Cardiac surgery is commonly associated with high blood loss and the need for allogeneic red blood cell (RBC) transfusion. In addition, 20–30% of cardiac surgical patients suffer from anaemia [1]. Brouwers et al. [2] analysed > 11,000 cardiac surgical patients and revealed a transfusion rate of > 50%. Karkouti et al. [3] analysed > 9000 patients undergoing cardiac surgery and revealed that severe intraoperative blood loss is associated with an 8.1-fold higher mortality rate. Studies indicate that allogeneic RBC transfusions lead to an increased number of adverse outcome events like postoperative low output failure [4].
In order to minimise the risks associated with peri- operative RBC transfusion, patient blood mana-ge-ment (PBM) has evolved. PBM is an approach to reduce anaemia (pillar 1), minimise iatrogenic blood loss (pillar 2), and optimise patient-specific tolerance of anaemia (pillar 3), in order to maintain the patient’s own blood volume. In particular, the second pillar comprises blood conservation strategies to manage perioperative bleeding including the use of cell salvage (CS) [5]. CS collects blood by aspiration from the operative field to produce autologous RBC for re-transfusion [6]. A meta-analysis based on 47 trials, involving 21 cardiac surgical trials, demonstrated that CS is efficacious in reducing the overall need for allogeneic RBC transfusion in cardiac surgical patients by 29% [7].
These studies, however, have several limitations. Sample sizes of trials were low. Study populations included in the in meta-analysis ranged between 24 and 541, with a majority (19 out of 21 studies [90.5%]) of studies analysing patient populations with less than 100 patients [7]. Trials with low numbers of patients are important to understand the impact of the intervention; however, they might often be underpowered to detect clinical differences of the intervention. Furthermore, the majority of the studies did not include patients undergoing aortic surgery, focusing instead on coronary artery bypass grafting (CABG) and cardiac valve surgery only. Aortic procedures are also known to account for high intraoperative blood loss, with a transfusion rate of up to 88% [2].
The implementation of PBM measures, including CS, is an integral part at the University Hospital Frankfurt [8–10]. Here, we evaluate the benefit of CS in a large number of patients undergoing elective cardiac surgery at a designated PBM centre. We hypothesised that the use of CS is associated with a decreased proportion of patients exposed to RBC transfusion as an individual measure in a comprehensive PBM program.
METHODS
Patients
This observational study is based on a multicentre observational epidemiological trial focusing on the implementation of PBM in surgical patients (Trial Registration: ClinicalTrials.gov, NCT02147795) [11]. The ethics committee of the University Hospital Frankfurt (Ref. 318/17) approved the study protocol and waived the requirement for written informed consent by patients.
A preoperative anaemia walk-in clinic was established for diagnosis and treatment of iron deficiency anaemia in patients undergoing major surgery (e.g. cardiac surgery), irrespective of underlying risk factors or type of cardiac surgery. All patients screened by the anaemia walk-in clinic are supplemented with iron/folic acid/vitamin B12 if required [12]. Physiological conditions of haemostasis (body temperature > 36°C; pH > 7.2; Ca2+ > 1.1 mmoL L-1), use of tranexamic acid, minimally invasive techniques, and point-of-care diagnostics or management of coagulopathy are ensured during surgery among other interventions. All of the above-mentioned measures are applied to all surgical patients. To improve further the outcome for cardiac surgical patients, extracorporeal volumes were reduced and retrograde autologous priming was implemented. Also, compliance to guideline-based transfusion triggers was implemented [10, 12].
We analysed medical records of patients scheduled for elective cardiac surgery requiring cardiopulmonary bypass (CPB) between November 2015 and October 2018. According to the use of CS, patients were divided into a CS group (patients received CS and autologous re-transfusion at the physician’s discretion) and a control group (no CS). CS was used intraoperatively. Anaemia was defined according to the World Health Organisation (WHO), with haemoglobin (Hb) concentrations < 12 g dL-1 in women and < 13 g dL-1 in men.
Inclusion criteria
Patients undergoing major surgery (e.g. cardiac surgery) with a ≥ 10% risk for RBC transfusion were assessed for preoperative anaemia management, irrespective of underlying risk factors or type of cardiac surgery. All adult patients (age ≥ 18 years) undergoing elective cardiac surgery and requiring CPB were included in the analysis. Coronary artery bypass grafting surgery, valve surgery or combined CABG and valve surgery, aortic surgery as well as left ventricular assist device (LVAD) implantation, and surgical interventions of structural heart changes were included.
Exclusion criteria
To avoid information bias, we excluded patients with incomplete medical records (no documented blood loss or re-transfused CS volume) and massive bleeding. Massive bleeding was considered as a severe bleeding (> 1000 mL) after sternal closure in the intensive care unit combined with minimally invasive re-intervention or surgical re-exploration. Because these cases represent emergency situations, only wide ranges and inaccurate amounts of blood loss were documented.
Cell salvage
Cardiac surgery is known for high intraoperative blood loss. According to the local blood conservation strategy guidelines, blood-collecting containers from the CS system are used if expected blood loss exceeds 500 mL. Therefore, in cardiac surgery, blood from the surgical field is collected in special blood-collecting containers from the CS system. These are only used for surgical suction before and after CPB. CS is available in every cardiac surgery, and intraoperative blood loss is calculated by the attending anaesthesio-logist. If blood loss exceeds 500 mL, the decision to set up the entire CS apparatus (C.A.T.S Plus Fresenius Kabi, Bad Homburg, Germany) to produce autologous blood for re-transfusion is dependent on preoperative risk stratification. Risk factors that influence the decision for the use of CS are comorbidities, redo cardiac surgery, Hb value, preoperative antiplatelet or anticoagulation therapy, CPB time (> 120 min), and aortic surgery. Also, the German transfusion guidelines are taken into consideration. Therefore, re-transfusion was conducted in patients with cardiovascular risk factors and Hb < 8 g dL-1 and risk for anaemic hypoxia [24]. In cases of blood loss < 500 mL, collected surgical blood was not processed by CS apparatus. An anaesthetic technician operated the CS device. If CS was used, all recovered blood, irrespective of recovered blood volume, was re-transfused. Salvaged blood volume was converted into units by dividing by 300.
Coagulation management and transfusion practice
All patients received standard anaesthetic induction with fentanyl, propofol, and rocuronium. Tranexamic acid 1 g was administered intravenously after anaesthesia induction. Before CPB, heparin 500 U kg-1 was given to achieve an activated clotting time (ACT) > 450 s, with additional boluses of 5.000–10.000 U if ACT decreased below 450 s at any time. All patients underwent CPB, blood cardioplegia was used, and blood gases were recorded every 30 minutes throughout the CPB. Tranexamic acid was given at a continuous rate of 400 mg/h during the CPB. Transfusion of RBC units was in accordance with the German transfusion guidelines. Briefly, RBC transfusion is recommended in asymptomatic patients with Hb < 6 g dL-1, in patients with cardiovascular risk factors with Hb between 6 and 8 g dL-1, or in patients with clinical symptoms of anaemic hypoxia [13]. During and after cardiac surgery, allogeneic RBC units were transfused when Hb was < 8 g dL-1, targeting Hb ≥ 8 g dL-1. After CPB, prota-mine was given at a matching dose of preoperative heparin (1 U protamine for 1000 U heparin). If ACT still exceeded baseline ACT, additional 20–50 U kg-1 protamine was administered. For further coagulation management, basic physiological conditions had to be ensured for haemostasis (body temperature > 36°C; pH > 7.2; Ca2+ > 1.1 mmol L-1). If the absolute number of platelets was lower than 100 nL-1, one unit of platelet was transfused plus 0.4 µg kg-1 desmopressin. In the case of platelet dysfunction due to a long CPB time (> 120 min) or platelet intake influencing medication (e.g. acetylsalicylic acid), rotational thromboelastometry and platelet function tests were done after CPB (body temperature > 36°C). Fibrinogen and prothrombin complex were given in accordance with the results from the rotational thromboelastometry.
Primary endpoints
Primary endpoints were the number of patients exposed to allogeneic RBC transfusions and the number of RBC units transfused per patient.
Secondary endpoints
Secondary endpoints were the amount of re-transfused blood from CS and postoperative Hb levels. Patient’s Hb levels were followed up by analysis of medical records until day 0 (day of surgery) and day 1.
Statistical analysis
Descriptive statistical methods (mean ± standard deviation [SD], median, interquartile range [IQR] [25%; 75%] or ratio) were used to analyse the data. To assess normality of continuous variables the Shapiro-Wilk test and graphic method using histograms were used (Suppl Figure 1). Non-normally distributed data (preoperative Hb-level, age, BMI, blood loss, RBC transfusion rate, and RBC units) were compared with the Wilcoxon-Mann-Whitney test. Categorical variables were compared with χ2 test or Fisher’s exact test. Correlation between transfusion of RBC units and blood loss was computed using Spearman’s rho because the data were not normally distributed. Patients were assigned to their groups by documented use of CS in medical records. Cell salvage was used in patients with high intraoperative blood loss (> 500 mL), therefore adjustment for blood loss diminished the sample size and was statistically invalid. Therefore, any amount of blood loss in both groups was included and considered for analysis. Consequently, simple linear regression analysis was performed using single data or mean values to study the association between intraoperative blood loss and allogeneic RBC transfusion. Linear regression slopes were compared using the F-test. In addition, propensity score matching was used for adjusted analysis including the variables age, gender, BMI, preoperative Hb level, preope-rative anaemia, ASA, and type of cardiac surgery as potential confounders. Matching (nearest neighbour approach with exact matching of ASA, gender, and preoperative anaemia) was performed with R (R foundation for statistical computing, Vienna, Austria) using the package MatchIt version 3.0.2. A P-value < 0.05 was considered to be statistically significant. The absolute standardised difference was also calculated to measure the effect size between both groups. Statistical analysis was performed using Graphpad Prism® (Version 8.0, Graphpad Software, San Diego).
RESULTS
Study population
Between November 2015 and October 2018 more than 2000 cardiac surgeries were performed at our hospital, of which 856 patients were screened in the anaemia walk-in clinic. A total of 152 patients were excluded from the analysis because of incomplete medical records and severe bleeding (Figure 1). In total, 704 patients were analysed; CS was used in 338 patients during surgery (CS group), and 366 patients underwent surgery without CS (control group). After performing propensity score matching the sample size was 618 patients (CS group, n = 309; control group, n = 309).
Adherence to the German transfusion guidelines and the hospital’s coagulation management by every attending anaesthesiologist was ensured by standardised processes during cardiac surgery. No difference was found between both groups in terms of age, BMI, and gender distribution. The preoperative Hb level of patients in the CS group was lower (13.4 [12.2–14.6] g dL-1) compared to the control group (13.9 [12.7–14.7] g dL-1) (P = 0.019). Accordingly, the prevalence of preoperative anaemia differed significantly between the groups: 33.1% (n = 112) in the CS group vs. 23.8% (n = 87) in the control group (P = 0.007) (Table 1). Overall, 331 patients underwent CABG surgery, 255 valve surgery, 55 combined CABG and valve surgery, 34 aortic surgery, 14 removal of myxoma, thrombus, or thymus, and 15 other types of cardiac surgery (Table 2).
TABLE 1
Demographic data of included patients
TABLE 2
Different types of cardiac surgery included in the study
[i] Data represented as count (percentage). CABG – coronary artery bypass grafting, LVAD – left ventricular assist device, ASD – atrial septum defect, PFO – patent foramen ovale, CS – cell salvage, Abs. Std. Diff. – absolute standardised difference. Matched comparison: patient data after propensity score matching.
Association between cell salvage and allogeneic red blood cell transfusion
Mean blood loss was significantly higher (1800 ± 935 mL) in the CS group compared to the control group (566 ± 371 mL; P < 0.0001), suggesting a higher rate of more complex surgical procedures in the CS group. The mean ± SD amount of re-transfused autologous blood from CS was 459 ± 297 mL. Amounts of re-transfused blood ranged from a mini-mum of 50 mL to a maximum of 1661 mL. Intraope-ratively, patients in the CS group received 1.1 ± 1.5 (median 0 [0–2]) allogeneic RBC units whereas patients in the control group received 0.5 ± 1.0 (median 0 [0–1]) units (P < 0.0001). Overall, 152 patients (45%) were exposed to allogeneic RBC transfusions in the CS group and 93 patients (25%) in the control group during surgery (P < 0.0001). Intraoperatively, the numbers of allogeneic RBC units per exposed patient were 2.4 ± 1.4 (median 2 [1–3]) in the CS group and 1.9 ± 1.2 (median 2 [1–2]) in the control group.
At the day of surgery but postoperatively (= day 0), use of allogeneic RBC units was similar between the CS group (0.1 ± 0.4 units) and the control group (0.1 ± 0.3 units; P = 0.28) (median 0 [0–0] vs. 0 [0–0] units). Overall, 23 patients (7%) received allogeneic RBC transfusions in the CS group and 18 patients (5%) in the control group (P = 0.33). The total number of allogeneic RBC units per exposed patient was 1.6 ± 0.8 (median 1 [1–2]) in the CS group and 1.4 ± 0.8 (median 1 [1–2]) in the control group (P = 0.46) (Table 3).
TABLE 3
Blood loss and RBC units before and after matching
Postoperative haemoglobin concentration
Regarding follow-up time, the postoperative mean Hb concentration was similar between the CS and the control group for day 0 (CS group: 9.6 ± 1.2 g dL-1 vs. control group: 9.6 ± 1.3 g dL-1; P = 0.62) and day 1 (CS group: 9.4 ± 1.1 g dL-1 vs. control group: 9.5 ± 1.3 g dL-1; P = 0.97) (Table 4).
TABLE 4
Perioperative Hb level before and after matching
Comparison of allogeneic red blood cell units between the cell salvage group and control group
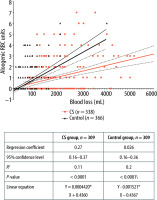
A regression analysis was performed to compare the need for RBC transfusion between patients in the CS group and the control group. Considering the range of minimal and maximum blood loss (400–6700 mL), CS was associated with a reduced use of allogeneic RBC transfusion by 5% (600 mL blood loss) to 55% (6700 mL blood loss). Overall, the allogeneic RBC transfusion rate was significantly associated with higher surgical blood loss in both groups (P < 0.001). However, comparing patients of both groups with similar blood loss, the use of allogeneic RBC units was higher in patients of the control group compared to the CS group (P < 0.001) (Figure 2). For further analysis, linear regression with matched data after propensity score matching was conducted. For intraoperative exemplary amounts of blood loss of 1000, 2000, and 3000 mL in regression analysis of adjusted data, analysis showed a significant association of (P < 0.001) higher mean use of allogeneic RBC units in the control group than in the CS group (1000 mL: 1.0 vs. 0.6 RBC units; 2000 mL: 2.2 vs. 1.1 RBC units; and 3000 mL: 3.4 vs. 1.6 RBC units). Thus, CS was significantly associated with a reduced number of allogeneic RBCs by 40% for 1000 mL, 49% for 2000 mL, and 52% for 3000 mL blood loss, compared to patients without CS. Overall, the analysis revealed that patients in the CS group required significantly fewer allogeneic RBC transfusions intraoperatively compared to the control patients (P < 0.001) (Figure 3).
FIGURE 2
Regression analysis of intraoperative blood loss and red blood cell (RBC) units transfused. Scatter plot to evaluate the correlation between the amount of blood loss and number of intraoperative transfused allogeneic RBC units. Values are shown for individual patients of the cell salvage (CS) group (red) and control group (black). Results of linear regression analysis are displayed with 95% confidence interval for both groups (dotted line). The difference between the slopes is significant (P = 0.0002). The regression coefficient provides information about the strength and direction of a relationship, R2 – coefficient of determination. R2 evaluates the scatter of the data points around the fitted regression line; it indicates the quality of the independent variables (blood loss) and can explain the variance of the dependent variables (RBC units). The R2 value is always between 0 (no goodness of fit) and 1 (perfect goodness of fit). Y – RBC units transfused, X – blood loss in mL
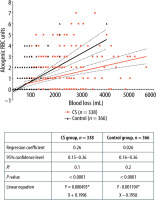
FIGURE 3
Regression analysis of intraoperative blood loss and red blood cell (RBC) units transfused with adjusted data. Scatter plot to evaluate the correlation between the amount of blood loss and number of intraoperative transfused allogeneic RBC units. Patients’ data after propensity score matching was used. Values are shown for individual patients of the cell salvage (CS) group (red) and control group (black). Results of linear regression analysis are displayed with 95% confidence interval for both groups (dotted line). The difference between the slopes is significant (P = 0.0004). Regression coefficient provides information about strength and direction of a relationship, R2 – coefficient of determination. R2 evaluates the scatter of the data points around the fitted regression line; it indicates the quality of the independent variables (blood loss) and can explain the variance of the dependent variables (RBC units). R2 value is always between 0 (no goodness of Fit) and –1 (perfect goodness of Fit). Y – RBC units transfused, X – blood loss in mL
DISCUSSION
The intraoperative use of CS in major surgery remains important [14] and is implemented in guidelines for peri-operative blood conservation [15]. This might be associated with the increasing number of implemented PBM programs. For example, Althoff et al. previously demonstrated that a multimodal PBM program addressing all 3 main pillars is effective in improving the clinical outcome in surgical patients. Analysis of 17 studies including > 235,000 patients revealed that PBM significantly reduced the RBC transfusion rate by 39% [16]. However, based on their study design, and because PBM is a bundle of care, the authors were not able to reveal which PBM measure was the most effective. Several PBM measures have been successfully implemented at the University Hospital Frankfurt [8, 10]. Although CS is a valuable technique to recover surgical blood loss, the cost effectiveness of CS and the reduction of adverse outcomes by reducing allogeneic RBC transfusion requirements is still under debate. Although the current evidence underlines the beneficial effects of CS [17–22], many national authorities may decline any approval of a prospective randomised comparison of groups with and without blood conservation strategies due to ethical issues. Here, we performed a sub-study of a multicentre observational epidemiological trial and evaluated the association between CS that was at the individual physician’s discretion and RBC transfusion rate in patients undergoing cardiac surgery at our PBM centre. A total of 856 patients underwent elective cardiac surgery. Blood loss was 3.2 times higher in the CS group compared to the control group and accounted for the increased intraoperative transfusion rate of allogeneic blood in the CS group (45%) compared to the control group (25%). Here, it should be noted that selection bias may occur because CS is especially used in patients with high intraoperative blood loss. Surprisingly, patients in the CS group received only 0.5 RBCs more (1.0 ± 1.5 units) compared to the control group (0.5 ± 1.0 units), although blood loss in the CS group was on average 1000 mL greater. Regression analysis was performed to evaluate the association between CS and RBC transfusion. Cell salvage was significantly associated with a reduced number of allogeneic RBCs by 52% for 3000 mL, 49% for 2000 mL, and 40% for 1000 mL blood loss. Xie et al. also found that CS was associated with a significant reduction of more than 3 RBC units in 150 patients undergoing cardiac surgery with a similar amount of intraoperative blood loss (CS group 2 ± 2.8 vs. control group 5.4 ± 3.3 RBC units; P < 0.0001) [23]. Wang et al. [24] conducted a meta-analysis including 31 trials, of which 19 displayed sample sizes below 50 undergoing cardiac surgery. Overall, CS was associated with a decreased rate of 0.4 RBC units per patient. Similarly, 321 patients undergoing off-pump CABG were analysed by Wang et al. [25] to evaluate the safety, efficacy, and cost-effectiveness of CS with different amounts of bleeding. Overall, the authors demonstrated that the efficacy of CS was dependent on the amount of surgical blood loss. In particular, when the amount of surgical blood loss was > 600 mL, CS significantly reduced the demand for allogeneic blood transfusions. Only a few studies have suggested that there are no beneficial effects from CS on the proportion of patients exposed to allogeneic blood transfusion or the number of transfused RBC units [26–29]. Lastly, there are also some risks that may be associated with CS, such as red cell injury leading to increased potassium, non-immune haemolysis, or air embolus [30].
Our study has some limitations. First, we used an observational design, and confounding factors cannot be excluded. Secondly, multiple types of surgeries with different times of CPB were included. This variety of data may account for the overall difference in intraoperative blood loss and transfused RBC units per patient in the 2 study groups. The results of a higher blood loss and RBC transfusion rate in the CS group suggests a higher rate of more complex cases in the CS group. This reflects everyday clinical practice, and therefore generalisability of our results is ensured. However, the higher blood loss in the CS group might also imply a selection bias, which may be unavoidable, due to the indication of CS in surgeries with high intraoperative blood loss based on international guidelines. Given the possible bias of linear regression analysis, this should be considered when comparing patients from the CS group and from the control group. Also, intraoperative blood loss was a subjective estimation made by the attending anaesthesiologist. Defining blood loss in cardiac surgery is challenging because blood loss is determined by the measurement of blood in collecting containers, visual assessment of blood in surgical towels, and recycling of CPB blood remaining in the circuit. This calculation is an uncertain but universally established practice. In addition, it would be interesting to assess additional risk factors like EuroScore, comorbidities, or redo cardiac surgical procedures. Also, a comparison of CPB and aortic cross-clamp would be of great interest and should be considered in future studies. Besides RBC transfusion, a holistic PBM program also includes non-RBC products (e.g. platelets or fresh frozen plasma) and antifibrinolytic drugs [16]. In this sub-study, however, we focused on transfused allogeneic RBC units only, because CS produces only RBC for autologous re-transfusion. Regarding the conversion of cell-salvaged blood to RBC units, it is noteworthy to mention that differences in haematocrit occur between cell-salvaged blood and allogeneic blood. For future studies, mean haematocrit achieved by CS should be measured, because 300 mL of cell-salvaged blood may differ from 300 mL of allogeneic blood. Also, the evaluation of coagulation management (e.g. use of prothrombin complex concentrate, fibrinogen, or results from rotational thromboelastometry) and transfusion of non-RBC products should be considered. Lastly, the term cell salvage could be renamed as cell recovery in further studies.
CONCLUSIONS
Here, we analysed > 800 patients undergoing cardiac surgery including CABG surgery, cardiac valve surgery, aortic surgery, and left ventricular assist device implantation with and without CS at a PBM centre. Overall, CS was associated with a significantly reduced number of allogeneic RBC transfusions, depending on intraoperative blood loss, by 52% for 3000 mL, 49% for 2000 mL, and 40% for 1000 mL blood loss compared to patients without CS. In conclusion, our data suggest that CS is associated with a decreased proportion of patients exposed to allogeneic RBC transfusions as an individual measure in a comprehensive PBM program.