Introduction
Psoriasis epidermal hyperproliferation is a chronic, multi-systemic inflammatory disease characterised by abnormal keratinocyte differentiation, T-lymphocyte infiltration, and increased cytokine expression [1]. It is present on the skin in 6–8% of patients arriving at the Dermatology Clinic [2]. The etiology of this disease is unknown; however, various factors such as genetics, trauma, infections, drugs, and smoking are associated with the aetiopathogenesis of the disease. Chronic inflammation and oxidative stress cause an increase in the prevalence of cardiovascular disease [3]. Atherosclerosis, hypertension, and valvular involvement are frequently encountered in patients with psoriasis. However, a recent study by Ahlehoff et al. determined that psoriasis was related to an increased risk of atrial fibrillation (AF) [4]. Other studies have also reported that secondary amyloid deposition was present in the conduction system, and correspondingly, atrial conduction disorders were frequently observed [5, 6]. The AF is the most common arrhythmia, and it is associated with an increased risk of stroke, heart failure, coronary artery disease, and cardiovascular mortality. Total atrial conduction time (TACT) is detected using echocardiography, and is prolonged in cases of discontinuous and heterogeneous propagation of sinus impulses [7]. Weijs et al. determined that prolonged TACT and AF were related [8]. Left atrial global longitudinal strain (LAGLS), which is detected using two-dimensional speckle tracking echocardiography (2D-STE), is a new tool currently under investigation for the prediction of AF. The determination of the relationship between TACT and LAGLS will contribute to the ability to predict AF, which is an arrhythmia with a high rate of morbidity and mortality in psoriasis patients, and is frequently seen in the clinic.
There are currently no studies exploring the role of LAGLS and TACT in the induction of subclinical electrophysiological changes in psoriasis patients; therefore, the goal of the current study was to investigate the relationship between TACT and LAGLS in patients with psoriasis.
Material and methods
This study was performed in accordance with the Helsinki Declaration after consent was obtained from the hospital Ethics Committee. All patients were informed prior to beginning the study, and their approval was granted.
Study population
Forty patients who were over the age of 18 years, had been diagnosed with psoriasis vulgaris for a minimum of 3 years via biopsy, and admitted to the Dermatology Department, Faculty of Medicine, Recep Tayyip Erdoğan University, Rize, Turkey (group 1), were selected as study participants. Additionally, 40 age- and sex-matched healthy individuals were included in this study as the control group (group 2). Patients and control subjects with a history of past or concurrent diseases such as hypertension (HT), diabetes mellitus, coronary artery disease (CAD), lung diseases and/or pulmonary HT, valvular heart diseases, liver or kidney diseases, collagen vascular diseases, rhythms other than sinus, any cardiovascular drug use, abnormal thyroid function, or serum electrolyte values were excluded from the study. Also, obese subjects (BMI ≥ 30 kg/m2) were excluded from the study. All subjects gave informed consent, and the local Ethics Committee approved the study. Patients with coexisting psoriatic arthritis (diagnosed by a rheumatologist) were excluded due to the possible adverse effects on heart rhythm. The age, gender, age of onset, duration of the disease, and drug history of each patient were recorded. Height, weight, body mass index (BMI), and waist circumference were also assessed in all patients and biochemical variables such as fasting glucose levels and a lipid panel were also analysed and Holter electrocardiography (ECG) was recorded.
Study design
A total of 80 patients, of whom 40 were psoriasis patients and the remaining 40 healthy controls, were included in the study. The group of psoriasis patients was named group 1, and the control group was named group 2.
Standard echocardiographic evaluation
The echocardiographic assessment was performed on patients during clinical remission in the routine follow-up period. Also, none of the patients used any drugs that affected the heart during the echocardiographic assessment. Transthoracic echocardiographic assessments were carried out using the S5–1 probe (5–1 MHz) transducer with a Philips iE33 Medical ultrasound Systems device. The patients were assessed in a left lateral decubitus position after a 5-minute rest period. Firstly, the pericardium, valve morphology, and wall motions were assessed using M mode and two-dimensional echocardiography. The LV ejection fraction (LVEF) and left atrium diameter (LA) were calculated using the parasternal long axis. In pulse wave (PW) Doppler examination of LV filling using an apical four-chamber view, the Doppler sample volume in parallel to the LV long axis and at the mitral annulus were measured and averaged. During the assessment, the early diastolic flow velocity (E), the late diastolic flow velocity (A), E/Em, the deceleration time (DT), and the isovolumetric relaxation time (IVRT) were recorded. In the apical four-chamber view, a 5-mm-width PWD sample volume was performed by placement on the point of intersection of the posterior wall and the mitral annulus. By ensuring that the sample volume was parallel with the wall axis, peak early (Em) diastolic flow velocity was recorded. LA volumes were determined using the biplane ‘area-length’ method from apical four- and two-chamber images, and their average was calculated. The LA maximum volume (LAVmax) was measured when the mitral valve was completely opened, and LA minimum volume (LAVmin) was measured when the mitral valve was completely closed, and the following parameters were calculated using these measurements: LAVreservoir (ml): LAVmax-LAVmin. All measurements were repeated during three consecutive heartbeats and averaged. The American Society of Echocardiography’s standards were used as a base for all the measurements [9].
2D speckle tracking echocardiography
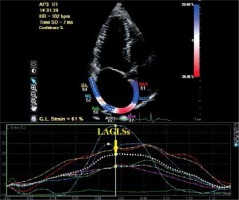
The apical four-chamber and two-chamber grey scale views were stored digitally. The records were then processed by TDI data and analysed off-line using dedicated software (QLAB, Philips). The global strain and the strain rate were calculated by averaging the values measured in 15 atrial segments. The frame rate was 50–80 frame/s. The global peak systolic LA myocardium strain during the left ventricular systole (LAGLSs), global peak systolic positive LA myocardial strain rate during left ventricular systole (LAGLSRs), global peak negative LA myocardial strain rate during early ventricular diastole (LAGLSRe), and global peak negative LA myocardial strain rate (LAGLSRa) during late ventricular diastole were obtained (Figure 1).
Atrial tissue Doppler imaging
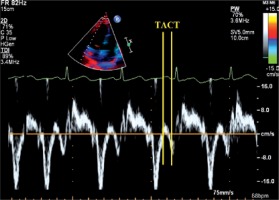
Total atrial conduction time – the time from initiation of the electrocardiographic P-wave (lead II) to the local lateral left atrial activation time [10] – was determined using transthoracic echocardiography. This was measured by placing the sample volume in the left atrial basal portion and then measuring the time between initiation of the electrocardiographic P-wave and the peak velocity, using tissue velocity imaging (Figure 2).
Evaluation of patients’ disease activity
The diagnosis of psoriasis vulgaris was performed by a dermatologist and/or was based on a description of characteristic lesions. The clinical severity was determined according to the Psoriasis Area and Severity Index (PASI) [11]. The PASI assesses four body regions: the head, trunk, upper extremities, and lower extremities. For each region, the surface area involved is graded from 0 to 6, and each of the three variables (erythema, thickness, and scaling of the plaques) is graded from 0 to 4. The scores from each of the regions were added to determine a PASI score ranging from 0 to 72. The Psoriasis Severity Index (PSI) was also used to evaluate the clinical signs (erythema, thickness, and scaling) on a scale of 0 (absent) to 3 (severe) [12]. The affected body surface area (BSA) was also evaluated. The Nail Psoriasis Severity Index (NAPSI) – a simple numerical tool for the evaluation of nail psoriasis – was used to quantify the degree of nail changes [13]. The NAPSI was assessed separately for each fingernail and toenail. This scale is used to evaluate the severity of nail-bed psoriasis and nail-matrix psoriasis. Each nail was evaluated for the presence or absence of nail-matrix disease (pitting, leukonychia, red spots in the lunula, nail-plate crumbling) and nail-bed disease (oil drop/salmon patch discolouration, onycholysis, nail-bed hyperkeratosis, and splinter haemorrhage). The sum of the scores for all of the nails is the patient’s NAPSI. Nail involvement was considered for the patients with a NAPSI score ≥ 1.
Twelve-lead electrocardiogram and P-wave duration analysis
A 12-lead ECG of all patients was recorded in the supine position (Montara Instrument EU 250 Electrocardiograph, Milwaukee, WI, USA). ECG recordings were obtained at a paper speed of 50 mm/s and 10 mm/mV amplitude. The beginning of the P-wave was defined as the point at which the first atrial deflection crossed the isoelectric line, and the end of the P-wave was defined as the point at which the atrial deflection returned to the isoelectric line. The P-wave durations (Pmax, Pmin) were calculated in all 12 ECG leads. The difference between Pmax and Pmin was defined as P-wave dispersion (PWD) [14]. Two ECG readers, who were blinded to the study, evaluated the ECGs. Initially, the measurements were performed manually with the help of callipers and a magnifying glass to define the electrocardiographic deflection.
Reproducibility
Bland-Altman analyses were performed in our laboratory to evaluate the inter- and intra-observer reproducibility of the total atrial conduction time measurements, which demonstrated minimal biases and tight limits of agreement (1.8+10 and 1.7+10 ms, respectively). The intra- and inter-observer reproducibility for the left atrium peak longitudinal strain was also evaluated. For the intra-observer assessment, images from 20 randomly selected patients were reanalysed after 1 week. The Bland-Altman analysis and the intraclass correlation coefficient (ICC) indicated good inter- and intra-observer agreement: the inter-observer agreement for the left atrium global longitudinal peak strain had an average difference of 2.0 (–1.9, 4.5) and the ICC was 0.89, while the intra-observer agreement determined that the average difference was 1.5 (–2.1, 3.2) and the ICC was 0.93.
Statistical analysis
The continuous variables are presented as means ± standard deviation, and the categorical variables as percentages. The Kolmogorov-Smirnov test was used to verify the normality of the distribution of the continuous variables. The statistical analysis of the clinical data between the two groups consisted of unpaired t-tests for parametric data and the Mann-Whitney U test analysis for nonparametric data. The correlations were assessed by means of Pearson and Spearman correlation coefficients, and the c2 test was used for the categorical variables. The analyses were performed using PASW 18 (SPSS/IBM, Chicago, IL, USA) software and a two-tailed p-value less than 0.05 was considered to indicate statistical significance.
Results
Demographic and clinical characteristics
The average duration of psoriasis was 7–8 years. High sensitivity C-reactive protein (hsCRP) was higher in the psoriasis patient group than the control group (group 1: 1 ±0.8; group 2: 0.6 ±0.3, p < 0.05). Except for premature atrial extra beats no sustained atrial arrhythmia was detected in the Holter ECG. The demographic and clinical characteristics of the groups are shown in Table I. The average age of study participants in group 1 was 36 (20–60) and was 37 (20–54) in group 2; this difference was not significant (p > 0.05). Additionally, no statistically significant differences between the groups in terms of gender, BMI, smoking, waist circumference, systolic and diastolic blood pressure, fasting glucose, low-density lipoprotein (LDL) cholesterol, triglyceride and heart rate were revealed. The average NAPSI value was 23.9 ±5, the PASI value was 3.4 ±1, and the PSI value was 2.8 ±0.8.
Table I
Demographic and clinical characteristics of the groups
Echocardiographic and electrocardiographic characteristics
The echocardiographic and electrocardiographic characteristics of the groups are shown in Table II. No statistically significant differences were observed between the two groups in terms of LVEF, E wave, A wave, E/Em, DT, IVRT, LA diameter, LAVmax, LAVmin, LAVreservoir and LAGLSRa (p > 0.05). However, the LAGLS value was significantly lower in the patients in group 1 compared to control subjects in group 2 (group 1: 28.2 ±7.4; group 2: 42.0 ±3.7, p < 0.05). Additionally, the LAGLSRs value was significantly lower in the subjects in group 1 compared to group 2 (p < 0.05). The LAGLSRe value was also significantly lower in group 1 compared to group 2 (p < 0.05). With regard to the atrial conduction characteristics, TACT was significantly longer in patients in group 1 compared to the controls in group 2 (group 1: 103.5 ±3.7; group 2: 99.1 ±4.4, p < 0.05). Additionally, PWD was higher in group 1 compared to group 2 (group 1: 59.5 ±4; group 2: 47.1 ±2.9, p < 0.05).
Table II
Echocardiographic and electrocardiographic characteristics of the groups
[i] Group 1 – psoriasis patients, group 2 – control group, LVEF – left ventricular ejection fraction, DT – deceleration time, IVRT – isovolumetric relaxation time, LA – left atrium, LAV max – LA maximum volume, LAV min – LA minimum volume, LAV reservoir – LAVmax. – LAVmin, LAGLSs – global peak systolic LA myocardium strain during left ventricular systole, LAGLSRs – global peak systolic positive LA myocardial strain rate during left ventricular systole, LAGLSRe – global peak negative LA myocardial strain rate during early ventricular diastole, LAGLSRa – global peak negative LA myocardial strain rate, TACT – total atrial conduction time, PWD – P-wave duration difference; p < 0.05 indicates a significant difference.
Evaluation of correlations
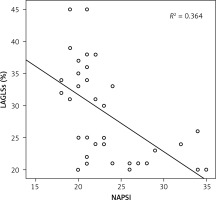
A significant and negative moderate correlation was observed between LAGLS and TACT (r = –0.57, p < 0.05), and a significant negative strong correlation was also determined between LAGLS and disease duration (r = –0.62, p < 0.05). Additionally, a significant negative correlation was detected between LAGLS and PASI, NAPSI (r = –0.45, r = –0.60; p < 0.05) (Figure 3), while a significant negative moderate correlation was observed between LAGLS and PWD (r = –0.65, p < 0.05). A significant positive moderate correlation between TACT and PWD (r = 0.42, p < 0.05) was determined. We also determined that a significant positive moderate correlation existed between TACT and disease duration (r = 0.52, p < 0.05).
Discussion
In the current study, we determined that LAGLS decreased, TACT was prolonged, and PWD was increased in psoriasis patients. Additionally, a strong negative relation between LAGLS and TACT was found. While a negative moderate correlation between LAGLS and PWD was observed, there was a positive moderate correlation between TACT and PWD. We detected a strong negative correlation between LAGLS and disease duration and parameters indicating severity of disease, while a positive moderate correlation was observed between TACT and disease duration and parameters indicating severity of disease. The incidence of psoriasis is between 0.6% and 6.5% in Europe [15]. It has been reported that psoriasis patients exhibit increased cardiovascular morbidity and mortality due to mechanisms such as chronic inflammation, endothelial dysfunction, and oxidative stress [16]. Therefore, it is important to predict the potential arrhythmias that cause morbidity and mortality in this patient group, including atrial arrhythmia. The increase in PWD observed in the psoriasis patient group suggests that chronic inflammation plays a role in the development of atrial conduction disorders. The AF is the most common atrial arrhythmia, and the increase in PWD, the prolongation of TACT, and the decrease in LAGLS are essential factors that facilitate the prediction of this arrhythmia. Ahlehoff et al. [4] demonstrated that the risk of AF increased in patients with psoriasis. Atrial fibrillation is the most common arrhythmia and confers a substantial stroke risk, ranging from < 1% to > 20% in the absence of anticoagulation [17]. The AF also may lead to heart failure, pulmonary embolism, and acute coronary syndrome. Therefore, the ability to predict AF is vital to prevent morbidity and mortality. As shown in our study, the increase of PWD and the prolongation of TACT, which indicated the presence of a disorder in sinus impulse conduction, are important in terms of predicting AF. Additionally, the decrease in LAGLS, which indicated the deformation of the atrial myocardium and the subclinical dysfunction, will facilitate the early diagnosis of atrial conduction disorders. The LAGLS and TACT may be utilised as new echocardiographic markers in the early diagnosis of subclinical electrophysiological changes in patients with psoriasis. Tsai et al. found, in a study performed on 52 patients with paroxysmal AF [18], that there was a strong relationship between AF and LAGLS. Various other studies have also shown that LAGLS decreased in response to physiopathological diseases related to the left atrium such as atrial fibrillation, heart failure, valvular diseases, hypertension, and cardiomyopathy [19–22]. While a decrease in LAGLS value was also observed in the current study, there was a strong negative relationship between the decrease and the disease duration. This result may stem from the fact that chronic inflammation and oxidative stress affect the left atrium myocardium. As the disease duration increases, the inflammation may lead to the development of fibrosis on the atrial wall. This also prolongs TACT by blocking the atrial conduction pathways with fibrosis. Atrial conduction abnormalities and left atrial mechanical dysfunction were demonstrated in the psoriasis patient group in the current study and a positive strong correlation with disease duration and severity was observed [23, 24]. This result is important for the ability to predict AF and other atrial arrhythmias. The capability to determine electrophysiological changes at the subclinical level will contribute to a reduction in cardiovascular morbidity and mortality.
Clinical applicability of the study results
The results of the current study will facilitate identification of patients who have an increased risk of developing cardiovascular disorders. Additionally, early detection of potential risk factors can facilitate the implementation of lifestyle modifications, including changes to the diet, exercise, and smoking cessation in patients exhibiting an increased cardiovascular risk. Intense medical treatments aimed at decreasing inflammation and oxidative stress would be beneficial for patients with advanced disease duration and severity, and would likely decrease cardiovascular morbidity and mortality. ECG, speckle tracking echocardiography, and atrial tissue Doppler imaging may be utilised as screening tests to assess the cardiovascular risk in patients undergoing systemic treatment. The establishment of preventive treatment strategies early during disease progression will be especially useful in young patients who exhibit a moderate or severe level of the disease.
The main limitations of the current report were the small number of patients included in the study, and the fact that no long-term follow-up of the patients was performed, especially those addressing atrial arrhythmia. Since our study was cross-sectional in design, our results cannot demonstrate causality.
In conclusion, we determined in the current study that LAGLS decreased, TACT was prolonged, and PWD increased in patients with psoriasis. There is a relationship between TACT, LAGLS, PWD and the duration and severity of disease. These results demonstrate that LAGLS, TACT, and PWD are important markers with the ability to predict AF, and are capable of detecting the subclinical electrophysiological changes in psoriasis patients. Since anti-arrhythmic preventive measures may not be effective in this specific patient subset, we believe that clinicians should be alert for patients experiencing dyspnoea and palpitations, knowing the increased prevalence of AF.