Introduction
Spinal surgery is one of the fastest-growing areas of treatment in medicine. In the last two decades, we have observed a large increase in the number of performed spinal procedures, for several reasons: improved imaging diagnostics, introduction of many new techniques and operational tools, and a significant increase in spine diseases associated with an aging population. For each operation it is necessary to use intraoperative imaging based on ionizing radiation, which greatly facilitates the work of the surgeon, but unfortunately exposes the person to the adverse effects of radiation.
Vertebral compression fractures (VCF) of the thoracolumbar spine have become an important medical and socioeconomic problem. Their etiology is multifactorial: most commonly observed low-energy VCF are sequelae of osteoporosis, while less frequently VCS are caused by trauma or spinal neoplasms. The choice of treatment modality depends on fracture pathogenesis and its morphology and on neurologic symptoms [1]. First described in 1987, percutaneous vertebroplasty (PV) has been ever since widely used to treat VCF without neurologic deficit, vertebral hemangioma or vertebral osteolytic lesions [2]. The PV belongs to the category of the so-called minimally invasive spine surgery (MISS) methods, advocated in last decades in the treatment of VCF (osteoporotic or traumatic) for decreased operative time, less morbidity and simplicity as compared to open surgery [1, 3, 4]. This method involves injection of radiopaque bone cement – most commonly polymethylmethacrylate (PMMA) – into the vertebral body. Conventionally the procedure is performed under fluoroscopic guidance provided by the isocentric C-arm unit [5, 6]. The pedicle access needle is introduced into the vertebral body and is followed by cement injection. Conventional needle placement is focused on anatomic landmarks of vertebrae. However, in certain conditions the visibility of target vertebra may be obstructed due to pathology (high osteoporosis, lytic lesion) and also imaging may be affected by surrounding structures (shoulders, scapulae, ribs and fat tissue).
In recent years, intraoperative spinal navigation techniques coupled with the 3D imaging system (O-arm) have been used to guide precise placement of transpedicular screws, and later to perform percutaneous kyphoplasty [7, 8]. Both mentioned guidance methods with the C-arm or O-arm use radiation posing certain risk for the medical personnel performing the spinal procedures. According to some authors, exposure of the operating room personnel seems to be underestimated [7, 9]. Matityahu et al. in a systematic review of exposures during orthopedic procedures highlighted that the highest radiation doses or effective doses were found in spinal surgery, especially for vertebroplasty and kyphoplasty [8].
This study aimed to compare the radiation emitted for vertebroplasty procedures performed using the O-arm combined with neuronavigation and under fluoroscopic guidance using the standard mobile C-arm. To the best of the authors’ knowledge, radiation emitted during C-arm-assisted and O-arm-assisted vertebroplasty has not been compared previously.
Material and methods
Population
The subjects of analysis were patients treated at the authors’ institution divided into 2 groups. The first group (O-arm group) comprised 29 patients (28 female and 1 male, aged 18–89 years, mean age: 66.7 years) treated from 2016 to 2017 with O-arm-guided vertebroplasty procedures performed in 44 vertebrae.
The reason for compression fractures in the examined group of patients was osteoporosis in 18 (62.06%) patients, pathological fracture in 8 (27.6%) patients and a fresh non-osteoporotic fracture in 3 (10.34%) patients. Fractured vertebrae were situated in the spinal segment from Th1 to L5 and dominated in the thoracolumbar junction. In 40% of cases fractures were multilevel, affecting 2 or 3 vertebrae. The second group (C-arm group) comprised 35 patients with 40 vertebrae treated using fluoroscopic guidance, among whom 26 (74.29%) were female and 9 (25.71%) were male. Patients’ age in this group ranged from 30 to 85 years (mean age: 68.9 years). In this group of patients, osteoporosis was also the main cause of spinal fractures (> 70% of patients).
Table I presents patients’ demographics and treated vertebrae.
Table I
Patients’ clinical and radiological features
Surgery
Trained operators performed all procedures in an operating room under general anaesthesia.
O-arm group
For the O-arm procedure we used the O-arm (Medtronic PLC, Littleton, Massachusetts, USA) integrated with Stealth Station Navigation System (Medtronic PLC, Louisville, Colorado, USA). Patients under general anaesthesia were placed prone on the radiolucent table. Sterile preparation of the operation site was followed by attachment of the reference frame to the spinous process and three-dimensional (3D) examination of the treated spinal segment with the O-arm. During the data acquisition (not exceeding 30 s), all medical personnel were outside the operating theatre. The data were automatically transmitted to the neuronavigation system (Figure 1) and used to guide the vertebroplasty needle into the target vertebra (Figures 2, 3). After proper needle placement the PMMA was injected under the real-time image given by the O-arm switched to the fluoroscopy mode.
Figure 1
Operating theatre, O-arm (Medtronic PLC, Littleton, Massachusetts, USA) integrated with Stealth Station Navigation System (Medtronic PLC, Louisville, Colorado, USA)
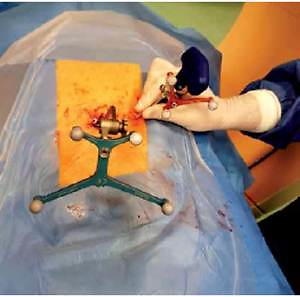
Figure 2
Patient under general anaesthesia placed prone on the radiolucent table. Operation site with the reference frame attached to the spinous process via a midline incision. The 3D tracer is attached to the vertebroplasty needle in order to perform continuous neuronavigation
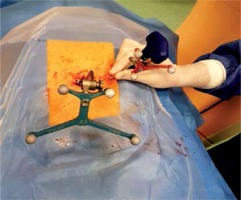
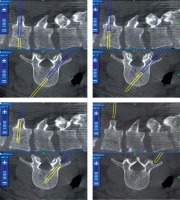
Figure 3
Navigation images showing transpedicular advancement of the vertebroplasty needle into the central zone of the target vertebra
The O-arm also enabled post-procedural 3D imaging to be obtained to estimate the result of vertebral body filling and to visualize potential extravertebral cement leak. Mean volume of PMMA injected per vertebra was 4.03 ml (range: 2.0–7.5 ml). Radiation dose and time were recorded by a built-in dosimeter. In the lumbar vertebrae mean volume of injected PMMA was higher than in the thoracic ones (respectively: 4.58 ml and 3.42 ml).
C-arm group
The traditional PV procedure was performed using radio-fluoroscopic guidance with a mobile C-arm fluoroscopy unit (Siremobil Compact L, Siemens, Erlangen, Germany). During the procedure, the X-ray source was primarily placed under a surgical table and could be rotated with the image intensifier from antero-posterior to lateral position. Exposure parameters were determined by an automatic mode and brightness control. The radiograph unit was equipped with a permanent dose area product meter and a beam-on timer. The radiation time and dose could be read directly from the unit. Patients under general anaesthesia were placed prone on the radiolucent table. The trajectory of the needle and PMMA injection were controlled by the C-arm multiple anteroposterior and lateral images of treated vertebra in pulsed or cine fluoroscopy.
Ethics
All patients provided written consent for treatment. For retrospective study of radiation doses approval of the Institutional Ethics Committee was not necessary.
Statistical analysis
Sets of radiation dose values without initial and final 3D scans, when the surgeon was outside of the operating room, obtained in the O-arm group were compared with the ones in the C-arm group. We used the data analysis software system Statistica version 9.0 (Stat Soft Inc., 2010) and Microsoft Excel software. Both sets of data were characterized by minimum, maximum, mean value, median and standard deviation. The following means of testing were used during this study: Student’s t-test, the Shapiro-Wilk test and the Mann-Whitney U test. The level of statistical significance was set at p = 0.05.
Results
In the O-arm group, mean radiation dose was 912.29 cGy/cm2 vs. 1772.13 cGy/cm2 noted in the C-arm group (Table II). The difference was statistically significant (p = 0.000069). In the O-arm group, mean radiation dose per vertebra was 601.28 cGy/cm2 vs. 1506.86 cGy/cm2 noted in the C-arm group (Table II). The difference was statistically significant (p = 0.000000004). In neither of the two groups were any surgery-related complications observed.
Table II
Values of intraoperative radiation exposure
Discussion
In recent years, MISS methods have become increasingly popular in treatment of various vertebral compression fractures. Percutaneous techniques such as vertebroplasty, kyphoplasty and transpedicular stabilization are replacing conservative methods of treatment and open surgical methods [1, 3, 4]. Vertebroplasty reinforces the mechanical strength and stabilizes the shape of the fractured vertebral body, which constitute the main advantages of this modality [10]. In consequence, the procedure reduces pain and need of medication and finally improves patients’ activity and quality of life.
Recently the World Health Organization has stated that osteoporosis is an emerging health problem in aging societies [11, 12]. In the year 2000, there were about 9 million osteoporotic fractures worldwide with 1.4 million vertebral fractures [13]. In Europe, 400,000 new vertebral fractures are diagnosed yearly and, according to the demographic prognostics, this number may be doubled by the year 2050 [12, 14–16]. Osteoporosis is responsible for 80% of vertebral fractures, while the remaining 20% are a result of trauma and tumors [15, 17]. Increasing use of MISS allows prediction of the growing number of patients treated with these modalities in the future. These techniques as compared with open surgery offer such advantages as smaller wounds, negligible blood loss, shorter hospitalization time, and lower risk of complications [3, 18–20]. However, the risk of radiation for patients and medical staff is the main drawback of MISS procedures, which are generally performed under fluoroscopic guidance [9].
There are many methods of radiological protection that minimize the potential risk of radiation to which surgeons and the rest of the operating room staff are exposed. To avoid long-term unfavorable effects of ionizing radiation exposure for many years, various radiological shields are used for the most sensitive parts of the human body. The hands of the surgeon, chest, abdomen, neck, thyroid and eyeballs are particularly sensitive when exposed to ionizing radiation.
On the basis of literature analysis, Srinivasan et al. proposed recommendations significantly reducing the exposure of spine surgeons and supporting personnel. The authors recommend the use of various types of lead shields, but also the proper setting of parameters and location of the fluoroscope. One of the recommendations to consider is use of the O-arm and neuronavigation in the case of multi-level changes of the spine [21].
In our department, the use of the O-arm with neuronavigation does not exempt the surgeon from observing the basic principles of radiological protection with lead shields protecting sensitive organs, proper setting and distance from the radiation source and leaving the operating room whenever possible.
The above-mentioned rules have to be applied when the O-arm is used in the fluoroscopy mode. During the acquisition of 3D images the operating room staff always leaves the operating room.
Efstathopoulos et al. stated that the eye lenses and hands of a surgeon are placed at high risk during vertebroplasty and kyphoplasty [22]. The dose value of ionizing radiation exposure for eye lenses and extremities is limited respectively to 150 mSv and 500 mSv per year [22]. A great deal of attention was paid to staff members who are frequently subject to and are more likely to suffer from radiation side effects than patients rarely exposed to radiation more than once. At present there are no studies comparing fluoroscopically assisted PV and PV guided by O-arm and navigation. The only available research describes differences between fluoroscopically assisted kyphoplasty and O-arm guided kyphoplasty [23, 24]. In his prospective study, Schils assessed the average radiation dose and exposure time during percutaneous kyphoplasty guided by O-arm combined with neuronavigation [23]. His study included 54 cases with 76 fractured vertebrae. Among them 70% were a direct result of osteoporosis, 24% were injuries unrelated to osteoporosis and the remaining 6% were pathological fractures caused by multiple myeloma [23].
The authors compared the intraoperative radiation dose during vertebroplasty performed under radio-fluoroscopic guidance (C-arm) versus vertebroplasty guided by O-arm combined with neuronavigation. It is important to note that in the O-arm group the radiation dose levels used for the analysis included only the doses to which the surgeon was exposed during the bone cement implantation (injection), without the initial and final 3D scans, when the personnel always leaves the OR. Thanks to the O-arm combined with navigation, access to the vertebral body is achieved under navigation, without the need of any fluoroscopic control. The present study aimed to determine which method of guidance offers less radiation exposure of patients and medical staff. Similarly to Schils’ study [23], this paper presents a comparison of the radiation dose and time of exposure during percutaneous vertebroplasty guided by O-arm combined with neuronavigation. In Schils’ study, the average time of fluoroscopy when using the O-arm combined with neuronavigation was 2 min shorter than in cases with use of the C-arm – 3.1 min and 5.1 min respectively [23]. Consequently, less time was needed to carry out the procedure: 38 min for the O-arm and 47 min for the C-arm [23]. Based on these results, Schils states that the O-Arm combined with neuronavigation allows the surgeon to perform kyphoplasty having only 1/3 of radiation typically required for a traditional procedure guided by fluoroscopy [23]. Moreover, Schils et al. reported that addition of the Cement Delivery System (CDS) (Medtronic Spine LLC, Sunnyvale, CA, USA) to the O-arm and navigation reduces the dose of radiation by up to 1/5 in comparison to a traditional method [25].
Izadpanah et al. measured radiation exposure and operation time in a kyphoplasty procedure with and without a navigation system [24]. Patients were divided into 2 groups: a group of 29 subject to computer navigated kyphoplasty and a control group treated by means of a conventional C-arm controlled kyphoplasty procedure. Results in the latter one revealed that the dose area product (DAP) applied to the patient was on average 1972 cGy cm2 for the thoracic spine and 2105 cGy cm2 for the lumbar spine. In the navigated group of patients, DAP was significantly lower: 1318 cGy cm2 (thoracic spine) and 1245 cGy cm2 (lumbar spine) [24].
Like in the studies by Schils and Izadpanah et al. [24] also our research showed a statistically significant (p < 0.05) reduction of radiation exposure of patients and surgeons when applying the O-arm computer navigated procedure. Here DAP applied was on average 912.29 cGy/cm2 (O-arm group) and 1722.13 cGy/cm2 (control group) per procedure and 601.28 cGy/cm2 (O-arm group) vs. 1506.86 cGy/cm2 (control group) per vertebra.
From a practical point of view PV treatment is technically much more difficult in the treatment of thoracic than lumbar vertebrae. This is due to the anatomy of the thoracic vertebrae: smaller, vertically positioned vertebral pedicles, worse quality of intraoperative X-ray images, especially with C-arm fluoroscopy, and higher possibility of severe neurological deficits due to the risk of thoracic spinal cord damage. These inconveniences usually increase the number of intraoperative fluoroscopy tests performed during the needle insertion into the vertebra. Therefore, we additionally performed a statistical analysis comparing the level of radiation dose between the thoracic and lumbar vertebrae in the O-arm group (lack of statistical significance) and dose of radiation between the thoracic and lumbar vertebrae in the C-arm group (also statistically non-significant). However, when comparing radiation dose levels between the O-arm and C-arm groups, the difference is statistically significant for thoracic vertebrae (p = 0.000005) and also for lumbar vertebrae (p = 0.00019) (Table III).
Table III
Values of intraoperative radiation exposure in thoracic and lumbar vertebrae
From results summarized in Table III another interesting conclusion can be drawn, that the use of the O-arm technique with neuronavigation is particularly useful in the treatment of thoracic vertebrae, which reflects our results – a very low radiation dose (528.14 cGy/cm2) and even lower than in the case of lumbar vertebrae (668.06 cGy/cm2), in contrast to the control group, where the dose was very high in the treatment of thoracic vertebrae (1666.72 cGy/cm2) and higher than in the lumbar vertebrae (1420.24 cGy/cm2).
In conclusion, percutaneous vertebroplasty guided by O-arm combined with neuronavigation significantly reduces the radiation exposure of surgeons compared to the C-arm fluoroscopy procedure.