, as the increased media attention to the crisis led to an inability to access prescription opioids. This inevitably led to the rise in popularity of a cheap, widely available option – heroin [11]. Corresponding with the rise in heroin use, this marked the peak of the annual prescribing rate and average daily morphine equivalents (MEQ) in a decade or so [12]. In 2015, nearly a quarter of drug overdose deaths were due to heroin making up over 15,000 deaths that year in the U.S. [2]. Despite best efforts from clinicians, government agencies, and public health workers, the problem was still rapidly growing, making any solution proposed seem unfeasible.
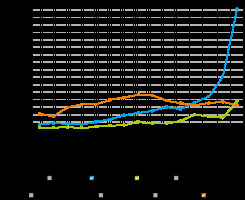
Finally, the last resurgence was seen in 2013 (Figure 1) and was likely due to synthetic analog derivatives of fentanyl such as acetylfentanyl, butyrylfentanyl, carfentanil and W-18 [13]. The sharpest increase in drug-related deaths due to fentanyl, however, occurred in 2016, accounting for 20,000 deaths in the U.S. [14]. A substantial amount of the fentanyl consumed in Canada has been noted to originate from overseas through illicit trade as opposed to prescription for chronic pain [15]. These astounding statistics provided the framework for motivation and need for hospital level changes through better education and more secure storage of these drugs. The Nova Scotian, British Columbian and Ontario’s government responded by utilizing a Narcotics Monitoring System to ensure adequate monitoring of prescription opioids [16].
CHRONIC POST-SURGICAL PAIN
Expectedly, when a patient undergoes a surgical procedure, some level of pain is experienced postoperatively, and the severity and duration vary from patient to patient [17]. When this pain develops after surgery and persists for at least 2 months, it is referred to as chronic post-surgical pain (CPSP) [18]. Opioid prescribing rates have increased as a result of an increased demand for pain management [19]. Additionally, in a recent review of US insurance data, surgical patients’ mean total oral MEQ per prescription has almost doubled from 240 mg in 2010 to 403 mg in 2016 [19].
The heterogeneity in the patient pain experience relies on a multitude of factors related to the patient, type of procedure, and postoperative recovery environment. The primary predictor of CPSP is a prior pain history, while poorly controlled acute postoperative pain is important to consider, as well as other patient-related factors including female gender and younger age [17, 20–22]. Surgical risk factors for CPSP such as the surgical approach (open vs. laparoscopic) and duration of surgery must be considered. Finally, psychosocial constructs that are often left unrecognized are increased preoperative anxiety, poor social supports, and engaging in catastrophizing behavior – predicting a negative pain outcome related to the surgery [17].
The literature suggests that patients are currently complaining more of pain. The PAIN OUT study demonstrated that patients in American society complain more of similarly painful experiences versus their European and international counterparts [23]. After similar orthopedic procedures, American patients rated higher “worst pain” scores than the international cohort (made up of European patients and patients outside of Europe). Interestingly, the difference in pain experiences was independent of typical risk factors such as female gender, younger age, high body mass index (BMI), chronic pain, and opioid use before surgery [23]. In a comparison of American and Dutch patients following hip and ankle surgery, American patients required post-discharge opioid prescriptions in 77% of cases compared to none among Dutch patients, reflecting as much a prescribing cultural difference as well as a philosophical pain management difference [24].
ISSUES OF POSTOPERATIVE OPIOID PRESCRIBING
Data demonstrate that a significant number of patients experience CPSP at three months postoperatively, particularly patients who have undergone thoracic and breast surgery [25]. Opioids are often prescribed to address this in the acute postoperative setting; however, once re-prescribed, this is typically done without a long-term plan, or with a plan to eventually wean these medications. Alam and colleagues noted that patients receiving an opioid prescription within the first week of surgery were 44% more likely to be taking them one year later compared to those who did not receive the same prescription [26]. Opioids were found to increase the risk of road trauma by 21% even at low doses (20–49 MEQ), while patients taking moderate to high doses of opioids were at 29–42% increased odds of motor vehicle accidents [27]. There are also data to suggest that opioid-related deaths have increased as a result of opioid prescribing, over half of which have been deemed unintentional [28]. Finally, opioid use was associated with a higher prevalence of constipation and reduced quality of life in patients with an opioid dependence [29].
Patients are ultimately left with inappropriately managed chronic pain and almost half of these patients continue to use opioids as more of a “band-aid” solution. The issue becomes more complex as these patients seek care from their primary care physicians and surgeons. These physicians often struggle with complex chronic pain management and lack the expertise to wean patients off opioids [25]. With CPSP estimated to cost U.S. $41,000–43,000 annually per patient, it is important that there are programs and processes in place to address this issue [25, 30]. Mayo Clinic recently developed guidelines to aid practitioners in prescribing opioids for adults upon discharge after common surgical procedures [31]. Educational interventions focused on educating surgeons on using NSAIDs and acetaminophen before prescribing opioids led to a 53% decrease in the amount of opioids prescribed to general surgery patients [32].
Although some success has been seen with the implementation of prescribing guidelines and educational interventions, new programs need to be implemented to address this systemic issue. This led to the introduction of a Toronto General Hospital based TPS with a focus on patients at high risk of developing chronic pain and patients with complex pain management and opioid weaning.
CONCEPT OF A TRANSITIONAL PAIN SERVICE
The TPS was created to effectively manage pa-tients’ perioperative pain long-term if needed, maintain function, reduce opioid consumption, and monitor the efficacy of these interventions. Approximately 7% of all major surgical interventions result in a pain disability problem one year later, and this has implications for both CPSP and long-term opioid use with respect to patient recovery. The TPS was launched in 2014 and it is the first program of its kind to target CPSP at three stages: 1) preoperatively, 2) postoperatively in the hospital setting, 3) postoperatively in the outpatient setting for up to 6 months after surgery [30, 33]. The services offered by the TPS are broken down into three categories: 1) Introduction and optimization of multimodal analgesia to both improve pain management and facilitate weaning from opioids; 2) non-pharmacologic interventions including physiotherapy and acupuncture; 3) psychological interventions by a trained pain psychology team around an acceptance and commitment therapy (ACT) model [30]. The focus of ACT is pain education and pain coping by fostering commitment in valued life activities, while encouraging acceptance and mindfulness of the distressing pain experience [34, 35].
The TPS treats two types of patients: opioid-naïve patients without a preexisting pain condition (5–10% risk for the development of moderate to severe CPSP) and patients presenting with a preexisting pain condition or on preoperative opioid medications (population ranges from 10–20%, 12.5% among patients at Toronto General Hospital) [30, 36]. This is managed through an interdisciplinary team which is composed of anesthesiologists, acute pain nurse practitioners, clinical psychologists, palliative care specialists, an exercise physiologist, and a patient-care coordinator [33]. With the goal of minimizing risk of developing CPSP before surgery and long-term opioid use postoperatively, the TPS identifies high-risk pain patients during the pre-admission clinic visit [36]. Patients that may have been missed during their pre-admission visit could still be referred to the service if they met any criteria including pre-surgical chronic pain, history of drug abuse, severe post-surgical pain, high post-surgical opioid consumption (> 90 MEQ/day), and emotional distress [30]. These patients are followed up as inpatients to avoid delays in discharge due to uncontrolled pain. Once discharged, early follow-up is arranged (within 1–2 weeks), such that they continue to be seen on an outpatient basis with the eventual goal of transitioning them back to their primary care physician.
EARLY SUCCESS OF THE TRANSITIONAL PAIN SERVICE
The TPS program based out of the Toronto Gene-ral Hospital has been operational for five years and there have been a number of notable successes. Between May 2014 and July 2017, the TPS analyzed 304 surgical patients after discharge from hospital. Of these, 251 had full data available for review and analysis. These patients were classified into two groups: opioid-naïve (0 MEQ/day) (45%) or opioid-experienced (> 0 MEQ/day) (55%).
Among opioid naïve patients, MEQ consumed were decreased from 106.7 post-surgery to 37.3 at the final TPS visit (6.6 months after surgery), translating to a 65% decrease [33]. Within the opioid-experienced group, 140.5 MEQ at discharge were decreased to 78.3 at the final TPS visit (5.2 months after surgery), resulting in a 44% decrease [33]. More importantly, among opioid-naïve patients, 44.5% of patients were no longer taking an opioid at 6 months after discharge, and 25.6% of opioid-experienced patients were completely weaned off opioids at 6 months. Among those still taking opioids, 45.5% and 55.4% of opioid-naïve and opioid-experienced patients saw some level of reduction in opioid consumption, respectively [33].
The above data demonstrate that the TPS has been effective in safely weaning patients from opioids in the postoperative period, in both opioidnaïve and experienced patients. At the same time, TPS involvement in the post-discharge period has also led to reductions in reported pain. In the thoracic surgery population, a cohort of patients followed by the TPS in the postoperative period reported a reduced level of pain and a faster trajectory towards “milder” pain as compared to a cohort without TPS involvement [37].
Within the TPS framework, importance is placed on the optimization of non-opioid adjuncts to enhance pain management and facilitate weaning of opioids. Such adjuncts include medical cannabis, which is rapidly gaining adoption in chronic pain management in Canada. For example, TPS was successfully able to facilitate opioid weaning in a post-liver transplant patient [38]. In this case, a patient requiring 30 mg of oral hydromorphone per day was successfully weaned to 6 mg per day through the introduction of medical cannabis as a pain adjunct. This not only produced significant reductions in opioid needs, but also improved self-reported functional status.
The success of the Toronto TPS experience has led to similar initiatives elsewhere. A similar model to the TPS has been adopted successfully in the United Kingdom [39], where non-postoperative patients with chronic pain participated in an interdisciplinary pain management program. This program involved psychologists, physiotherapists, nurses and physicians in a comprehensive program similar to that of the TPS. They also relied heavily on ACT-based theory. Overall, participants saw a reduction in the total oral morphine equivalents dose, as well as the number of medication classes used. This interdisciplinary approach to pain management has seen success, further establishing the need, importance, and feasibility of this model.
FUTURE PLANS FOR THE TRANSITIONAL PAIN SERVICE
The implementation of the TPS program, and its early success, have allowed for planning to expand the scope of the program, and increase accessibility. These plans include engaging community and family physicians to facilitate referral of non-Toronto General Hospital surgical patients to the program [30]. In addition, there are plans to develop rehabilitation programs incorporating yoga and mindfulness to improve physical and psychological wellbeing. With the increasing availability and adoption of mobile technology, the TPS has recently engaged patients in self-reporting of pain scores and functioning through mobile apps. This facilitates ongoing tracking and documentation, and allows patients to participate in their pain management more actively. Finally, the future of pain management must involve providing support and expertise to allow for the development of similar programs at other hospitals, allowing easier access for patients in need.
RECOMMENDATIONS FOR INITIATING SIMILAR SERVICE IN OTHER HOSPITALS
There are three main considerations when considering the implementation of a program similar to the TPS. These are institutional support, buy-in from surgeons, and comprehensive multidisciplinary involvement.
In terms of incentivizing institutional support, the potential cost and patient care benefits of a TPS program are clear. It is estimated that CPSP costs the system on average $41,000-43,000 per patient annually (U.S. System) [40]. Our own analysis based on the experience at Toronto General Hospital has revealed significant potential benefits within the Canadian healthcare system [25]. With 4000 patients receiving major surgery at the Toronto General Hospital annually, new cases and worsening cases of post-surgery chronic pain make up 5% and 12.5%, respectively, of all major procedures. With a gross underestimated annual cost of $5000 annually, it can be estimated that these patients access over $2.5 million of health care services annually from the system. These funds could be used to service other areas within patient pain management, or other areas of healthcare. Outside of the financial benefits, the availability of services such as the TPS can also alleviate strain on other departments within the hospital. This includes potentially reducing emergency department visits for patients needing assistance with pain management or prescriptions, which is a common cause for emergency department visits [41]. This allows the emergency departments and acute care clinics to focus on managing patients with acute or emergent medical issues. The combined potential to improve both efficiency in health care spending, as well as overall patient care, should provide sufficient incentive to most institutions.
The engagement of surgeons for support of a TPS program is equally important, and also straightforward. Pain management and medication prescribing is a common topic of both postoperative clinic visits with surgeons, and telephone calls to surgeon offices [42]. While issues of this nature are important, they often occupy a significant portion of busy surgical clinic visits and potentially distract from discussion of surgical or prognostic details. Additionally, some pain complaints are complex, and beyond the scope of what a surgeon may feel comfortable addressing. A TPS program would allow faster access to pain management specialists and allow surgeons to focus on important areas where they have more expertise.
Finally, the success of any program similar to TPS hinges upon the need for multidisciplinary involvement. The availability of non-pharmacologic interventions including physiotherapy and behavioral interventions grounded in Acceptance and Commitment Therapy are imperative to the improvement in pain management and reduction in opioids. Preliminary outcomes from the Toronto General Hospital showed that patients who participated in ACT saw a 17% mean pain score reduction vs 8% reduction for those patients who did not participate [43]. ACT patients also saw a significant reduction in pain interference scores – one’s pain experience leading to anxiety or depression – compared to those who did not participate in ACT.
CONCLUSIONS
In summary, this review provided a brief history of the opioid epidemic and defined the concept of chronic post-surgical pain. Furthermore, the rationale for the TPS was described (i.e. created in order to address patient’s pain management needs through a multidisciplinary model) and preliminary data regarding early successes were reported. Finally, recommendations were given to hospitals that are aiming to implement a similar pain management program, with insight into hurdles that may be faced and potential means to mitigate them.