Current issue
Archive
Manuscripts accepted
About the Journal
Editorial office
Editorial board
Abstracting and indexing
Subscription
Contact
Ethical standards and procedures
Most read articles
Instructions for authors
Article Processing Charge (APC)
Regulations of paying article processing charge (APC)
OBSTETRICS AND GYNAECOLOGY / STATE OF THE ART PAPER
The role of vitamin D in perinatology. An up-to-date
review
1
Second Department of Obstetrics and Gynecology, Center of Postgraduate Medical
Education, Warsaw, Poland
2
First Department of Obstetrics and Gynecology, Center of Postgraduate Medical
Education, Warsaw, Poland
3
European Observatory of Health Inequalities, State University of Applied Sciences,
Kalisz, Poland
4
Department of Obstetrics and Gynecological Nursing, Faculty of Health Sciences,
Medical University of Gdansk, Gdansk, Poland
5
Students’ Scientific Association at the First Department of Obstetrics and
Gynecology, Medical University of Warsaw, Warsaw, Poland
Submission date: 2018-03-02
Final revision date: 2018-06-24
Acceptance date: 2018-07-05
Online publication date: 2019-01-30
Publication date: 2021-07-16
Arch Med Sci 2021;17(4):992-1005
KEYWORDS
TOPICS
ABSTRACT
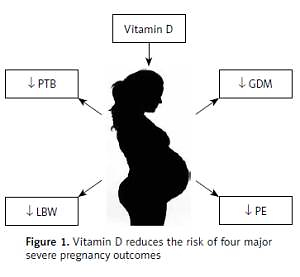
The role of vitamin D in perinatology is a subject of major interest in current medicine. There is growing evidence about the role of maternal vitamin D levels in pregnancy outcomes. The aim of this review is to summarize the current literature about the role of vitamin D in perinatology. Evidence from this review suggests associations between low levels of maternal vitamin D and higher risk of certain obstetrical complications. Vitamin D has been found to be related to preeclampsia, gestational diabetes mellitus, low birth weight, and preterm birth. The current literature supports vitamin D supplementation in pregnant women, but more high-quality data are necessary. The problem that remains is how to achieve an optimal 25-hydroxyvitamin D level. To determine the real benefits of vitamin D supplementation in pregnancy, we need high-quality trials in larger groups.
Share
RELATED ARTICLE
We process personal data collected when visiting the website. The function of obtaining information about users and their behavior is carried out by voluntarily entered information in forms and saving cookies in end devices. Data, including cookies, are used to provide services, improve the user experience and to analyze the traffic in accordance with the Privacy policy. Data are also collected and processed by Google Analytics tool (more).
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.