Introduction
Intercellular communication is an important mechanism involved in physiological processes and associated with many diseases and injuries [1]. In multicellular organisms, individual cells communicate with each other to exchange their nutrients and signals, which are necessary for their biological processes such as proliferation, differentiation, apoptosis, wound healing, and homeostasis for maintenance of the organization. There are multiple strategies multicellular organisms have evolved, such as neural or endocrine systems which mediate long-range interactions or direct physical or cell–cell contact which mediate short-range interactions. Besides the field that was solely defined previously by the simple exchange of endocrine, autocrine and epicrine agents, some more complex forms of intercellular communication have been witnessed in the last few years that include microvesicles of various types, including exosomes, bioelectrical mechanisms, including local field potentials, volume transmission and special communication cells known as telocytes [2].
The gap junctions formed by connexin proteins facilitate intercellular communication between neighboring cells by allowing the transfer of ions and small signaling molecules [3]. Gap junctions are intercellular channels formed by connexins, a family of transmembrane proteins with the capacity to form gap junctions, and by the gap junctions, the cytoplasm of adjacent cells is directly linked. The most ubiquitously expressed connexin isoform in human tissues is connexin 43 (Cx43) [4]. With their important roles in maintaining the cell functions, dysregulations of connexin expression and function are involved in numerous pathogeneses, including cancer. It is evidenced that disorder in gap junctions and their structural subunits, connexins, often exists in, and sometimes causes, various proliferative disorders, including cancer [5].
Cancer has long been a major public health problem worldwide and is the second leading cause of death in the United States, where 1,685,210 new cancer cases and 595,690 cancer deaths were projected to occur in 2016 [6]. In China, with increasing incidence and mortality, cancer has become the leading cause of death and it was estimated that 4,292,000 new cancer cases and 2,814,000 cancer deaths would occur in 2015 in China [7]. Cancer is associated with many concerns and gastric cancer has an important place in the worldwide incidence of cancer and cancer-related deaths [8–13]. Gastric cancer is the fifth most common cancer and third leading cause of cancer-related death worldwide [14]. It was projected that there would be 26,370 new gastric cancer cases and 10,730 gastric cancer deaths in the United States in 2016 [6]. In China, 6,791,00 new gastric cancer cases and 498,000 gastric cancer deaths are expected [7]. Although ample evidence suggests the role of the gap junction and its connexins such as connexin 43 in a variety of human cancers, detailed examination in specific cancers, such as gastric cancer, is still not abundant; also, a number of contradictory studies have reported both pro- and anti-proliferative effects that often depend on the cell or tissue type or the microenvironment [5]. More data on the role of the gap junction and its connexins such as Cx43 involved in gastric cancer remain necessary.
This study investigated the changes in gap junction ultrastructure and Cx43 in normal gastric tissue and in carcinomatous gastric tissue at different degrees of differentiation in order to provide a novel research direction for gastric cancer pathogenesis as well as to provide potential targets for drug treatment.
Material and methods
Objects
Sixty-four samples of gastric carcinomatous tissue were surgically removed for collection from the patients in the affiliated hospital of Chengde Medical College from July 2010 to February 2013. In addition, 29 samples of normal gastric tissue were also collected (normal tissue was located more than 10 cm away from the carcinomatous edge). All of the samples were collected after formal written consent was obtained, in accordance with the guidelines of the Ethics Committee of the Affiliated Hospital of Chengde Medical College. None of the specimens had been treated with preoperative chemotherapy or radiotherapy. Among the 64 gastric carcinomatous samples, 47 samples were from male patients, and 17 samples were from female patients. The patients’ age range was 45–67 years, with a median age of 56.5 ±10.0. Twenty-six samples were of well-moderately differentiated adenocarcinoma, and 38 samples were of poorly differentiated adenocarcinoma. Twenty-seven of the samples did not have lymph node metastasis, whereas 37 cases did have lymph note metastasis. The TNM stage I and TNM stage II were merged as one group, while TNM stage III and TNM stage IV were merged as another group, thus it is kept consistent with the Figure 1. Fifty of the samples had serious penetration of carcinoma tissue, and 14 cases did not have serious penetration as detected by transmission electron microscopy, Western blotting, and RT-PCR.
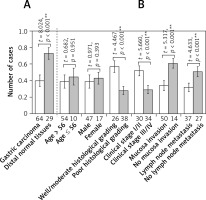
Figure 1
Level of Cx43 protein expression in normal and carcinomatous gastric tissues determined by western blot. Error bars indicate standard deviation of the means. Double asterisks indicate t-test p < 0.01. A – Cx43 protein expression in normal and carcinomatous gastric tissues. B – Cx43 protein expression in carcinomatous gastric tissues with different clinicopathological features
Reagents and instruments
The reagents for preparing the electron microscopy sample were as follows: glutaraldehyde, osmic acid, Epon812, DDSA, uranium acetate, lead citrate, and so on. These were purchased from Sigma Aldrich in the United States. The Western blotting detection reagents and anti-Cx43 rabbit anti-human primary antibody were purchased from Bioworld Technology, Co, Ltd., Nanjing, China. RIPA organization cracking liquid (BB120031) was purchased from the Shanghai Bestbio Company (Bestbio, Shanghai, China). The BCA protein concentration determination kit, HRP-labeling sheep anti-rabbit secondary antibodies, and hypersensitive ECL luminous fluid were all purchased from the Beijing Solarbio Science and Technology Co., Ltd (Beijing, China). The remaining reagents (such as 30% acrylamide, β-mercaptoethanol, 10% SDS, 10× ponceau S, 10% ammonium persulfate, 1.5 M/1.0 M Tris-HCl, TBST, 1× electrophoresis buffer and transmembrane liquid, developing powder, fixing powder, etc.) were from the Chengde Medical College Foundation Institute of Molecular Laboratory. The RT-PCR kit and DNA marker were purchased from the Dalian Treasure Biological Engineering Co., Ltd (Dalian, China). The PCR primers were designed and synthesized by the Beijing Parkson Gene Technology Co., Ltd. (Beijing, China). The primer sequences of Cx43 are 5′-TCTCGCCTATGTCTCCTCCTGG-3′ (upstream primer) and 5′-AGTTAGAGATGGTGCTTCCCGC-3′ (downstream primer), with amplified fragments of 156 bp. The primer sequences of P85 α are 5′-TGCTATGCCTGCTCTGTAGTGGT-3′ (upstream primer) and 5′-GTGTGACATTGAGGGAGTCGTTG-3′ (downstream primer), with amplified fragments of 175 bp. The primer sequences of β-actin are 5′-AGCGGGAAATCGTGCGTGAC-3′ (upstream primer) and 5′-ACATCTGCTGGAAGGTGGAC-3′ (downstream primer), with amplified fragments of 453 bp. The following instruments were used in this study: JEM-1200EX TEM (JEOL Ltd., Tokyo, Japan), LELCA ULTRACUT UCT Ultra microtome (LELCA, Vienna, Austria), ultraviolet spectrophotometry (DU800; Beckman, Brea, USA), enzyme standard instrument MK3 (Thermo, Waltham, USA), voltage steady flow electrophoresis apparatus (Shanghai Xin Industrial Co., LTD., Shanghai, China), PTC-220-PCR amplification (MJ Research, Inc., Watertown, USA ), and 2020 D fluorescent UV digital imager (GoldSpring, Beijing, China).
Transmission electron microscopy (TEM) assay
Tissue samples were sliced into three 1-mm tissue blocks and fixed in 2.5% glutaraldehyde. Then, the samples were dehydrated step by step and embedded in Epon for ultrastructure observation by TEM technology.
Western blotting assay
Tissue were extracted with lysis buffer (150 mM NaCl, 1% NP-40, 0.1% SDS, 2 mg/ml aprotinin and 1 mM PMSF (Solarbio, Shijiazhuang, China)) for 30 min at 4°C. Extracts were centrifuged at 15,000g for 15 min at 4°C. Supernatants containing total protein were then harvested. Aliquots, each containing 50 mg of protein, were separated by 12.5% SDS-PAGE and transferred to PVDF membranes at 80 V or 120 V for 2 h at low temperature. The membranes were blocked in 5% skim milk for 2 h, and proteins were detected using monoclonal antibodies overnight at 4°C. Proteins were visualized using anti-rabbit IgG conjugated with horse radish peroxidase (HRP) for 2 h at 28°C, respectively. Proteins were detected using the ECL Western blotting detection reagents and using IPP image analysis software for analysis of band density. The relative protein levels were calculated based on GAPDH as the loading control.
RT-PCR assay
Total RNA was extracted from gastric cancer tissues and adjacent normal tissues using a Trizol kit. The RT-PCR conditions for Cx43 were as follows: 35 cycles of 94°C pre-denaturation for 5 min, 94°C denaturation reaction for 30 s, 58°C annealing reaction for 30 s, and 72°C extension reaction for 1 s followed by a 72°C extension reaction for 5 min. The RT-PCR conditions for β-actin were as follows: 30 cycles of 94°C pre-denaturation reaction for 5 min, 94°C denaturation reaction for 30 s, 55°C annealing reaction for 30 s, and 72°C extension reaction for 1 min followed by a 72°C extension reaction for 5 min. Then, 2% agarose gel electrophoresis was used for detecting and quantifying the PCR-amplified products. Positive staining of the electrophoresis gel resulted in the appearance of the corresponding sites. β-actin was used as the internal standard, and the gel electrophoresis results were measured by an electrophoresis image analysis system.
Results
Ultrastructure of gap junction observed by electron microscopy
In the normal gastric tissue intercellular junction in the glandular cavity, tight junctions, desmosomes, and gap junctions were observed. The intercellular junction structure was damaged in the well-moderately differentiated gastric cancer tissues and was seriously damaged in the poorly differentiated gastric carcinoma (Figure 2).
Figure 2
Ultrastructure of gap junction in normal gastric tissue, well-moderately differentiated and poorly differentiated gastric carcinoma tissue. The gap junction ultrastructure in the tissues was determined by electron microscopy. A – Complete gap junctions in normal gastric tissue (TEM 100000×, bar = 500 nm). B – Incomplete gap junctions in well-moderately differentiated gastric carcinoma tissue (TEM 100000×, bar = 1 μm). C – Incomplete gap junctions in poorly differentiated gastric carcinoma tissue (TEM100000×, bar = 1 μm)
Expression of Cx43 protein detected by Western blotting
The relative Cx43 protein content in the 29 cases of normal gastric tissue was 0.82 ±0.04, and in the 64 cases of gastric cancer tissue, it was 0.51 ±0.11. The differences are statistically significant (p < 0.05). The relative Cx43 protein content in the well-moderately differentiated gastric cancer tissues was 0.64 ±0.04, whereas in the poorly differentiated gastric carcinoma, the content was 0.42 ±0.06. The relative content in TNM stage I/II was 0.67 ±0.03, and it was 0.37 ±0.09 in TNM stage III/IV. The relative Cx43 protein content in the group in which there was serious membrane penetration was 0.45 ±0.09, and in the group without serious membrane penetration, it was 0.72 ±0.03. The relative expression level of the Cx43 protein in the group with lymph node metastasis was 0.41 ±0.07, and in the group without lymph node metastasis, it was 0.65 ±0.04. In the independent two-samples t-test, the aforementioned differences had statistical significance (p < 0.05, p < 0.05, p < 0.05, p < 0.05), whereas the differences in the Cx43 protein expression level between different sex and different age groups were not statistically significant (both p > 0.05, Figures 1, 3).
Expression of Cx43 mRNA detected by RT-PCR
The relative Cx43 mRNA content in the 29 cases of normal gastric tissue was 0.73 ±0.07, and in the 64 cases of gastric cancer tissue, the content was 0.40 ±0.07. The differences are statistically significant (p < 0.05). The relative Cx43 mRNA content in the well-moderately differentiated gastric cancer tissues was 0.57 ±0.07, whereas the relative content in the poorly differentiated gastric carcinoma was 0.28 ±0.05. The relative content in TNM stage I/II was 0.52 ±0.06, and the relative content in TNM stage III/IV was 0.29 ±0.05. The relative Cx43 mRNA content in the group with serious membrane penetration was 0.34 ±0.06, and in the group without serious membrane penetration, the content was 0.61 ±0.06. The relative expression level of Cx43 mRNA in the group with lymph node metastasis was 0.32 ±0.05, and in the group without lymph node metastasis, it was 0.51 ±0.06. According to the independent two-samples t-test, the differences had statistical significance (p < 0.05, p < 0.05, p < 0.05, p < 0.05); however, the differences in the Cx43 mRNA expression level between different sex and different age groups were not statistically significant (both p > 0.05, Figures 4, 5).
Figure 4
Lanes of Cx43 mRNA expressed in gastric cancer and normal gastric mucosa tissues determined by RT-PCR. 1 – Normal gastric mucosa, 2 – well and moderately differentiated gastric cancer, 3 – poorly differentiated gastric cancer
Figure 5
Level of Cx43 mRNA expression in normal and carcinomatous gastric tissues determined by RT-PCR. Error bars indicate standard deviation of the means. Double asterisks indicate t-test p < 0.01. A – Cx43 mRNA expression in normal and carcinomatous gastric tissues. B – Cx43 mRNA expression in carcinomatous gastric tissues with different clinicopathological features
Discussion
Gap junction intercellular communication (GJIC) is a means of communication for adjacent cells that is mediated by gap junctions. It is involved in the passing of information among cells and plays an important role in the regulation of cell proliferation and differentiation. The main channel component is the gap junction protein connexin 43 (Cx-43). At present, both domestic and foreign researchers are conducting research on the relationship between GJIC and cancer development. This is because a defect in Cx expression, a loss in GJIC function, and intercellular communication disorders can result in the loss of normal contact between cells. As a result, regulatory signals cannot reach malignant cells from normal cells, and thus unlimited proliferation, material energy metabolic abnormalities, and other disorders result. These factors may contribute to tumor occurrence [15–18]. Due in part to this possibility, with the consideration that cancer, associated with many concerns, has long been a major public health problem worldwide [6, 14, 19–21], gap junctions and gap junction protein have become hot topics in current bioscience research. This study investigated the changes in gap junction ultrastructure and Cx43 in normal gastric tissue and in carcinomatous gastric tissue at different degrees of differentiation in order to provide a novel research direction for gastric cancer pathogenesis as well as to provide potential targets for drug treatment.
Direct information transfer between cells is achieved by intercellular junctions, which include gap junctions, desmosome junctions, and tight junctions. Tight junctions do not have communication capabilities, and there is little research on the communication capabilities of desmosome junctions. However, the biological function of gap junctions has recently received a great deal of attention. The gap junction is a kind of plate connection with a large contact area, and it is more common in the deep side of epithelial cells where the adjacent cell membrane is separated by 2 nm. The adjacent cell membrane contains many hexagonal particles (6–8 nm in size) that are known as connexons. Each connexon is composed of six transmembrane intrinsic protein molecules, which form a central tunnel that is 2 nm in diameter. The tunnels of adjacent connexons on the plasma membranes of corresponding connected cells form direct channels between the cells. This tiny pipe-type channel is composed of six sub-units and the shifting of the six sub-units turns on or turns off the channel [19, 22]. Through observing normal and cancerous gastric tissue ultrastructure via electron microscopy, we observed that the normal gastric mucosal gap junction structure is complete, whereas the gap junction structure of gastric cancer is damaged, and the poorly differentiated gastric cancer tissue’s gap junction structure has a greater degree of damage. It can be concluded that because there is a lack of cell gap junction communication function, the GJIC function is reduced or suppressed after the cells have transformed. This may be an important mechanism for promoting carcinogenesis, and the lack of gap junctions may be involved in tumor occurrence.
Gap junctions are the structural basis of cellular communication, and the diversity of gap junction function is based on the diversity of the molecular structure of gap junction proteins. One such membrane protein is Cx43, which is the structural basis of cell metabolism coupling and impulse conduction. Cx43 is an important member of the gap junction protein family and plays an important role in the intercellular information exchange and regulation of cell proliferation and differentiation in normal cells. Furthermore, it is also the main connecting protein in the GJIC of gastric cancer tissues [18].
Decreased or absent Cx43 expression is closely related to the occurrence and development of various tumors [23–30]. The inhibition of Cx43’s effect on tumor growth is considered through the following: some growth inhibitors are distributed by gap junctions (GJ), such as through cAMP, to participate in GJ-dependent growth inhibition so that GJIC function is decreased or absent, and the control signals from normal cells cannot reach the malignant cells, and the resulting unrestricted growth can lead to tumor occurrence [24, 25, 27, 28]. In normal cell carcinoma, the expression of the gap junction protein is decreased, and there is GJIC dysfunction, which inhibits the function of cellular communication and allows for abnormal growth of tumor cells. Thus, Cx43 plays an important role in tumor malignant progression. The finding of the study also confirmed that the protein expression of Cx43 in normal gastric mucosa tissues was higher than that in carcinoma tissues, which indirectly confirms that there was gap junction communication dysfunction in the process of cell carcinogenesis. Tang B [28, 31] and other researchers have found that the expression of Cx43 in gastric cancer cells is significantly lower than that in the adjacent normal cells. Moreover, the Cx43 expression in gastric cancer cells in the lymph nodes without metastasis was significantly higher than that of gastric cancer cells in the lymph nodes with metastasis. This suggests that low expression of Cx43 may contribute to the occurrence of gastric carcinoma.
In conclusion, this study confirmed that the expression of Cx43 in gastric cancer tissues was lower than that in normal gastric mucosa tissues, and the expression of Cx43 in gastric cancer with lymph node metastasis was lower than that without lymph node metastasis. Furthermore, the expression of Cx43 in gastric cancer in TNM stage I/II was higher than that in TNM stage III/IV. Therefore, we speculate that Cx43 may be involved in various tumor factors that affect the occurrence of gastric cancer and that Cx43 can affect the biological behavior of gastric cancer. As a result, decreased Cx43 expression levels and abnormal gap junction intercellular communication function may be involved in the progression of the malignant degree of gastric cancer and the metastasis of tumor cells.