Central venous pressure (CVP) represents a parameter indicating the mean right atrial pressure. In daily practice, it is measured using a central venous catheter advanced via the internal jugular or subclavian (but also femoral or axillary) veins and placed in the superior vena cava near the right atrium. Normal values range from 3 to 6 mmHg [1]. CVP is usually employed to evaluate the volume status of critical patients [2]. However, invasive CVP measurement is often time-consuming, requires special monitoring equipment and trained personnel, and is burdened by some complications such as local haematoma, accidental arterial puncture, pneumothorax, infection, and venous thrombosis [3].
Point-of-care ultrasound provides a non-invasive, safe, quick, and cheap tool for the evaluation of the volume status in critically ill patients [4]. Ultrasound (US)-measured inferior vena cava (IVC) and internal jugular vein (IJV) diameters have been demonstrated to predict the volume status in venti-lated patients [5–7], but few data are available in spontaneously breathing patients [8–11]. CVP values might be subjected to the influence of heart function and intra-abdominal pressure (IAP) [12]. However, in the studies analysing the association between CVP and US measures of venous filling, information about heart function or IAP values are often lacking.
The present study was designed to investigate the relationship among US indicators of venous filling and invasive CVP in spontaneously breathing patients. IVC diameters, IJV diameters, IVC collapsi-bility index, and IJV ratio were used as ultrasonographic measures of venous filling. At the same time, all subjects underwent echocardiography and intra-abdominal pressure measurement.
METHODS
Study design
This is an observational cross-sectional study conducted in adult patients attending the Intensive Care Unit of the “Santa Maria della Scaletta” Hospital of Imola (Italy).
Inclusion criteria were as follows: age ≥ 18 years; spontaneously breathing patients; and supine position. Exclusion criteria were as follows: cerebral ischaemia, carotid artery stenosis, or bradycardia.
Written informed consent was obtained from patients or their legal guardians. The hospital’s insti-tutional ethical committee approved the study (Prot. N. 124 CE; Cod. CE: 14111).
Among 56 patients screened for inclusion, 41 were recruited. The main cause of exclusion was the ina-bility to perform all US measures.
Ultrasound examination
The ultrasound study was performed using an echo-Doppler Philips HD 11 XE (Royal Philips Electronics, Netherlands) equipped with a 12–3-MHz linear array, steerable pulsed-wave Doppler, and simultaneous ECG recording. A cardiac probe (1–5 MHz, phased array) was used for IVC imaging and transthoracic echocardiography. A single experienced sonographer, blinded to the CVP measurement, performed the ultrasound examinations. Bedside ultrasound images were obtained with the patient in the supine position.
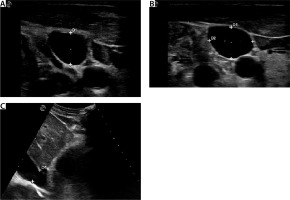
Vascular ultrasound examinations were performed as previously reported [8–10] (Figure 1). Briefly, IJV was imaged in a transverse plane, 2 cm above the clavicle, at the end of expiration. The anteroposterior diameter (AP-IJV) and transverse dia-meter (T-IJV) were recorded, and then the IJV ratio was calculated as AP-IJV/T-IJV [8]. IVC measurements were registered in a longitudinal plane with a cardiac transducer in the subxiphoid position. The IVC diameter was measured at 3–4 cm from the junction of the IVC and right atrium. B-mode was used to register a cine loop of the IVC over 2 respiratory cycles. Two IVC diameters were measured: the maximum anterior-posterior dimension at the end-expiration (IVCD-max) and the minimum anterior-posterior dimension at the end-inspiration (IVCD-min). These measures allowed us to calculate the IVC collapsibility index as IVCD-max – IVCD-min/IVCD-max × 100.
FIGURE 1
Ultrasound aspect of internal jugular vein and inferior vena cava. A) Internal jugular vein (IJV) in transverse plan (D1: anteroposterior diameter). B) IJV vein ratio calculation (D1: antero posterior diameter, D2: transverse diameter). C) Inferior vena cava (IVC) in subxiphoid view (D1: IVC diameter)
Echocardiographic imaging was performed by another experienced examiner, blinded to the vascular and CVP measurements. Left ventricular systolic function was calculated as the ejection fraction using volumetric measurements following the biplane method of disks (modified Simpson’s rule) in 2- and 4-chamber views [13]. Right ventricular (RV) systolic function was evaluated as tricuspid annular plane systolic excursion (TAPSE). TAPSE was measured using M-mode in the apical 4-chamber view. The M-mode cursor was positioned parallel to the RV free wall as it meets the tricuspid annulus. A TAPSE measurement < 17 mm is highly specific for RV dysfunction [13].
The CVP waveform from the central venous catheter and a single-lead electrocardiogram rhythm strip was registered a few minutes after the ultrasound examination. The CVP was measured at the end-expiration with the patient in a supine position. At the same time, the intra-abdominal pressure (IAP) was measured using a Foley cathe-ter filled with 250 mL of normal saline in the patient’s bladder. The normal IAP value is 5–10 mmHg. The Simplified Acute Physiology Score II (SAPS II) was also calculated for every individual [14].
Statistical analyses
All the analyses were performed using Stata/SE Statistical Software 13.1 (StataCorp, College Station, Texas, USA). Categorical variables were reported as percentages and all the continuous variables as mean ± standard deviation. The Mann-Whitney test was used to compare variables among the patient groups.
The Pearson or Spearman’s rank correlation coefficients were used, as appropriate, to assess the correlation among IVC diameters, IJV diameters, collapsibility index, and IJV ratio, and CVP.
The receiver operating characteristic (ROC) curves were plotted to compare the specificity and sensitivity of each ultrasound measurement as predictors of a “low” CVP (≤ 8 mmHg). The ROC curves for predicting “high” CVP (> 8 mmHg) were not performed due to the low number of subjects (n = 12). Multivariable linear regression analysis was used to test the independent association between ultrasound-derived indexes and CVP. A P-value < 0.05 was considered statistically significant.
RESULTS
Overall, we collected data from 41 spontaneously breathing patients. The mean age was 75 years (56% male). The median SAPS II score was 38. Sepsis was the most frequent diagnosis at admission.
Median CVP, intra-abdominal pressure, and TAPSE values were 7 mmHg (range 2–20 mmHg), 8 mmHg (range 5–18), and 25 mm (range 15–30 mm), respectively.
The mean intra-abdominal pressure and TAPSE were 9.7 mmHg and 25 mm. A low CVP (< 8 mmHg) was found in 71% (29/41) of cases.
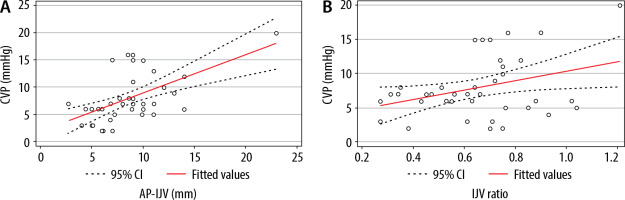
Figures 2 and 3 illustrate the bivariate correlation analyses of each ultrasound measurement with CVP. The results demonstrated a significant correlation between the following: CVP and AP-IJV (r = 0.58, P = 0.0001); CVP and IJV ratio (r = 0.35, P = 0.03); and CVP and IVCD-max (r = 0.35, P = 0.02). Conversely, the IVC collapsibility index was not related with CVP (r = –0.24, P = 0.13).
FIGURE 2
A) Correlation between the central venous pressure (CVP) and inferior vena cava (IVC) max diameter: r = 0.35, P = 0.02. B) Correlation between the CVP and IVC collapsibility index: r = 0.24, P = 0.13
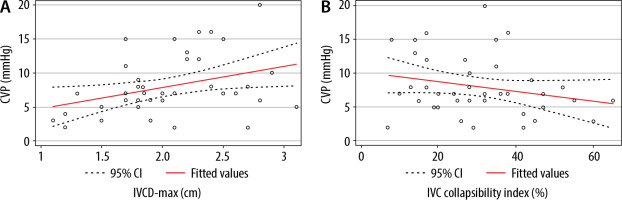
FIGURE 3
A) Correlation between the central venous pressure (CVP) and internal jugular vein (IJV)anteroposterior (AP) diameter: r = 0.58, P < 0.001. B) Correlation between the CVP and IJV ratio: r = 0.35, P = 0.03
Differences in clinical and ultrasound parameters between subjects with “low” and “high” CVP are shown in Table 1. The “high” CVP group had significantly lower LVEF, and significantly higher values of AP-IJV and IJV aspect ratio.
TABLE 1
Characteristics of patients and ultrasound measurements
[i] SAPS – Simplified Acute Physiology Score, MAP – mean arterial pressure, LVEF – left ventricular ejection fraction,
The AUROC values of each tested variable for predicting “low” CVP (≤ 8 mmHg) are shown in Table 2. The AP-IJV had an AUC value of 0.79 (95% CI: 0.63–0.91), whereas the IJV ratio (0.68 [95% CI: 0.51–0.82]), IVCD-max (0.66 [95% CI: 0.49–0.80]), and IVC collapsibility index (0.62 [95% CI: 0.45–0.77]) showed lower AUC values.
TABLE 2
Test characteristics of ultrasound technique in predicting central venous pressure < 8 mmHg among patients in spontaneous breath
Furthermore, an AP-IJV value ≤ 7 mm showed the best performance in predicting a CVP < 8 mmHg, with 88% sensitivity and 60% specificity. Collapsi-bility index value > 17, IVCD-max ≤ 2.1 cm, and IJV ratio ≤ 0.63 had moderate sensitivity and specificity in predicting low CVP.
Finally, in multivariable linear regression analysis, only the AP-IJV showed a significant association with CVP: β coefficient = 0.63 (95% CI: 0.30–0.96) with P < 0.001.
DISCUSSION
The results of the present study demonstrated that AP-IJV, the IJV ratio, and IVCD-max significantly correlated with the invasive CVP, whereas IVC collapsibility index did not show any association. Among them, AP-IJV showed the best correlation with CVP (r = 0.58, P = 0.0001) and good accuracy in predicting low CVP (≤ 8 mmHg), with a sensitivity of 88%, a specificity of 60%, and an AUROC of 0.8.
In the last decades, point-of-care ultrasound has gained increasing popularity because it represents a non-invasive, safe, quick, and cheap tool for the evaluation of volume status in critical patients [5–11]. In particular, some guidelines have proposed the evaluation of intravascular volume status based on sonographic examination of the IVC [4]. In a recent systematic review, it was found that US measurement of IVC diameter and collapsibility represent a valid method for estimating CVP and right atrial pressure [11]. Furthermore, point-of-care ultrasound has proven useful also in paediatric intensive care units because it is radiation free, non-invasive, quick, and can be done at the patient’s bedside [15, 16]. However, the IVC ultrasound assessment is not feasible in the case of obesity, abdominal air interposition, or surgical wounds. For this reason, seve-ral investigations focussed on ultrasound evaluation of the IJV as a surrogate indicator of CVP [17–19].
The first study investigating the application of IJV-US-derived measures in the estimation of volume status date back to 2000. In these pioneering observations, Lipton described the sonographic patterns of IJV in patients with low, normal, and elevated CVP [20]. Later, Donahue et al. [9] explored the correlation between IJV-US measures and CVP in a cohort of 34 non-ventilated ICU patients. The results of this pilot study demonstrated that patients with CVP ≤ 10 cm H2O showed a mean IJV diameter of 7.0 mm, whereas patients with CVP > 10 cm H2O had a mean diameter of 12.5 mm. Furthermore, a strong intra- and interobserver agreement was found, with a correlation coefficient of 0.92 [9]. Similar results were obtained by Keller et al. [10] in a sample of 44 spontaneously breathing patients. The authors demonstrated that the IJV aspect ratio strongly predicted a CVP of 8 mmHg, with an area under the receiver operating characteristic curve of 0.84.
The predictive power of US measurement of IJV was also validated against the right heart catheterization. Indeed, an increase in IJV cross-sectional area > 17% during Valsalva excludes elevated right atrial pressure [21] and predicts 30-day re-hospitalization in patients with acute decompensated heart failure [22].
Of note, few studies have compared the accuracy of US-IJV and US-IVC in predicting CVP [8, 23, 24]. The results of these investigations are varied and often conflicting. In detail, a cross-sectional study comparing 3 point-of-care ultrasounds found that IVC diameter is a stronger predictor of CVP than the IVC collapsibility index or the IJV aspect ratio [8]. On the other hand, Avcil et al. [24] demonstrated that IJV had the best diagnostic performance in estimating CVP when compared with US-IVC measures (IVC-max, IVC-min, IVC collapsibility).
The present study demonstrates that AP-IJV strongly correlated with CVP (r = 0.58, P = 0.0001) in both simple and multivariable linear regression analysis. It also shows sensitivity and specificity comparable with those of previous studies [9, 24]. The original purpose of our investigation was the assessment of right and left systolic cardiac function and intra-abdominal pressure. As is known, all these variables might alter the CVP values. In the present study, patients had normal right ventricular function and normal intra-abdominal pressure. Therefore, the values of tested variables (CVP, IJV, and IVC diameters) were due to the intravascular volume status and not the cardiac dysfunction or elevated intra-abdominal pressure.
The present study has some shortcomings that need to be discussed. First, the small sample of subjects prevents a definite conclusion being drawn about this topic. Second, ultrasound examination is a highly operator-dependent tool, and this makes it difficult to compare the results of different studies. Finally, we employed CVP as a reference method to estimate the volume status of patients. However, it has been demonstrated that dynamic parameters (such as pulse pressure variation, stroke volume variation, passive leg raising, etc.) are better predictors of fluid responsiveness than static parameters (such as CVP and others).




