Introduction
Lipoprotein(a) (Lp[a]) is a low-density lipoprotein (LDL)-like particle that binds to the protein apoprotein(a) (this protein has a structure homologous to plasminogen) [1]. Due to the unique features of Lp(a) (i.e. thrombogenic and atherogenic properties) [2], Lp(a) is recognized as a risk factor of cardiovascular disease (CVD) [3–6]. Of interest, the blood Lp(a) levels are largely determined by genetic factors [7].
Familial hypercholesterolemia (FH) is an autosomal dominant genetic disease [8]. The main causative genes are related to LDL clearance by the LDL receptor (LDLR), leading to high blood levels of LDL cholesterol (LDL-C) [9]. FH is recognized as a risk factor of CVD [10].
A combinatory relationship between LDL-C and Lp(a) for CVD has been previously suggested, although the previous studies did not specifically examine FH subjects [11, 12]. A few studies also show that FH subjects with high Lp(a) levels had an increased risk of CVD, but these studies were conducted in somewhat specific settings (e.g. in the coronary care unit at a single hospital [13] or during a cascade test screening for family members of FH subjects [14]). As such, the relationship of Lp(a) with CVD outcomes has yet to be characterized in a population limited to FH.
Whether or not the impact of Lp(a) on CVD can be modulated in FH, a condition of high LDL-C levels, is still of interest. In the current study, we therefore conducted a meta-analysis of available studies to determine the relationship of Lp(a) with CVD outcomes in FH subjects.
Material and methods
Candidate articles for this study were searched using PubMed up to April 25, 2020 using the following keywords: “familial hypercholesterolemia” AND (“lipoprotein(a) (Title/Abstract)” AND “cardiovascular disease (Title/Abstract)” AND risk NOT review (Publication type)). Cardiovascular disease was defined as coronary heart disease and stroke (cerebrovascular disease). Studies that focused on the relationship of Lp(a) with CVD in FH were included. Studies written in a language other than English and for a non-adult population were excluded. The title and abstract of all studies identified through the search were screened. In addition, the reference lists of the retrieved articles were searched for further identification of relevant studies.
Statistical analysis
The studies reporting the CVD risk by Lp(a), as odds ratio (OR) and risk/hazard ratio (HR) with 95% confidence interval (CI), were finally collected. When multivariate estimates were reported, the maximally adjusted estimates were extracted. The OR in cross-sectional studies and the RR and HR in cohort studies were used to assess the risk of CVD by high Lp(a) levels compared to low Lp(a) levels. Random-effects meta-analyses for CVD outcomes were performed in the Review Manager software program (RevMan ver. 5.3) using the generic inverse variance method. The results were expressed as the OR and HR with 95% CIs, and heterogeneity was assessed using I 2 statistics. In addition, when the OR and HR were considered approximately equivalent as relative risks, an integrated meta-analysis was performed.
Results
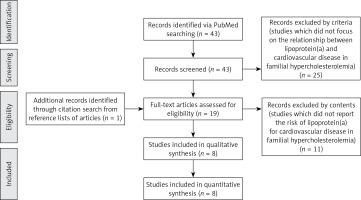
Figure 1 shows the flow for selecting articles that reported the relationship of Lp(a) with CVD outcomes. Of the 43 initially identified articles, 25 that did not focus on this relationship were excluded. After reviewing the full text and the reference lists of all retrieved articles, eight articles [15–22] met the criteria. The 8 studies (8,378 participants and 1,458 CVD outcomes) included 2 cross-sectional studies and 6 cohort studies.
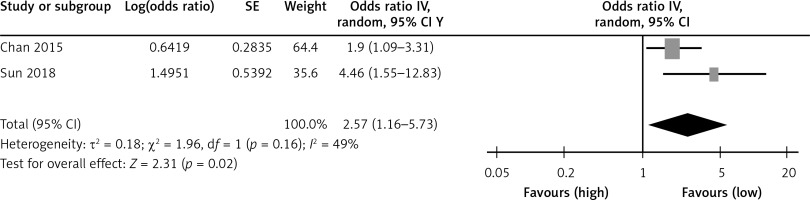
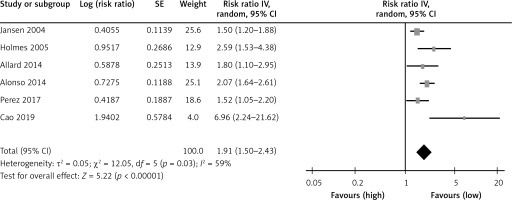
Table I shows a summary of the association of Lp(a) with CVD in the two cross-sectional studies. As shown in Figure 2, the two studies found high Lp(a) levels to be positively associated with CVD compared to low Lp(a) levels (OR = 2.57; 95% CI: 1.16–5.73; I 2 = 49%). A summary of the prediction of Lp(a) on CVD is shown in Table II in six cohort studies. High Lp(a) levels predicted CVD compared to low Lp(a) levels with HR (1.91; 95% CI: 1.50–2.43; I 2 = 59%; Figure 3). In addition, the integrated relative risk of all studies was 1.97 (95% CI: 1.57–2.46; I 2 = 53%).
Table I
The association of Lp(a) with CVD by odds ratio in two cross-sectional studies
| Authors [ref. no.] | Year | Subjects (n) CVD/non-CVD | Age [years] CVD/non-CVD | Confounders | Cut-off level of Lp(a) | OR (95% CI) |
|---|---|---|---|---|---|---|
| Chan [15] | 2015 | 64/326 | 57/43 | – | 50 mg/dl | 1.90 (1.09–3.31) |
| Sun [16] | 2018 | 61/87 | 49/45 | Age, sex, smoking, hypertension, diabetes mellitus, body mass index, high-density lipoprotein cholesterol, statin, family history of CVD | 60 mg/dl | 4.46 (1.55–12.83) |
Table II
The prediction of Lp(a) for CVD by risk ratio in three cohort studies
| Authors [ref. no.] | Year | Subjects (number) CVD/non-CVD | Age [years] CVD/non-CVD | Confounders | Cut-off level of Lp(a) | HR (95% CI) |
|---|---|---|---|---|---|---|
| Jansen [17] | 2004 | 782/1618 | 50.4/42.0 | Sex, smoking, hypertension, diabetes mellitus, body mass index, high-density lipoprotein cholesterol, triglyceride, homocysteine | 30 mg/dl | 1.50 (1.20–1.79) |
| Holmes [18] | 2005 | 61/327 | 53.0 | Sex, smoking, hypertension, total cholesterol/high-density lipoprotein cholesterol, low-density lipoprotein cholesterol | 56 mg/dl | 2.59 (1.53–4.39) |
| Allard [19] | 2014 | 74/221 | 67.7/58.0 | Sex, smoking, diabetes mellitus, body mass index, high-density lipoprotein cholesterol, low-density lipoprotein cholesterol, triglyceride, family history of premature CVD | 60 mg/dl | 1.8 (1.1–2.9) |
| Alonso [20] | 2014 | 247/1713 | 44.4 | – | 50 mg/dl | 2.07 (1.64–2.61) |
| Pérez De Isla [21] | 2015 | 122/185 | 44.5 | Age, sex, smoking, hypertension, diabetes mellitus, waist circumference, body mass index, high-density lipoprotein cholesterol, low-density lipoprotein cholesterol, triglyceride, history of CVD, statin | 50 mg/dl | 1.52 (1.05–2.21) |
| Cao [22] | 2019 | 35/358 | 50.3/48.5 | triglyceride, total cholesterol | 52.0 mg/dl | 6.96 (2.24–9.32) |
Discussion
The current meta-analysis reviewed the relationship of Lp(a) with CVD outcomes in FH subjects. The findings from the meta-analysis showed that a high Lp(a) level remained a risk factor for CVD among FH subjects.
This meta-analysis revealed the risk of CVD as an OR of 2.57 and HR of 1.91, as well as an integrated relative risk of 1.97. In an earlier meta-analysis of the relationship of Lp(a) with CVD in a general population, an RR of 1.57 for CVD was demonstrated when the group with the high Lp(a) level was compared to that with the low level [5]. In a cohort study of hypercholesterolemic subjects with LDL-C of ≥ 3.1 mmol/l (121 mg/dl), the HR of 1.64 for CVD was demonstrated when the group with a high Lp(a) level (≥ 44 mg/dl) was compared to that with a low level [11]. The risk for CVD with Lp(a) among FH subjects, as observed in the current meta-analysis, seemed to be similar to or slightly high relative to the risks described for these data [5, 11].
The threshold of Lp(a) causing CVD may be debatable [3, 11]. While there is an opinion that a linear relationship without a threshold exists between the Lp(a) levels and CVD outcomes among general populations [23], an Lp(a) of 30–50 mg/dl is often used as the cut-off level for CVD [24, 25]. The European guideline states that the desired Lp(a) level for the prevention of CVD is < 50 mg/dl for general populations and CVD subjects [26]. As most studies included in the current meta-analysis used cut-off levels of 50 or 60 mg/dl (as shown in Tables I and II), this might induce a somewhat high risk in FH subjects relative to general populations and generic/common hypercholesterolemic subjects [5, 11].
While LDL-C is a main target for the management in FH subjects, the current study findings suggest the need for attention to Lp(a) levels during management. Statins (HMG-CoA reductase inhibitors) are representative lipid-lowering drugs in hypercholesterolemic subjects, including FH subjects [27–29]. Therapies involving statins can increase or decrease Lp(a) levels [27–29]. Whether or not the statin effects influence the relative risk for CVD with Lp(a) in FH subjects is unclear. Recently, proprotein convertase subtilisin/kexin type 9 (PCSK9) inhibitors have been used to treat high LDL-C levels in conditions such as FH, and these inhibitors reduce not only LDL-C but also Lp(a) levels [30]. The effect of reduction of Lp(a) by intervention with PCSK9 inhibitors on the relationship between Lp(a) and CVD should be explored in the future.
Several limitations associated with the current study warrant mention. First, there were not many studies included for the purpose of this topic. Second, confounding factors (e.g. obesity, hypertension) in the adjusted analysis were not fully unified across the studies.
In conclusion, the current meta-analysis revealed that FH subjects with high Lp(a) levels could have a high risk of CVD, and in addition to LDL-C, attention should be paid to Lp(a) levels in FH subjects.