Introduction
Atherosclerosis, a chronic progressive disease relating the immune or inflammatory response to vascular tissue damage, can affect coronary, cerebral, and lower extremities arteries [1–3]. Arteriosclerosis obliterans (ASO) of the lower extremities represents a spectrum of disorders causing normal limb dysfunction and may be associated with an increased risk for claudication, resting pain and gangrene [4, 5]. Open surgery, endovascular therapy, and physical therapy (e.g. exercise) are the major methods to cure ASO [6]. Nevertheless, the risk of patients developing post-angioplasty restenosis within 1 year after the these treatments, such as balloon angioplasty procedures and stent implantations, is high [7]. The damage caused by ASO includes a series of highly specific cellular and molecular responses; changes in the phenotype of vascular cells, such as platelets or inflammatory cells, that release growth factors and cytokines to induce vascular smooth muscle cells (VSMCs) are observed after the initial arterial injury [8].
The mechanism of ASO is associated with VSMCs’ proliferation and migration, which are stimulated by the inflammatory response [9, 10]. Significant differences are found in the expression levels of some microRNAs, such as miR-21, miR-22, miR-24 and miR-1298, between ASO and normal arteries [11]. Among these microRNAs, miR-21 plays a critical role in various biological processes in the cardiovascular system, regulating heart and vascular function; it is also associated with proliferation, migration and apoptosis of VSMCs [12, 13]. miR-21 is one of the well-characterized miRNAs: previous research has shown that the expression of miR-21 was remarkably elevated in cardiovascular cells and such overexpression has also been found in other cardiovascular diseases, such as obliterative vascular disease, ventricular hypertrophy and ischemic heart disease [13, 14].
Mitogen-activated protein kinases (MAPKs) were found to be potential mediators in the inflammatory response and atherosclerosis [15, 16]. Pathways of two important members of the MAPK family, ERK1/2 and AKT, were proved to regulate multiple biological processes including cell survival, proliferation and glycogen metabolism in VSMCs [17, 18].
Recently studies reported that inhibition of the PI3K/AKT signaling pathway also inhibited atherosclerosis progression [19, 20] but enhanced the stability of atherosclerotic plaques [21]. Compared with normal vessel tissues, the tissues from atherosclerotic lesions showed a significant increase in the expression of ERK1/2 protein [22, 23]. It is still unclear, however, if miR-21 and ERK and AKT pathways are involved in the regulation of ASO. In this study, we developed an ASO injury rat model to determine whether the expression of miR-21 was stimulated under the ASO condition. A cell model was used to investigate whether AKT and ERK1/2 pathways were involved in the miR-21-induced proliferation and migration of human aortic smooth muscle cells (HASMCs). We also attempted to determine whether expression of AKT and ERK1/2 could modulate the miR-21-induced alteration of cell shape in HASMCs.
Material and methods
Cell culture and cell transfection
HASMCs were maintained in DMEM medium (Sigma) supplemented with 10% fetal bovine serum (FBS) (Hyclone), penicillin (100 U/ml) (Sigma) and streptomycin (100 mg/ml) (Sigma). Cells were incubated at 37°C in a humidified atmosphere of 5% CO2 and 95% air, and only cells within the subculture passage 4 to 10 were used in subsequent experiments. Cells were grown to 80–90% confluence. Serum-free medium was replaced and the cells were incubated for 24 h to reach 90% confluence to induce quiescence. A miR-21 mimic was synthesized and was transfected into HASMCs using Lipofectamine 2000 (Invitrogen) transfection reagent. Cells were incubated for 6 h in serum-free medium, which was then replaced with complete growth medium. Transfection was initiated when the cells reached 70–90% confluence. Cells were incubated at 37°C for 2–4 days and using a microscope to confirm the efficiency of transfection. RNA and protein were extracted and collected after transfection and analyzed by western blot and real-time PCR.
Western blot analysis
HASMCs or the vessel tissue were lysed and extracted. The protein concentrations were determined by the bicinchoninic acid (BCA) assay (Thermo Fisher Scientific). Protein extract was separated by 10% or 6% SDS-PAGE and transferred to a nitrocellulose or polyvinylidene difluoride (PVDF) membrane. After 1 h incubation in blocking solution, membranes were immersed using primary antibodies (anti-AKT (Abcam), anti-pAKT (Abcam), anti-ERK1/2 (Abcam), anti-pERK1/2 (Abcam)) at 4°C overnight. Subsequent to washing with TBST, the membranes were incubated with 1 : 5000 dilution secondary antibodies for 1 h. The membranes were detected with the enhanced chemiluminescence (ECL) system. Density of each band was analyzed using Image J.
Cell apoptosis assays
Apoptotic cells were detected with a terminal deoxyribonucleotidyl transferase-mediated dUTP-digoxigenin nick-end labeling (TUNEL) assay kit (Abcam). Nuclear morphology was stained by 4,6-diamidino-2-phenylindole (DAPI) staining (Sigma). HASMCs were washed with PBS and fixed with 4% paraformaldehyde at 4°C, followed by cell membrane permeabilization with Triton X-100. The apoptosis rate was quantified by counting cells with apoptotic nuclei by using a fluorescence microscope.
Quantitative real-time PCR
HASMCs or the vessel tissues were harvested, and quantitative stem-loop RT-PCR for detection of miR-21 level was determined with SYBR Green Master Mix kits (Thermo Fisher Scientific). The expression of miR-21 was normalized to rRNA U6. Single-strand cDNA was synthesized from total RNAs using a Reverse Transcription System Kit (Promega, Madison, WI).
Immunohistochemistry staining
The rat normal and ASO arterial tissues were fixed with paraformaldehyde (PFA) in PBS for 30 min. Transverse sections (10 μm) of frozen vessels were prepared using coated slides, which were fixed with PFA (4%). Tissue sections were permeabilized with Triton-X-100 (0.2%) in PBS for 10 min. Primary and secondary antibodies were applied to the tissue at room temperature for 1 h. The primary antibodies used were as follows: miR-21 (Abcam), AKT (Abcam), ERK1/2 (Abcam) and actin (Abcam). The slides were washed with PBS after co-incubation with each antibody and were then analyzed under a fluorescence microscope. The animal experiments were approved by the Affiliated Hospital of Guangdong Medical University.
Proliferation, invasion and migration assay BrdU incorporation
HASMCs were transfected with miR-21 mimics or miR-21 inhibitor. Cells were incubated either in the presence or absence of AKT and ERK inhibitors at 37°C. Cell proliferation was measured at 24 h, 48 h and 72 h after transfection with a BrdU assay kit, respectively. The culture medium was removed and replaced with BrdU labeling solution, after which the HASMCs were incubated in the BrdU labeling solution at 37°C. The cells were fixed with paraformaldehyde and stained with DAPI after washing with PBS, and then measured by immunocytochemistry.
Transwell assay and wound healing assay
HASMCs were plated with a density of 2 × 104 cells/well in the top compartments inserted into the wells and complete medium (600 μl) was added to the lower reservoir chamber. Subsequent to incubation at 37˚C for 6–24 h, the cells without migration were scraped off whereas the cells that migrated to the bottom side were stained with 10% Giemsa. The migration ability was expressed by counting the stained cells on the bottom surface.
The wound closure assay was performed to evaluate the ability of the cells to migrate. The HASMCs were plated with a density of 1 × 106 cells/well in 12-well plates and were then scratched. The average distance that cells moved from the edge of the scratch towards the center was measured after 24 h.
Statistical analysis
Statistical analysis was performed using GraphPad Prism. Data were reported as means ± (SEM) from three independent experiments. Statistical analysis was performed using the t-test to compare differences between two groups. Multiple group comparisons were analyzed using ANOVA with a post hoc test. P < 0.05 was considered to indicate a statistically significant result.
Results
miR-21 expression levels were increased in ASO arteries
To determine whether miR-21 expressed in the ASO arteries, quantitative RT-PCR was used to measure the expression level of miR-21 in ASO and normal arterial walls. As shown in Figure 1 A, compared with the normal arteries, the miR-21 expression level was significantly increased in the ASO arteries. Subsequently, the expression pattern of miR-21 in different layers of ASO artery was investigated. As shown in Figure 1 B, the expression level of miR-21 in the media of ASO artery was significantly higher than that in the layer of adventitia and intima. Immunofluorescence results confirmed that the expression of miR-21 was situated in the media of the ASO artery (Figure 1 C). The results indicated that miR-21 was upregulated in the ASO artery.
Figure 1
Characteristics of miR-21 expression and AKT and ERK 1/2 activation in ASO artery. A – The miR-21 expression level in normal and ASO artery, determined by qRT-PCR. B – miR-21 expression in different layers of ASO arteries, determined by qRT-PCR. C – Immunohistochemistry of miR-21 expression level in ASO sections, miR-21 (red) and α-actin (green). D – Tissues from ASO artery were resolved by SDS-PAGE and protein levels were detected using anti-AKT antibody (1 : 5000), anti-p-AKT antibody (1 : 2000), anti-ERK antibody (1 : 5000), anti-p-ERK antibody (1 : 2000) and anti-β-actin antibody (1 : 10000). Statistical analysis was performed according to Material and methods (data are reported as mean ± SD, Student t-test, **p < 0.01, #p < 0.05, ##p < 0.01)
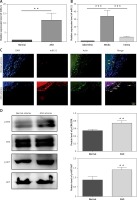
The expression of AKT and ERK1/2 were aberrant in the ASO arteries
In order to determine whether expression of AKT and ERK1/2 was involved in the pathogenesis of ASO, expression levels of p-AKT and p-ERK1/2 were compared between normal and ASO arteries using western blot. As shown in Figure 1 D, the expression levels of p-AKT and p-ERK1/2 were significantly increased in the ASO artery. The results demonstrated that expression of p-AKT and p-ERK1/2 was involved in the pathogenesis of ASO.
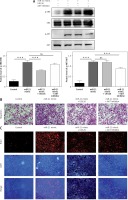
Down-regulation of AKT and ERK1/2 expression inhibited the miR-21-induced proliferation and migration of HASMCs
To determine whether miR-21 was involved in the regulation of HASMC motility, HASMC was transfected with miR-21 mimics. Quantitative real-time RT-PCR was used to measure the expression level of miR-21 in miR-21 mimic and miR-21 mimic + AKT or ERK1/2 inhibitor groups. It was found that HASMC migrated through uncoated porous filters (8.0 µm) in the Transwell migration assay. As shown in Figure 2 B, transfection of miR-21 mimics significantly accelerated the migration rate of HASMCs co-incubated with miR-21 mimics compared to that of the control group. To investigate whether miR-21 could promote cells growth, proliferation of cells in which miR-21 had been enhanced by the BrdU incorporation assay was examined. As expected, the cells transfected with miR-21 mimics showed a significantly increase in the number of BrdU-positive cells compared to the control group (p < 0.05; Figure 2 C). To explore the roles of the AKT and ERK1/2 signaling pathway in miR-21-induced proliferation and migration, HASMCs were transfected with miR-21 mimics and were then exposed to AKT or ERK inhibitors (Figure 2 A). It was found that co-treatment with miR-21 mimics and AKT or ERK inhibitor could attenuate the miR-21-induced proliferation and migration of HASMCs (p < 0.05; Figure 2 B). Inhibition of the AKT or ERK activity might protect HASMC from miR-21-induced aberrant growth and motility. The results indicated that miR-21 overexpression promoted HASMC migration and proliferation. Furthermore, inhibition of AKT or ERK activity attenuated the aforementioned miR-21-induced effects.
Figure 2
Effects of AKT and ERK1/2 expression inhibited miR-21-induced proliferation and migration of HASMC. A – Western blot showed suppression of AKT and ERK activation by using AKT inhibitor and ERK inhibitor U0126. B – HASMC migration rate was examined after transfection with miR-21 mimic and co-treatment with AKT or ERK1/2 inhibitor, determined by wound healing assay and Transwell assay. C – Representative images illustrate that BrdU expression was significantly decreased in miR-21 mimic + AKT or ERK1/2 inhibitor groups. Statistical analysis was performed according to Material and methods (data are reported as mean ± SD, Student t-test, **p < 0.01, #p < 0.05, ##p < 0.01)
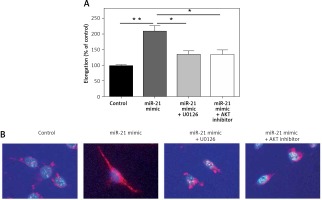
Down-regulation of AKT and ERK1/2 expression altered miR-21 induced cell shape alteration in HASMCs
To explore whether miR-21 promoted increasing expression of HASMCs α-actin, miR-21 mimics were transfected into HASMCs to induce overexpression of miR-21. Cell shape alteration was determined with a confocal microscope and the cell elongation ratio was examined by measuring the longest cord and the longest axis perpendicular to it. Cell cytoskeleton was visualized by immunocytostaining of smooth muscle α-actin (Figure 3 B). Results from cell elongation assays showed that the cells co-treated with miR-21 mimics and AKT or ERK inhibitor could attenuate the miR-21-induced high elongation ratio of HASMCs (p < 0.05; Figures 3 A). The results indicated that miR-21 not only played an important role in promoting HASMCs α-actin, but also altered HASMCs’ shape via AKT and ERK signaling pathways.
Figure 3
MiR-21 altered cell shape in HASMCs via AKT and ERK1/2 signaling pathway. A – Immunostaining was used to alter the cell shape followed by examination of cell elongation ratio. B – Results of smooth muscle α-actin (red), and cell nuclear stain (blue) under confocal microscope demonstrated that overexpression of miR-21 altered cell shape and co-treatment with ERK or AKT inhibitor could be used to reverse the effect. Statistical analysis was performed according to Material and methods (data are reported as mean ± SD, Student t-test, **p < 0.01, #p < 0.05, ##p < 0.01)
Discussion
Although miRNAs have been well established as key mediators in multiple physiological and pathological processes of cardiovascular disease, the mechanism remains unclear. Recent studies concluded the pathology of ASO from clinical tests, but only ASO in relatively late stages could be detected based on the pathology known. As a result, finding a biomarker for detecting ASO in earlier stages is important. It is well accepted that miR-21 may play important roles in diverse vascular remodeling [24]. Wang et al. reported that the level of miR-21 significantly increased in ASO arteries [11]. However, signaling pathways involved in the regulation of proliferation and migration of VSMCs in ASO remain unclear. In this study, we firstly determined that the inhibition of AKT and ERK1/2 expression could attenuate miR-21-induced proliferation and migration of HASMCs. It was also found that the expression level of miR-21 significantly increased in the ASO arteries compared to that in the normal arteries. This finding was supported by quantitative RT-PCR and immunofluorescence staining results (Figures 1 A, C). Expression of miR-21 was detected in different layers of the ASO artery, and it was found to be located in the media, combined with smooth muscle cells. The results indicated that aortic smooth muscle cells might partially contribute to the overexpression of miR-21 in the pathogenesis of ASO.
Activation of AKT and activation of ERK1/2 kinase are two important pathways regulating metabolism, proliferation and migration of VSMCs [25]. In the process of atherosclerosis, cell death and survival of VSMCs correlate with atherosclerotic plaque instability and stability, respectively [26]. Regulatory molecules including ERK1/2, AMP kinase, PI3K, Akt and mTOR can maintain cellular quality of lipoprotein metabolisms, such as of obesity and atherosclerosis [27]. In the present study, it was observed that activation of p-AKT and p-ERK1/2 was significantly increased in the ASO artery, consistent with previous studies which found that angiogenesis was induced by miR-21 by the activation of AKT and ERK1/2 signaling pathways [28]. The present study added new proof to show that the expression of p-AKT and p- ERK1/2 was involved in the pathogenesis of ASO.
To further study the role of miR-21 in HASMC motility, miR-21 mimics that can specifically stimulate the expression of miR-21 in HASMC were used and quantitative RT-PCR was used to detect the expression level of miR-21. It was found that overexpression of miR-21 significantly accelerated HASMC migration and proliferation rates, consistent with the previous study [13]. To explore the function of the AKT and ERK1/2 signaling pathways in miR-21-induced proliferation and migration, HASMCs were transfected with miR-21 mimics and were then exposed to AKT or ERK inhibitor. It was found that the inhibition of AKT or ERK1/2 could block both miR-21-induced migration and proliferation of HASMCs. The results confirmed that miR-21 functioned partially by activating AKT and ERK1/2 expression and restored the motility of HASMCs.
Recently studies demonstrated that miR-21 could regulate the smooth muscle α-actin protein rate and altered cell shape change by directly targeting TPM1 in HASMCs [11]. It is still unclear, however, if cell shape changes in HASMCs correlate with AKT or ERK signaling pathways. In this study, it was proposed that miR-21 modulated the cytoskeleton by reducing the contraction of actin filaments via activating AKT and ERK1/2 expression. The cells co-treated with miR-21 mimics and AKT or ERK inhibitor could attenuate the miR-21-induced high elongation ratio of HASMCs. In summary, miR-21 not only played an important role in promoting HASMCs’ α-actin, but also altered HASMCs’ cell shape via AKT and ERK signal pathways.
In conclusion, in the present study, we demonstrated that the expression of miR-21 in HASMCs is an aspect worth considering for the treatment of cardiac diseases. We found that miR-21 was significantly increased in the ASO process and promoted the motility of HASMCs. Irregular function of HASMCs serves a critical role in arteriosclerosis and ASO. The characteristics of miR-21 in the present study allow great progress in the understanding of the pathogenesis of ASO. The inhibition of AKT or ERK activity could attenuate the miR-21-induced artery dysfunction effects as well as altering HASMCs’ cell shape. The research provided a potential therapeutic approach and new targets for preventing and treating ASO in earlier stages.


