Introduction
Arterial ischemic stroke (AIS) is a rare, heterogeneous disease in which multiple risk factors have an impact on its development. The pathology and the impact of potential risk factors of childhood AIS are still unclear. The most commonly reported states predisposing to AIS in childhood are central nervous system (CNS) arteriopathies, traumas, infections, cardiac diseases, metabolic diseases and mitochondrial diseases, with approximately half of all cases being cryptogenic [1, 2]. In adults, atherosclerosis is the main cause of most strokes.
The clinical manifestation of AIS depends on the localization of CNS lesions. Circulation disorders in the intracranial artery and central cerebral artery are characterized by paresis, less paralysis, central paresis or paralysis of the facial nerve and aphasia. Sometimes the focal symptoms are accompanied by headaches, rarely by seizure. If, however, disorders are related to the posterior vertebrae of cerebral vascularization, the features of the cerebellar syndrome will predominate among the clinical symptoms. The location of vascular lesions in patients with AIS has also been a contributor to the commonly used classification of ischemic stroke [3].
A history of pediatric AIS is related to various neurological consequences such as motor impairment (the most common is hemiparesis), seizures or intellectual delay [1, 2]. Therefore, the problem of post-stroke deficits is noteworthy because of their impact on daily activity as well as the costs of medical care and rehabilitation. Ischemic stroke occurring in the perinatal period influences cognitive and behavioral deficits, which become more evident during the subsequent years of life [4]. Earlier studies have reported that post-stroke cognitive problems may be associated with stroke subtypes [4], or epilepsy, since the presence of seizures limits plasticity of the developing brain [5].
In the present study we analyzed the frequencies of risk factors, stroke symptoms and post-stroke consequences in Polish pediatric patients depending on stroke subtype.
Material and methods
Study group
The study was retrospective and based on 77 patients recruited in the Department of Pediatric Neurology at the Medical University of Silesia in Katowice (Poland) between 2002 and 2012. The inclusion criteria for the study group were: first arterial ischemic stroke; age at onset up to 18 years, white Caucasian. The exclusion criterion from the study group was death after the first AIS or during the recurrence of stroke.
Neurological and cardiology examinations
A detailed neurological examination of all patients in the acute phase of the disease and at follow-up was performed. Children also underwent cardiology (electrocardiography, ultrasound cardiography) and neuroimaging studies with computed tomography and/or magnetic resonance imaging.
Focal cerebral arteriopathy was defined according to Amlie-Lefond et al. criteria [6] as focal stenosis with abnormalities of the arterial wall not attributed to specific diagnoses such as moyamoya, arterial dissection, vasculitis, or post-varicella angiopathy.
The following heart diseases were diagnosed in the recruited patients: common ventricle, sponge cardiomyopathy, mitral valve defect, Ebstein’s anomaly and congenital rubella syndrome.
Chronic diseases or congenital malformations occurring in the study of patients included: arterial hypertension, moyamoya disease, Hirschsprung’s disease, nephrotic syndrome, Schimke syndrome, thrombocytopenia, type I diabetes, MELAS syndrome (mitochondrial encephalomyopathy, lactic acidosis, and stroke-like episodes), epilepsy (with the onset in the first year of life), adaptive disorders and recurrent respiratory infections.
Clinical manifestation of arterial ischemic stroke
Arterial ischemic stroke was identified by the World Health Organization’s International Classification of Diseases. The stroke subtypes were evaluated according to the Oxfordshire classification into partial anterior circulation infarct (PACI), posterior circulation infarct (POCI), lacunar anterior circulation infarct (LACI) and total anterior circulation infarct (TACI) [7].
In the present group of patients the following most frequently observed neurological symptoms of AIS were analyzed: hemiplegia, hemiparesis, nerve VII paralysis, disturbances of consciousness, headache and dizziness, aphasia and convulsions.
Neurological consequences of arterial ischemic stroke at follow-up
At the follow-up, the following consequences of AIS were analyzed: hemiparesis, seizures, aphasia and other movement disorders. Post-stroke seizures in pediatric patients with AIS were defined according to Beslow et al. [8] as: early/acute symptomatic seizures (up to 7 days after stroke onset), remote symptomatic seizures (beyond 7 days from stroke presentation), and at least two recurrent, not provoked, seizures occurring after the acute phase of stroke. In the present analysis the term ‘seizures’ was used in relation to all patients who experienced any type of seizures, early or late, as well as ‘normal outcome’ for outcomes without neurological deterioration.
Statistical analysis
Statistical analysis was performed using Statistica 12.0 software (STATSOFT; Statistica, Tulsa, OK, USA). Mean values (M) and standard deviations (SD) were estimated for continuous variables, or absolute numbers (n) and relative numbers (%) of occurrence of items of categorical variables. The Kruskal-Wallis H test was used to compare continuous variables between four types of AIS. The Mann-Whitney U test was used to compare continuous variables between the children with a normal outcome and the children with at least one neurological consequence. The stochastic independence χ2 test with Yates’s correction was used to compare categorical variables between stroke subtypes as well as between the normal outcome group and the children with at least one consequence. Odds ratio (OR) estimates were used for occurrence of at least one neurological consequence vs. no neurological consequence, with age as the independent variable. In order to interpret it, an OR was calculated and expressed as a percentage. The significance level was set at p ≤ 0.05 in all of the statistical tests.
Results
Risk factors of arterial ischemic stroke depending on stroke subtypes
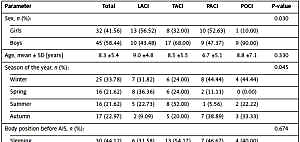
We analyzed 77 children with AIS, of whom 25 (32%) had TACI subtype, 23 (30%) had LACI, 19 (25%) had PACI and 10 (13%) had POCI stroke subtype. Table I shows basic characteristics of the study group as well as risk factors associated with AIS in children. In the study group, most children with POCI (90%) and TACI (68%) stroke subtypes were male, whereas LACI and PACI occurred in girls and boys with similar frequency. The age of examined children with AIS ranged from 1 to 18 years old (8.3 ±5.4 on average), and age did not significantly differ between stroke subtypes at onset.
Table I
Factors associated with AIS in children and stroke subtypes
There were significant differences between types of AIS and seasons of the year at which the AIS occurred. The TACI subtype appeared most frequently in the summer, LACI during winter or spring, and PACI in the winter or autumn.
When we analyzed the position of the patient’s body at onset of AIS we found that 44% of children were sleeping, 32% were walking, 21% were sitting and 3% were standing. However, body position was not associated with stroke subtypes.
Most children with AIS obtained 10 Apgar points after birth (38%), 24% were scored with 9 points, 28% with 8 points, 9% with 7 points, and one child with 6 points. Almost all the examined children with AIS were born at the full term of pregnancy (from the 38th to 40th week of pregnancy), there being only 1 child who was born in the 35th week of pregnancy. In 19% of children with AIS there was a history of complicated pregnancy and 6% presented with low birth weight (below 2500 g). Stroke subtype was not associated with Apgar points, birth weight, fetal age, or complications of pregnancy. In 13% of children, there was a history of upper respiratory infection before AIS; however, its occurrence did not differentiate stroke subgroups.
On the other hand, we observed that focal cerebral arteriopathy (FCA) and chronic diseases were associated with type of AIS. Focal cerebral arteriopathy was present in most children with TACI (80%) and PACI (79%) subtypes, in 50% children with POCI, and was less common in children with LACI (35%). Chronic diseases coexisted in 43% of children with PACI, 36% with TACI, 22% with POCI, and were least common in children with LACI (one child).
Characteristics of the clinical presentations of arterial ischemic stroke depending on stroke subtype
Table II presents the number of infarct foci and locations of morphological changes as well as the frequency of AIS symptoms in the analyzed pediatric patients. We observed that one infarct focus was present in almost all children with LACI and PACI subtypes, in 71% of children with TACI and in half of patients with POCI. Two infarct foci were present in one third of patients with POCI subtype, in every 8 patients with TACI, in 1 patient with PACI and in one with LACI. Three or more infarct foci occurred in almost 17% of patients with TACI and in 1 child with POCI (11%). We also found that the number of locations of morphological changes significantly differentiated stroke subtypes. A single location of the morphological changes, i.e. the temporal lobe, was present in most children with LACI (86%) and POCI (80%), and in 61% of children with PACI. By contrast, in most children with TACI subtype several locations of morphological changes were observed (61%).
Table II
Number of infarct foci, location of morphological changes and AIS symptoms in the study group
In the analysis of AIS symptoms we found that hemiplegia was significantly associated with stroke subtype. It was most frequent in children with TACI stroke (33%) while none of the children with POCI had hemiplegia because of the focus localization. Symptoms other than hemiplegia did not differentiate any of the analyzed subgroups of patients. Hemiparesis was present in 85% of the total study group, nerve VII paralysis in 57%, disturbances of consciousness in 72%, headache and dizziness in 32%, aphasia in 37% and convulsions in 16%. Other symptoms were significantly more common in children with POCI stroke subtype (90%) compared to children with other subtypes.
Neurological deficits following pediatric arterial ischemic stroke
Neurological outcomes following pediatric AIS are shown in Table III. The number, type and coexisting outcomes were related to stroke subtype. A normal outcome was observed in 43% children with LACI, 30% with POCI, 16% with PACI and only in one child with TACI. Most children with LACI and PACI had one deficit (52% and 63%, respectively), while most children with TACI had two or more consequences. When types of consequence were analyzed, we observed hemiparesis in almost all children with TACI stroke, less frequently in children with PACI (two thirds), and in almost 50% of children with LACI or POCI.
Table III
Neurological consequences following pediatric AIS
Seizures (early and late seizures were analyzed jointly) were present in 40% of patients with TACI, in 30% of patients with POCI, in 26% of patients with PACI, and in 9% of patients with LACI stroke. Aphasia occurred in every fifth child with TACI or POCI, in one child with PACI, and in none of the children with LACI. When we analyzed coexisting consequences, we observed that a normal outcome or only hemiparesis was present with equal frequency, each in 43% of patients with LACI stroke, while in children with TACI the most frequent was hemiparesis (40%) or hemiparesis and seizures (32%), and in almost 50% of children with PACI only hemiparesis was found.
Additionally, we divided the entire study group into two subgroups: a subgroup with a normal outcome (n = 17) and a subgroup with at least one post-stroke consequence (n = 60). Table IV shows the comparison between the two subgroups. In the case of age at AIS onset, a significant difference was observed between subgroups. Children with a normal outcome were older at the time of brain ischemia. The OR value for age equaled 0.894 and shows that the odds of outcomes after ischemic stroke decreases by an average of 10.6% if the child is 1 year older at the time of stroke occurrence. In the case of birth weight, as well as the number of morphological changes, the localizations were different between the subgroups and bordered on significance. In every child with hemiplegia as a stroke symptom, it evolved into hemiparesis at follow-up.
Table IV
Comparisons between cases with a normal outcome and those with at least one neurological consequence
Discussion
Arterial ischemic stroke in childhood is a rare, multifactorial disease; thus its occurrence, development and consequences may be the result of the simultaneous presence of several related risk factors.
Previously, focal cerebral arteriopathy has been recognized as a frequent risk factor for childhood stroke. Focal cerebral arteriopathy was also observed to be related to post-stroke seizures [8, 9]. Amlie-Lefond et al. observed seizures in 23% of arteriopathy patients [6]. In Polish patients, FCA was found to be a strong predictor of post-stroke epilepsy (p = 0.010, HR = 10.50) [9]. In the present study, 13% of patients developed post-stroke epilepsy while other available data demonstrated even greater frequencies [10, 11], which appear to be related to geographical differences. In the study of deVeber et al. [3] seizures were extremely high in neonates (88%), and 37% of older children also experienced seizures. We also observed that the presence of any type of seizures after stroke (early or late) may depend on stroke subtype (the difference was close to significance, p = 0.077). Seizures were most common in patients with TACI and least common in children with LACI stroke. In most children with AIS, FCA was transient although at least some of the factors contributing to the onset of FCA were constant and unmodified, such as genetically determined factors associated with coagulation. Presumably both, environmental factors, such as upper respiratory tract infections and some genetic risk factors, may precede the onset of AIS in children.
Due to the heterogeneity of AIS, different sets of genetic and non-genetic risk factors may exist and be related to the etiology and neurological burden of the disease in different populations. Among these factors, thrombophilia, hyperhomocysteinemia, dyslipidemia or MTHFR, FII, or FV gene polymorphisms can be distinguished [12–14]. Previous multicenter genetic studies, of 80 children suffering from AIS, showed that post-stroke epilepsy may be correlated with carrier state of the G allele of the –174G>C polymorphism within the IL-6 gene [15]. On the other hand, our earlier pilot data also provided some indication of an association between other candidate polymorphisms (561A>C in the E-selectin gene or insertion-deletion polymorphism of the ACE gene) and a serious post-stroke state with noticeable consequences [16, 17]. However, both of the studies were based on a small number of patients and have not been confirmed in a larger group.
In the present study, we observed that AIS is more common in boys than girls (58% vs. 42%) and these frequencies are in agreement with other findings [18]. Westmacott et al. [19] demonstrated that particularly boys with unilateral neonatal stroke are at increased risk for cognitive deficits during the school years. A possible explanation for this may be the immaturity of the male brain at birth compared to the female one, with its particular susceptibility to damage caused by stroke [19]. At present, we found significant differences in sex distribution between the stroke subtypes (p = 0.030). Most children with POCI and TACI stroke subtypes were male. Interestingly, there might be some genetic determinants of AIS development dependent on gender. Earlier, the 677C>T polymorphism within the MTHFR gene was suggested to significantly increase the risk of AIS in boys [20].
Arterial ischemic stroke in our patients appeared most frequently in winter (34%), then autumn (23%) and in summer and spring with the same frequency (22%). In adults, low ambient temperature of the day of stroke onset increased the risk of ischemic stroke as well as high-speed wind, especially in older patients [21]. Higher frequency of stroke in winter or autumn may also be linked with the greater frequency of infections, which are common in the colder seasons of the year. It was demonstrated that the risk of pediatric AIS is increased by infections [22]. Previously, it was observed that major infections (e.g. bacterial meningitis or sepsis) have been associated with stroke in children. However, minor infections, mostly upper respiratory tract infections, occur more frequently in childhood [23, 24]. Infections were demonstrated to be related to stroke in adults since they were observed in 10% of adults with AIS [23]. Minor infections may affect the prothrombotic state, which is caused by inflammatory mediators [25] and may lead to hypercoagulability. In most studies, frequency of the prothrombotic state ranged from 20% to 50% in children [26]. A significantly elevated level of procalcitonin was suggested to be a sensitive and specific predictor of systemic bacterial infection in children with chronic kidney disease [27]. In our study, in almost 13% of children respiratory infections preceded ischemic stroke. In 60% of them, AIS appeared in the autumn-winter period.
The factors related to pregnancy and childbirth did not differ between the analyzed stroke subgroups, and 38% of children obtained 10 Apgar points after birth. The available data indicate that most children suffering from AIS usually come from well-conceived, full-term pregnancy and the newborns usually have good Apgar scores and show normal somatic development [28].
We demonstrated that number, type and coexisting consequences were related to stroke subtype. In our study, 22% of analyzed children were characterized by a normal outcome without deterioration, while a single deficit was present in 49% of patients and two, three or four consequences were present in 29% of patients. Sreenan et al. [29] observed a normal outcome in 32% of children with perinatal stroke, while single or multiple deficits were demonstrated in 17% and 50%, respectively. We observed significant differences in the presence of hemiparesis in the analyzed subgroups of patients. Hemiparesis affected almost all children with TACI stroke. Additionally, in all children with hemiplegia the evolution to hemiparesis was observed. In children without hemiplegia in the acute phase of the disease, evolution of the grade of hemiparesis was observed during follow-up. When we compared children with a normal outcome and children with some neurological deficits we found that children with neurological deficits were significantly younger at the time of brain ischemia compared to children with a normal outcome. This finding is in agreement with previously published data based on patients with perinatal stroke, in which the young age of the patient at onset was significantly related to a poor neurological outcome [30]. In this study, the authors also observed infarction in the right middle cerebral artery territory as well as fever at presentation as risk factors [30].
Previously, some data concerning factors influencing stroke outcomes in adults were published. Worsened clinical outcome and increased risk of in-hospital death were associated with hyperglycemia on admission in the group of patients with ischemic and hemorrhagic stroke [31]. However, it was also observed that prior transient ischemic attacks (TIAs) may have a neuroprotective effect on the subsequent ischemic stroke, as well as neurological outcomes [32].
The limitation of our study is the rather low number of patients, which is mainly due to the fact that they were recruited from a single medical center. Additionally, we did not consider the inherited issues of cardiovascular diseases or abnormalities in lipid or fibrinogen levels in the analyzed patients, whereas those problems are of great importance in regard to the risk of AIS [33–36].
In conclusion, our study shows that the presence and number of neurological outcomes depend on stroke subtypes. There is also a significant relationship between the presence of post-stroke deficits and age at onset. Children with at least one deficit were younger at the time of AIS occurring than children with a normal outcome. However, further studies are needed to confirm and identify these patterns and associations.