While most of the doubts regarding bupivacaine safety have been dispelled – both through extensive research into its pharmacokinetics and the spread of ultrasound navigation – some concerns regarding specific scenarios persist. When the delivered dose approaches the recommended threshold, absorption and further metabolism of local anesthetics become a concern. Even when injected properly, large doses of bupivacaine may result in potentially toxic plasma concentrations [1–3]. Furthermore, an inadvertent intravascular injection would increase the risk of severe local anesthetic systemic toxicity (LAST). In recent years, this problem has become particularly relevant in Ukrainian hospitals due to an unprecedent incidence of exposure to potentially toxic doses of bupivacaine. Soldiers and civilians wounded during the Russian invasion often undergo intensive pain management during primary stabilization, medical evacuation and numerous surgical revisions. Such patients require either repeated high-dose perineural injections or a continuous infusion via a catheter, commonly supplemented by sedation or general anesthesia (GA) [4]. All these factors alone or in combination significantly increase the risk of LAST due to resorption of the local anesthetic or delayed elimination.
The LAST syndrome is based on two key adverse effects of local anesthetics – neurotoxicity and cardio-toxicity [5]. The neurotoxic element usually manifests earliest as seizures, which could not be detected under a combination of GA and regional anesthesia due to neuromuscular blockade and deep sedation. Characteristic cardiac arrhythmias [6] occur when higher plasma concentrations of bupivacaine are reached. When regional and GA are combined, electrocardiogram (ECG) remains the main tool for diagnosing clinically significant LAST.
The choice of a hypnotic with a better safety profile for combined anesthesia is controversial [7]. Propofol is expected to have a few protective pro-perties when administered in parallel with bupivacaine since it contains lipid emulsion [8]. However, previous research has demonstrated that it is inferior to concentrated lipid emulsions for resuscitation in LAST cases [9]. Also, propofol itself possesses cardiodepressive and vasoplegic properties, potentially contributing to cardiovascular collapse.
The purpose of the present study was to compare the effects of two hypnotic agents (propofol and sevoflurane) on the systemic toxicity of determined plasma concentrations of bupivacaine.
MATERIAL AND METHODS
This study was approved by the Bioethics Committee of the Bogomolets National University on 26.09.2022 (protocol #160). It is part of a research program funded by the Ministry of Health of Ukraine: “Optimization of the algorithm for performing regional anesthesia techniques in patients with trauma to reduce the risks of systemic toxicity of local anesthetics and nerve injuries”.
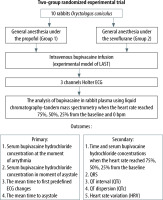
This was a 2-group randomized experimental study. In this study ten domestic rabbits (Oryctolagus cuniculus domesticus) were divided into 2 groups depending on the type of anesthetic used for GA: propofol and sevoflurane. Intramuscular sedation was delivered initially. After tracheal intubation, mechanical ventilation was initiated. In the first group, propofol (1%) was initiated at the rate of 1.8 mg kg–1 min–1. In the second group, sevoflurane was delivered at the end tidal concentration of 4 vol%. At 30 min after tracheal intubation, heart rate and QRS duration were recorded as “baseline” and intravenous (i.v.) bupivacaine (0.25% solution) at the rate of 1.0 mg kg–1 min–1 was initiated. Blood samples were obtained when predefined ECG changes were observed and when the heart rate (HR) reached 75%, 50%, 25% of the baseline and 0 bpm (15 s after the final systole). The analysis of bupivacaine in rabbit plasma was carried out using liquid chromatography-tandem mass spectrometry on a Shimadzu LCMS-8050 instrument according to the method developed at the Bogomolets National Medical University.
During the study, the serum concentration of bupivacaine hydrochloride was compared at the time of previously identified rhythm disturbances and at the time of asystole. Furthermore, the time to onset of arrhythmia and asystole at identical doses of bupivacaine hydrochloride was recorded.
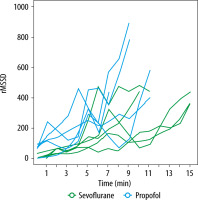
In addition, intracardiac conduction parameters were evaluated: HR, QRS, QT interval (QTi), QT dispersion (QTc), HR variation (HRV). HR variability was estimated as the root mean square of successive R-R interval differences (rMSSD).
Anesthesia protocol
All animals were fasted for 6 hours before the procedure. Access to water was not restricted. In both groups, rabbits were sedated via an intramuscular injection of xylazine 10 mg kg–1, tile-tamine 7.5 mg kg–1 and zolazepam 7.5 mg kg–1. An additional 1 mg kg–1 of tiletamine and zolazepam were administered intravenously in 30-second intervals until pedal withdrawal and ear pinch reflexes were absent. Rabbits were intubated with a 3.0 mm cuffed endotracheal tube, followed by an i.v. bolus of rocuronium 0.6 mg kg–1. Fentanyl infusion was initiated at a rate of 0.005 mg kg–1 h–1. For mechanical lung ventilation a Getinge Flow-C ventilator was used with tidal volume adjusted to maintain normocapnia (35–45 mmHg) at a stable respiratory rate (30 breaths per minute), FiO2 of 0.3, positive end-expiratory pressure of 1 mmH2O (0.01 kPa), I : E ratio of 1 : 1.5. A 22-gauge venous catheter was then placed in the femoral vein to be used exclusively for blood sampling. It was flushed with 100 IU mL–1 unfractionated heparin solution. First 0.2 mL of every obtained blood sample was discarded to minimize dilution. In the first group, animals were anesthetized with propofol 1% intravenous infusion at the rate of 1.8 mg kg–1 min–1. In the second group, 4.0% sevoflurane was delivered. Rocuronium 0.3 mg kg–1 injections were repeated every 20 min in both groups. After the experiment, all rabbits were euthanized using thiopental 200 mg kg–1 i.v. injection.
Vital parameter monitoring
Vital parameter monitoring included ECG monitoring initiated before induction of anesthesia and Holter monitoring to assess ECG patterns occurring during intravenous bupivacaine infusion. An analysis of pulse oximetry indicators was carried out, and SpO2 was kept at a level not lower than 95%. After tracheal intubation, pressure-controlled ventilation was initiated, aiming to maintain etCO2 within the range of 35–45 mmHg (4.6–6 kPa). Rectal thermometry was performed every 5 min in the period after tracheal intubation and before bupivacaine infusion, with target temperature ranging from 38.0°C to 40.0°C. Blood pressure monitoring was not available.
Laboratory analysis
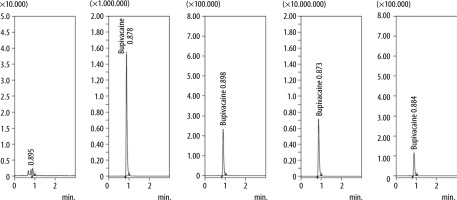
The blood samples were centrifuged at 4000 rpm for 15 min and frozen before transport to the laboratory. The serum bupivacaine hydrochloride concentrations were measured using liquid chromatography-tandem mass spectrometry on the Shimadzu LCMS-8050 [4]. The method is based on the extraction of bupivacaine hydrochloride from samples of rabbit plasma with methanol (simultaneously with coagulation of proteins), purification of extracts by centrifugation, and subsequent quantitative determination of bupivacaine by high-performance liquid chromatography with mass spectrometric detection in the multiple reaction monitoring mode. The bupivacaine hydrochloride in rabbit plasma was determined in the range 0.001–150 μg mL–1 using two methods of sample preparation, which provide determination at the range 0.001–0.600 μg mL–1 (method No. 1) and the range 0.5–150 μg mL–1 (method No. 2). The limit of quantitative determination (LOQ) of bupivacaine hydrochloride in rabbit plasma is 0.001 μg mL–1. The limit of detection (LOD) of bupivacaine hydrochloride in rabbit plasma is 0.0005 μg mL–1 (Figure 1).
ECG analysis
Obtained Holter ECG data were processed using the official software provided by the manufacturer (3 Channels ECG Holter System_TF V5.5.2.5). The first 15 complexes in any given minute were used to calculate mean values of RR, QRS, QT durations, and HR variability (rMSSD formula), rounded down. HR and QTc values were derived from these mean measurements. QTc interval was calculated using Bazett’s formula. Criteria for “ECG changes” used for blood sampling included: QRS widening > 20% from the base value, R' or S' wave appearance, or arrhythmia, whichever occurred first. Arrhythmia was defined as variability of R-R interval length exceeding 50 ms in 3 consecutive QRS complexes or the occurrence of 5 premature ventricular contractions in each minute. For real-time analysis, a single-lead strip was conti-nuously printed at 100 mm s-1 up to the moment of detection of predefined ECG changes.
Statistical analysis
The Shapiro-Wilk test was used to test continuous variables for normality of distribution. Student’s t-test was applied to normally distributed values to check for statistically significant differences. For non-parametric data, a two-sided Mann-Whitney U test was used when comparing two groups. Results were considered statistically significant at P ≤ 0.05.
Outcomes
Primary outcomes were time and serum bupivacaine hydrochloride concentrations at the onset of arrythmia and at the occurrence of asystole.
Time and serum bupivacaine hydrochloride concentrations when the HR reached 75%, 50%, 25% of the baseline, QRS, QTi, QTc, and HRV were the secondary outcomes (Figures 2–4).
RESULTS
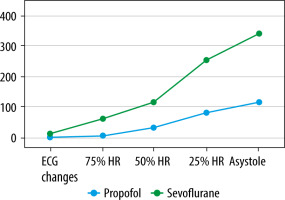
The two groups did not significantly differ in age or weight (Table 1). The mean time to first predefined ECG changes was 131 ± 25.02 s, 95% CI: 100–162 for the propofol group and 223 ± 34.11 s, 95% CI: 180–265 for the sevoflurane group (P = 0.001). No significant difference was observed in time to reach 75% and 50% HR relative to the baseline, while the 25% HR time was shorter for the propofol group (480 ± 117 s, 95% CI: 335–625 vs. 673 ± 146 s, 95% CI: 491–854, P = 0.05). Time to asystole was shorter in the propofol group (110.7 ± 22.22 s, 95% CI: 49.02–172.4 vs. 226.6 ± 98.61 s, 95% CI: 104.1–349, P = 0.047).
TABLE 1
Rabbits’ mean age and weight
Mean serum bupivacaine hydrochloride concentration was lower for the propofol group at the moment of occurrence of the first ECG changes (2.542 ± 1.415 mg mL–1, 95% CI: 0.785–4.299 vs. 6.997 ± 2.197 mg mL–1, 95% CI: 4.27–9.725) and asystole (110.7 ± 22.22 mg mL–1, 95% CI: 49.02–172.4 vs. 226.6 ± 98.61 mg mL–1, 95% CI: 104.1–349).
The mean HR and QT interval did not significantly differ at any calculated point. QRS duration was longer for the sevoflurane group at 4, 5, and 6 min. HRV (rMSSD) was higher in the propofol group at 2 and 11 min, and QTc was higher in the sevoflurane group at 6 and 7 min. Mean serum bupivacaine hydrochloride concentration was lower for the propofol group at the moment of occurrence of the first ECG changes (2.542 ± 1.415 mg mL–1, 95% CI: 0.785–4.299 vs. 6.997 ± 2.197 mg mL–1, 95% CI: 4.27–9.725) and asystole (110.7 ± 22.22 mg mL–1, 95% CI: 49.02–172.4 vs. 226.6 ± 98.61 mg mL–1, 95% CI: 104.1–349) (Tables 2–4).
TABLE 2
Mean time (seconds) to reach target points
[i] ECG-changes – time of detection of first ECG changes (bradycardia, QRS prolongation, QT-prolongation, Brugada-like pattern), 75% HR – time when the heart rate reached 75% of the baseline, 50% HR – time when the heart rate reached 50% of the baseline, 25% HR – time when the heart rate reached 25% of the baseline, asystole – time from the beginning of bupivacaine hydrochloride intravenous infusion to the moment of asystole.
TABLE 3
Mean serum bupivacaine hydrochloride concentrations (mg mL–1) at target points
[i] Standard deviation is provided for normally distributed data, median and minimum-maximum range are specified otherwise (variable marked with *).
[ii] ECG changes – serum bupivacaine hydrochloride concentration when first ECG changes (bradycardia, QRS prolongation, QT-prolongation, Brugada-like pattern) were detected, 75% HR – serum bupivacaine hydrochloride concentration when the heart rate reached 75% of the baseline, 50% HR – serum bupivacaine hydrochloride concentration when the heart rate reached 50% of the baseline, 25% HR – serum bupivacaine hydrochloride concentration when the heart rate reached 25% of the baseline, asystole – serum bupivacaine hydrochloride concentration at the moment of asystole.
TABLE 4
Registered ECG parameters and quantity of valid measurements for a specific minute, mean value for each group (P – propofol, S – sevoflurane) and P-value for Student’s t-test or Mann-Whitney U test (indicated with *)
DISCUSSION
A series of experiments demonstrated that when bupivacaine hydrochloride is administered intravenously during GA using sevoflurane, the time to the occurrence of the first ECG changes and to the moment of asystole is significantly longer compared to propofol-based anesthesia. The serum bupivacaine concentration required for the deve-lopment of arrhythmia and cardiac arrest is also significantly higher in the sevoflurane group. It can be assumed that sevoflurane has a better safety profile compared to propofol during anesthesia combined with regional blocks.
There is a lack of studies comparing the effects of sevoflurane and propofol on the development of LAST. Ohmura et al. [10] performed a similar study in 1999, during which they employed more rigorous cardiovascular monitoring but did not measure bupivacaine plasma concentrations, leaving the question of drug distribution in hemodynamically deteriorating rats open. Our results regarding time to dysrhythmia and time to asystole are similar, although we are unable to contribute any data regarding mean arterial pressure and seizure activity.
Propofol is expected to have a few protective properties when administered in parallel with bupivacaine since it contains lipid emulsion. However, previous research has demonstrated that it is inferior to concentrated lipid emulsions for resuscitation in LAST cases [11–13]. Also, propofol itself possesses cardiodepressive and vasoplegic properties, which has been mentioned before.
Several mechanisms are proposed to explain the greater resistance of sevoflurane-anesthetized rabbits to the arrhythmogenic effects of bupivacaine. The simplest hypothesis is based on the comparatively less prominent hypotensive action, resulting in better myocardial perfusion. Experiments on isolated perfused rat hearts demonstrated a lack of potentiation of the bupivacaine cardiotoxic effect when sevoflurane is added to the system [8]. Sevoflurane preconditioning also appears to diminish the severity of the ischemic reperfusion injury [14, 15]. This effect has not been examined sufficiently in the context of bupivacaine toxicity during combined regional and GA, making it a possible direction for further research. Currently, available data suggest that bupivacaine does not affect the cardioprotective effect of sevoflurane preconditioning [9].
It is debatable whether the later occurrence of ECG changes improves patient safety. In this study, the delay coincided with delayed complete circulatory arrest. On the other hand, early identification of excessive local anesthetic delivery would allow for its earlier termination and treatment initiation. Despite decades of research, the choice of a hypnotic agent for combined anesthesia remains a controversial matter.
It is important to note that the risk of LAST is further augmented by extensive pharmacotherapy. This is particularly important for wounded soldiers who have a long recovery process. The usual prolonged treatment of mine-blast trauma with antibiotics, analgesics, and antidepressants often diverges from best practices due to the chaotic nature of wartime medical evacuation [16]. Bupivacaine metabolism may be affected through several mechanisms that are not routinely considered. Selective serotonin reuptake inhibitors (notably escitalopram) inhibit CYP3A4, which may cause accumulation of bupivacaine in plasma [17, 18]. Drug-induced toxic hepatitis is often diagnosed in these patients and should also be considered.
FUTURE RESEARCH
Plasma concentration thresholds of clinically apparent neurotoxicity have previously been identified in healthy volunteers undergoing intravenous infusion of bupivacaine [19]. Research into delayed effects of critical doses of extravascularly administered bupivacaine in humans under GA cannot be conducted without endangering their well-being beyond the ethically permissible boundaries. We intend to conduct further observational studies that incorporate ECG monitoring in human patients who receive large daily doses of local anesthetics to detect minimal consistent changes while measuring corresponding serum local anesthetic concentrations and examining clinical outcomes.
STUDY LIMITATIONS
No blood pressure monitoring or echocardio-graphy was performed, making it impossible to objectively determine the point when the cardiovascular collapse occurred. Lack of EEG also means that the clinically important impact of the anesthetic agents on the central nervous system was not assessed. The results of the study only reflect the effects of slow intravenous infusion and can only be applied to long-term bupivacaine exposure scenarios to a limited extent.
CONCLUSIONS
At similar infusion rates, the cardiotoxic action of bupivacaine may be detected on an ECG earlier when propofol is used as an anesthetic agent compared with sevoflurane, as it develops at lower plasma drug concentrations in rabbits. A complete circulatory arrest occurs later and at higher plasma concentrations when sevoflurane anesthesia is conducted, which suggests that it may be a safer agent when exposure to high concentrations of bupivacaine is suspected or anticipated. Continuous ECG analysis may be useful in the early detection of bupivacaine cardiac toxicity when propofol or sevoflurane is being used.