Purpose
Worldwide, cancer of corpus uteri is the sixth most commonly diagnosed malignancy in women (4.4% of the total cases of cancer) [1]. Uterine carcinosarcoma (UCS), uterine clear cell carcinoma (UCCC), and uterine papillary serous carcinoma (UPSC) are uncommon histological sub-types, and constitute less than 15% of all endometrial cancers [2-5]. These histologies represent an aggressive disease, characterized by local relapse and rapid metastatic dissemination, leading to a poor prognosis [2-5]. The distribution is predominant in older non-obese women and primarily affects African-American women [2-5].
Due to their rarity, prospective randomized studies have been hampered and thus, there is a lack of robust recommendations to support the optimal adjuvant treatment strategy in such patients. In this scenario, we conducted a systematic review to define the role of adjuvant vaginal interventional radiotherapy (VIRt) in UCS, UCCC, and UPSC managements. Also, the aim of this review was to propose it as a useful clinical tool for further expert consensus.
Material and methods
A systematic research using PubMed and Cochrane library was performed to identify relevant articles evaluating the efficacy of VIRt in patients with early-stage UCS, UCCC, and UPSC, published from January 1990 to July 2020. A search in ClinicalTrials.gov and PROSPERO was conducted to detect ongoing and recently completed trials and systematic reviews, respectively. The following medical subject headings (MeSH) and keywords were used: “endometrial neoplasms”, “brachytherapy”, “endovaginal radiotherapy”, “uterine clear cell carcinoma”, “uterine carcinosarcoma”, and “uterine papillary serous carcinoma”. The search was restricted to English language publications, including humans. Studies were eligible if patients had a proven early-stage UCS, UCCC, and UPSC treated with adjuvant VIRt. Review articles, conference papers, case reports, commentaries, letters, and book chapters were not included. Reference lists of previously published reviews were explored. Four independent investigators with > 5 years of experience on endometrial cancer screened citations in titles and abstracts in order to identify appropriate papers. Eligible citations were retrieved for full-text reviews. Any disagreement was resolved by a consensus. When two articles appeared to report results with overlapping data, only the data representing the most recent publication were included in the review. The following parameters were obtained from all included studies: first author’s surname, publication year, sample size, median age, treatment, and clinical outcomes, such as disease-free survival (DFS), local control (LC), overall survival (OS), cancer-specific survival (CSS), and toxicity rates.
Results
We organized the results by a histological sub-type. The following key areas were covered: 1) uterine carcinosarcoma (UCS), 2) uterine clear cell carcinoma (UCCC), 3) uterine papillary serous carcinoma (UPSC). In total, there were no prospective studies. Based on selection criteria, only data from the VIRt treatment arms were extracted and considered for the analysis.
Uterine carcinosarcoma (UCS)
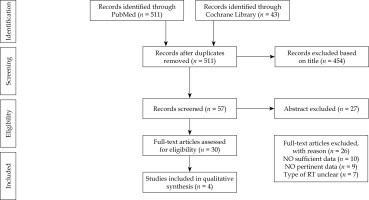
Literature search resulted in 511 articles. After exclusion by title and abstract, and after exclusion of conference papers, case reports, commentaries, letters, book chapters, reviews, and non-English language publications, 30 papers were assessed via full text for eligibility. Of these, 26 articles were excluded due to insufficient data, leaving 4 studies assessing the clinical efficacy of VIRt in DFS (Figure 1). All studies were retrospective [6-9]. Our review identified 432 patients with an average age of 67 years (range, 27-96 years) and a median follow-up of 35 months (range, 24-48 months). All patients underwent total abdominal hysterectomy and bilateral salpingo-oophorectomy (TAH/BSO). Pelvic lymph nodes dissection was performed in 79% to 97% cases, while para-aortic lymphadenectomy in 53% to 68% cases. Peritoneal cytology was performed in most patients (94-98%), and omental sampling in 42% to 78% cases. The presence of positive lymphovascular invasion was reported in 191 patients out of 412 patients analyzed. Adjuvant chemotherapy was administered in 55% to 74% of patients, with intervals of at least one week between chemotherapy and VIRt. Chemotherapy consisted of platinum/taxane doublets. Adjuvant exclusive VIRt was delivered to the proximal two-thirds of the vagina and prescribed at an average dose of 21 Gy (range, 21-30 Gy) in 3-5 fractions, at a depth of 0.5 cm or at vaginal surface of the upper half of vagina. Studies including VIRt plus chemotherapy as a treatment, reported a vaginal cuff and pelvic relapse rate that ranged from 0% to 9% and from 12% to 45%, respectively. The 2- to 5-year average LC was 91% (range, 74.2-96%), DFS 88% (range, 82-94%), and OS 79% (range, 53.8-84.3%). The average 5-year CSS was 70% (range, 70-94%) and G3-G4 toxicity was 0% (Table 1). In case of adjuvant treatment based on external beam radiotherapy plus VIRt and chemotherapy, both vaginal and pelvic relapse rate were 0-1%. Moreover, the distant relapse rate ranged from 6% to 87% of patients (Table 2). Stage IB-II were associated with poor outcomes, including presence of lymphovascular invasion, age > 65 years, and cervical involvement. Concurrent chemo-radiotherapy and VIRt (alone or boost) were associated with higher DSS and freedom from vaginal recurrence, respectively [6, 8, 9]. UCS showed worse outcomes when compared to UPSC, but these differences were not statistically significant in patients treated with chemotherapy plus VIRt [6, 7].
Table 1
Characteristics and outcomes of the included studies
| Author [ref] | Period | Study | Histology, no. of pts | Treatment, no. of pts | Age (years), median (range) | VIRt Gy/fr | CT % of pts | FU months (range) | DFS | LC | Toxicity G3-G4 | OS | CSS |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Desai et al. [6] | 2000-2011 | Obser retrospect | UPSC – 112 Stage I – 97 Stage II – 15 UCS – 60 Stage I – 55 Stage II – 5 | CT alone – 12 CT + RT – 4 VIRt – 21 VIRt + CT – 111 RT – 6 Obs – 18 | 67 (27-96) | 21/3 fr | 74% | 48 (3-139) | 5-y UPSC – 80% 5-y UCS – 52% VIRt + CT Actuarial relapse rate UPSC 5-y – 15% UCS 5-y – 29% | 5-y UPSC – 96% 5-y UCS – 91% | N.A. | VIRt + CT All-cause mortality UPSC – 10% UCS – 22% | 5-y UPSC – 89% 5-y UCS – 70% VIRt + CT Disease-related mortality UPSC 5-y – 10% UCS 5-y – 22% |
| Brown et al. [7] | 2000-2013 | Obser retrospect | UCS – 33 Stage I – 32 Stage II – 1 | VIRt – 15 VIRt + CT – 18 | 68 (47-93) | 21/3 fr 24/6 fr | 55% | 24 (9-90) | 2-y – 66% | 2-y VC – 94% 2-y PC – 87% | 6% | 2-y – 79% | |
| Gunther et al. [8] | 1990-2011 | Obser retrospect | UCS – 155 Stage I – 98 Stage II – 11 Stage III – 46 | VIRt – 21 VIRt + CT – 12 RT – 13 RT + CT – 12 Obs – 45 | 67 (36-92) | 30/5 fr 15/3 fr | 60% | 42 | All stage 5-y DSS – 57.2% I stage – 60.9% II stage – 53.3% All stage CT + RT 5-y DSS – 67.5% RT 5-y DSS – 39% Stage I EBRT – 64.9% No EBRT – 45.5% No CT – 59.6% CT – 50.9% | All stage 5-y PDC – 74.2% RT 5-y PDC – 85% No RT/VIRt – 70% I stage RT – 100% No RT – 76% (p = 0.05) | N.A. | All stage 5-y – 48.6% I stage – 53.8% II stage – 30% | |
| Guttmann et al. [9] | 1990-2014 | Obser retrospect | UCS – 118 Stage I – 100 Stage II – 18 | CT – 19 RT – 9 (11 + CT) VIRt – 7 (15 + CT) RT + VIRt – 8 RT + VIRt + CT – 12 Obs – 37 | 67 | 21/3 fr 12/2 fr | 58% | 28 (1-244) | 3-y RT – 57.33% 3-y CT/RT – 69% | 3-y RT – 64.3% 3-y RT + CT – 91.7% 3-y VIRt – 96% | N.A. | 3-y RT – 47.9% 3 y RT + CT – 84.3% | |
| Tétreault- Laflamme et al. [10] | 2006-2012 | Obser retrospect | Stage I UPSC – 30 UCCC – 6 Mixed – 21 | CT + VIRt – 32 CT + VIRt + EB – 25 | 62 (47-83) 65 (52-79) | 31.05/3 fr 21/3 fr | 100% | 35 (7-62) 61 (12-96) | 3-y I – 88% 3-y I – 84% | 3-y I – 97% 3-y I – 100% | 0% 1 pt GI | 3-y I – 100% 3-y I – 94% | |
| Qu et al. [11] | 2004-2015 | Obser retrospect | Stage IA UPSC – 266 Mixed – 112 UCCC – 36 | Obs – 209 CT alone – 27 RT – 10 CT + RT – 19 VIRt – 41 RT + VIRt – 14 RT + CT + VIRt – 36 | 68 (60–76) | 35/5 fr 21/3 fr | 34% | 32 (0-142) | Any treatment 5-y DFS – 77% 5-y Obs – 70% 5-y CT – 80% 5-y RT – 38% 5-y CT + RT – 92% 5-y VIRt – 9.5% 5-y RT + VIRt – 30% 5-y RT + VIRt + CT – 85% | Any treatment 5-y LC – 94% 5-y Obs – 82% 5-y CT – 91% 5-y RT – 68% 5-y CT + RT – 100% 5-y VIRt – 97% 5-y RT + VIRt – 93% 5-y RT + VIRt + CT – 100% | N.A. | Any treatment 5-y OS – 81% 5-y Obs – 83% 5-y CT – 88% 5-y RT – 53% 5-y CT + RT – 89% 5-y VIRt – 3% 5-y RT + VIRt – 37% 5-y RT + VIRt + CT – 90% | Any treatment 5-y CSS – 92% 5-y Obs – 90% 5-y CT – 93% 5-y RT – 86% 5-y CT + RT – 89% 5-y VIRt – 93% 5-y RT + VIRt – 100% 5-y RT + VIRt + CT – 90% |
| Barney et al. [12] | 1998-2011 | Obser retrospect | Stage I UPSC – 74 UCCC – 21 Mixed – 8 | VIRt – 68 VIRt + CT – 35 | 66 (44-86) | 21/3 fr | 34% | 36 | 5-y UPSC – 92% 5-y UCCC – 87% | 5-y UPSC – 100% 5-y UCCC – 100% 5-y mixed – 80% | N.A. | 5-y UPSC – 85% 5-y UCCC – 83% | |
| Townamchai et al. [13] | 2005-2012 | Obser retrospect | Stage IA/IB – 34 Stage II – 3 UPSC – 26 UCCC – 9 Mixed – 2 | VIRt – 7 VIRt + CT – 30 | 63 (52-80) | 24/6 fr | 81% | 24 (2-71) | 2-y – 89.3% 2-y UPSC – 90% 2-y UCCC – 83% | 2-y – 93.5% 2-y UPSC – 95% 2-y UCCC – 100% | 0% | 2-y – 100% | |
| DuBeshter et al. [14] | 1995-2001 | Obser retrospect | Stage I/II UPSC – 16 UCCC – 8 | VIRt – 24 | 67 (27-96) | 30-40/4 fr | N.A. | 31 (10-63) | 5-y – 75% | 5-y IA-IC – 100% 5-y II – 50% | N.A. | 5-y – 81% | |
| Armbruster et al. [15] | 2000-2016 | Obser retrospect | Stage IA/IB – 90 Stage II – 20 UCCC – 44 Mixed – 68 | RT ± CT – 10 CT alone – 12 CT + VIRt – 111 VIRt – 21 Obs – 12 | 65 (34-94) | N.A. | 40% | 44 (3-264) | Median RFS – 4.32 y No adj therapy – 2.77 y Adj therapy – 4.71 y (N.S.) | N.A. | 0% | Median – 9.80 y No adj therapy – 0.52 y Adj therapy – 11.29 y (N.S.) | N.A. |
| Desai et al. [17] | 1996-2010 | Obser retrospect | Stage IA – 56 Stage IB – 10 Stage II – 11 UPSC – 51 Mixed – 26 | VIRt + CT – 61 VIRt – 16 | 67 (27-88) | 21/3 fr | 79% | 62 (6-182) | 5-y – 88% 5-y IA – 91% 5-y IB-II – 78.5% | 5-y IA – 96.2% 5-y IB-II – 87.7% | N.A. | 5-y – 90% 5-y IA – 94.3% 5-y IB-II – 80% | |
| Robbins et al. [18] | 1998-09 | Obser retrospect | Stage I – 45 Stage II – 2 UPSC – 56 | CT + VIRt – 33 CT + VIRt + EB – 23 | 64 (59-83) 69 (59-81) | 37.5/6 fr | 100% | 55 (18-151) | 5-y VIRt – 80.5% 5-y VIRt + EB – 67.3% | 5-y VIRt – 87.1% 5-y VIRt + EB – 72% | N.A. | 5-y IA – 71.9% 5-y II – 45.5% | 5-y I – 84.8% 5-y I-II – 78.9% |
| Donovan et al. [19] | 2000-2014 | Obser retrospect | Stage IA UPSC – 63 | Obs – 32 VIRt + CT – 21 | 68 (45-90) | 31.5/3 fr | 100% | 38 (3-114) | 5-y – 73.5% 5-y – 74.7% | 5-y – 88% 5-y – 98% | N.A. | 5-y – 79.6% 5-y – 95% | |
| Alektiar et al. [20] | 2000-2006 | Obser retrospect | Stage IA – 7 Stage IB – 12 Stage IC/II – 6 UPSC – 25 | VIRt + CT – 25 | 67 (53-80) | N.A. | 88% | 30 (17-79) | 5-y IA – 83% 5-y IB – 92% 5-y IC-II – 83% | 5-y – 100% | 0% | 5-y – 88% | |
| Kiess et al. [21] | 2000-2009 | Obser retrospect | Stage IA – 30 Stage IB – 4 Stage II – 7 UPSC – 29 Mixed – 12 | VIRt + CT – 41 | 67 (51-80) | 18-21/3 fr | 95% | 58 (11-119) | 5-y I – 88% 5-y II – 71% | 100% | 10% | 5-y I – 93% 5-y II – 71% | |
| Mahdi et al. [22] | 2000-2012 | Obser retrospect | Stage IA UPSC – 92 Mixed – 11 | Obs – 43 VIRt – 12 CT alone – 21 CT + VIRt – 27 | 66 (49-90) | N.A. | 46% | 47.7 | 5-y – 88.5% 5-y – 95.6% 5-y – 77% 5-y – 95.6% | 5-y – 89.1% 5-y – 97.4% 5-y – 89.1% 5-y – 97.4% | N.A. | 5-y – 90.5% 5-y – 93.3% 5-y – 85.5% 5-y – 93.3% |
[i] no. – number, Pts – patients, VIRt – vaginal interventional radiotherapy, Gy – Gray, UCS – uterine carcinosarcoma, UCCC – uterine clear cell carcinoma, UPSC – uterine papillary serous carcinoma, CT – chemotherapy, FU – follow-up, Obs – observation, DFS – disease-free survival, LC – local control, G – grading, OS – overall survival, adj – adjuvant, CSS – cancer-specific survival, Obser retrospect – observational retrospective study, RT – radiotherapy, EB – external beam radiotherapy, y – years, fr – fraction, RFS – recurrence-free survival, VC – vaginal control, PC – pelvic control, N.A. – not available
Table 2
Characteristics of relapse rate of the included studies
| Author [ref] | Histology, no. pts | Treatment | FU months (range) | Local control | Vaginal cuff relapse rate | Pelvic relapse rate | Distant relapse rate |
|---|---|---|---|---|---|---|---|
| Desai et al. [6] | UPSC – 112 Stage I – 97 Stage II – 15 UCS – 60 Stage I – 55 Stage II – 5 | CT alone – 12 CT + RT – 4 VIRt – 21 VIRt + CT – 111 RT – 6 Obs – 18 | 48 (3-139) | UPSC 5-y – 96% UCS 5-y – 91% | UPSC – 4% UCS – 9% | UPSC – 10% CS – 13% PA UPSC – 5% PA UCS – 16% Peritoneal UPSC – 9% UCS – 18% | UPSC – 14% UCS – 38% |
| Brown et al. [7] | UCS – 33 Stage I – 32 Stage II – 1 | VIRt – 15 VIRt + CT – 18 | 24 (9-90) | 2-y VC – 94% 2-y PC – 87% | Vaginal alone – 0 Vaginal + distant metastases – 6% | 12% | 6% |
| Gunther et al. [8] | UCS – 155 Stage I – 98 Stage II – 11 Stage I/II – 46 | VIRt – 21 VIRt + CT – 12 RT – 13 RT + CT – 12 Obs – 45 | 42 | 5-y PDC – 74.2% | VIRt ± CT – 9% RT + CT – 0% Obs – 25% | VIRt ± CT – 45% RT + CT – 0% Obs – 17% No RT stage I – 21% No RT stage II – 25% | Site – PA VIRt ± CT – 27% RT + CT – 100% Obs – 42% RT + CT – 0% |
| Guttmann et al. [9] | UCS – 118 Stage I – 100 Stage II – 18 | CT – 19 RT – 9 (11 + CT) VIRt – 7 (15 + CT) RT + VIRt – 8 RT + VIRt + CT – 12 Obs – 37 | 28 (1-244) | 3-y RT – 64.3% RT + CT – 91.7% VIRt 3-y – 96% | Obs – 44% CT – 20% RT – 29% RT + CT – 1% | Obs – 6% CT – 0% RT – 29% RT + CT – 0% | Obs – 44% CT – 40% RT – 43% RT + CT – 87% |
| Tétreault- Laflamme et al. [10] | Stage I UPSC – 30 UCCC – 6 Mixed – 21 | CT + VIRt – 32 CT + VIRt + EB – 25 | 35 (7-62) 61 (12-96) | 3-y I – 97% 3-y I – 100% | VIRt – 6% VIRt + EB – 0% | VIRt – 7% VIRt + EB – 11% | VIRt – 0% VIRt + EB – 11% |
| Qu et al. [11] | Stage IA UPSC – 266 Mixed – 112 UCCC – 36 | Obs – 209 CT alone – 27 RT – 10 CT + RT – 19 VIRt – 41 RT + VIRt – 14 RT + CT + VIRt – 36 | 32 (0-142) | Any treatment 5-y LC – 94% 5-y Obs – 82% 5-y CT – 91% 5-y RT – 68% 5-y CT + RT – 100% 5-y VIRt – 97% 5-y RT + VIRt – 93% 5-y RT + VIRt + CT – 100% | All strategies – 12% VIRt + CT – 3% RT + CT – 0% RT + CT + VIRt – 0% | All strategies – 12% | All strategies – 54% Obs 5-y – 14% |
| Barney et al. [12] | Stage I UPSC – 74 UCCC – 21 Mixed – 8 | VIRt – 68 VIRt + CT – 35 | 36 | UPSC 5-y – 100% UCCC 5-y – 100% Mixed – 80% | 2% | 2% | 4% |
| Townamchai et al. [13] | Stage IA/IB – 34 Stage II – 3 UPSC – 26 UCCC – 9 Mixed – 2 | VIRt – 7 VIRt + CT – 30 | 24 (2-71) | 2-y – 93.5% UPSC 2-y – 95% UCCC 2-y – 100% | 2.7% | 5% PA – 11% Peritoneal – 2.7% | None |
| DuBeshter et al. [14] | Stage I/II UPSC – 16 UCCC – 8 | VIRt – 24 | 31 (10-63) | 5-y IA – 100% 5-y IB – 100% | 4% | None Ascites – 4% | 4% |
| Armbruster et al. [15] | Stage IA/IB – 90 Stage II – 20 UCCC – 44 Mixed – 68 | RT ± CT – 10 CT alone – 12 CT + VIRt – 111 VIRt – 21 Obs – 12 | 44 (3-264) | N.A. | Pelvic – 24% | Nodal – 22% | 54% |
| Desai et al. [17] | Stage IA – 56 Stage IB – 10 Stage II – 11 UPSC – 51 Mixed – 26 | VIRt + CT – 61 VIRt – 16 | 62 (6-182) | 5-y IA – 96.2% 5-y IB/II – 87.7% | 3% | 6% PA – 6% Peritoneal – 8% | 10% |
| Robbins et al. [18] | Stage I – 45 Stage II – 2 UPSC – 56 | CT + VIRt – 33 CT +VIRt + EB – 23 | 55 (18-151) | VIRt 5-y – 87.1% VIRt + EB 5-y – 72% | Stage I VIRt – 3% VIRt + EB – 0% Stage II VIRt – 0% VIRt + EB – 0% | Stage I VIRt – 10% VIRt + EB – 1% Stage II VIRt – 50% VIRt + EB – 56% | Stage I VIRt – 16% VIRt + EB – 7% Stage II VIRt – 50% VIRt + EB – 56% |
| Donovan et al. [19] | Stage IA UPSC – 63 | Obs – 32 VIRt + CT – 21 | 38 (3-114) | 5-y – 88% 5-y – 98% | Obs – 12.5% VIRt + CT – 3% | Obs – 6% VIRt + CT – 0% | Obs – 12.5% VIRt + CT – 9% |
| Alektiar et al. [20] | Stage IA – 7 Stage IB – 12 Stage IC/II – 6 UPSC – 25 | VIRt + CT – 25 | 30 | 5-y – 100% | None | 4% PA – 12% Peritoneum – 8% | 12% |
| Kiess et al. [21] | Stage IA – 30 Stage IB – 4 Stage II – 7 UPSC – 29 Mixed – 12 | VIRt + CT – 41 | 58 (11-119) | 100% | None | 3% PA – 7% Peritoneum – 7% | 7% |
| Mahdi et al. [22] | Stage IA UPSC – 92 Mixed – 11 | Obs – 43 VIRt – 12 CT alone – 21 CT + VIRt – 27 | 47.7 | 5-y – 89.1% 5-y – 97.4% 5-y – 89.1% 5-y – 97.4% | Obs/CT – 11% VIRt ± CT – 2.6% | Obs/CT – 3% VIRt ± CT – 5.1% | Obs/CT – 20% VIRt ± CT – 10.3% |
[i] no. – number, Pts – patients, VIRt – vaginal interventional radiotherapy, Gy – Gray, UCS – uterine carcinosarcoma, UCCC – uterine clear cell carcinoma, UPSC – uterine papillary serous carcinoma, CT – chemotherapy, FU – follow-up, Obs – observation, DFS – disease-free survival, LC – local control, G – grading, OS – overall survival, CSS – cancer-specific survival, RT – radiotherapy, EB – external beam radiotherapy, y – years, fr – fraction, RFS – recurrence-free survival, VC – vaginal control, PC – pelvic control, PDC – pelvic disease control, PA – para-aortic, N.A. – not available
Uterine clear cell carcinoma (UCCC)
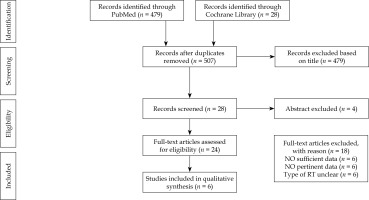
The literature search resulted in 507 articles. After the initial screening of titles and abstracts, 24 papers were identified. Of these, 18 articles were excluded due to insufficient data, leaving 6 studies assessing the clinical efficacy of VIRt in DFS (Figure 2). All studies were retrospective [10-15]. In total, 335 patients (UCCC – 124, mixed – 211), with an average age of 65 years (range, 62-67 years) and a median follow-up of 35 months (range, 24-61 months) were enrolled. All but one paper [15] reported TAH/BSO, pelvic and para-aortic lymph nodes dissection, peritoneal cytology, omental sampling, and the presence of positive lymphovascular invasion. In those studies including not unique and specific histology, data concerning UCCC, UPSC, and mixed histologies were not separately considered. In 112 patients with mixed or pure UCCC endometrial carcinoma, pelvic and para-aortic lymph nodes dissection was reported in 81% and 62%, respectively [15]. Adjuvant chemotherapy was administered in 34% to 100% of patients, with intervals of at least one week between chemotherapy and VIRt. Chemotherapy consisted of platinum/taxane doublets. Adjuvant VIRt was prescribed typically to the proximal two-thirds of the vagina at an average dose of 21 Gy (range, 21-31.5 Gy) in 3-6 fractions, at a depth of 0.5 cm or at vaginal surface of the upper half of vagina. When specifically reported for UCCC histology, the 2- to 5-year average LC was 100%, DFS 83% (range, 82-90%), and OS 93% (range, 83-100%),with G3-G4 toxicity of 0% (Table 2) [12, 13].
In the study by Armbruster et al., in 112 patients with early-stage UCCC, the median OS was longer in patients who received adjuvant treatment compared to those who did not receive adjuvant therapy, but this difference was not statistically significant [15]. On multivariate analysis, age ≥ 70, positive lymphovascular invasion [15] stage IB-IC [12, 14], and UCCC vs. mixed histology were associated with poorer outcomes. Better outcomes were associated with adjuvant therapy, i.e. chemotherapy plus radiotherapy and/or VIRt [11, 15]. In selected UCCC and UPSC stage I patients (with pelvic and aortic lymph nodes staging, omental sampling, and peritoneal washing), exclusive VIRt (with or without chemotherapy) can be an option, in particular in stage IA patients (without myometrial invasion) [10, 12, 14].
Uterine papillary serous carcinoma (UPSC)
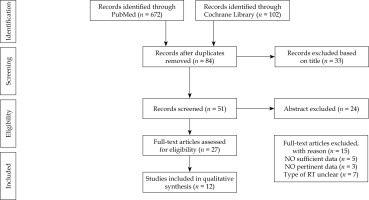
Literature search details of the VIRt role in UPSC were described in our previous systematic review [16]. Here, we presented a brief overview of the main data (Figure 3). A total of 12 retrospective studies were finally analyzed on the basis of their relevance to the scope of this review [6, 10-14, 17-22]. Our review identified 1,092 patients (UPSC – 866, mixed – 226), with an average age of 67 years (range, 27-96 years), 924 with FIGO stage I disease (when specifically described 608 patients with stage IA and 46 with stage IB), and 66 patients with FIGO stage IC-II, with a median follow-up of 36 months (range, 21-62 months). All patients received TAH/BSO. Pelvic lymph nodes dissection was performed in most cases (79-100%), and para-aortic lymphadenectomy was reported in 23% to 100% of cases. Peritoneal cytology ranged from 79% to 100%, and omental sampling ranged from 38% to 100%. The presence of positive lymphovascular invasion was reported in a median of 26% of patients (range, 4.7-70%). Adjuvant chemotherapy was administered in 34% to 100% of patients, with intervals of at least one week between chemotherapy and VIRt, mainly consisted of platinum/taxane doublets. Adjuvant VIRt consisted of an average dose of 21 Gy (range, 12-37.5 Gy) in 3 fractions prescribed at a depth of 0.5-0.7 cm or at vaginal surface. Details of clinical outcomes are listed in Tables 1 and 2.
In patients treated with VIRt plus chemotherapy, the studies reported a median rate of vaginal cuff, pelvic, and distance relapse of 3%, 4%, and 8%, respectively.
The average 5-year LC was 97% (range, 87.1-100%), DFS 84% (range, 74.7-95.6%), OS 93% (range, 71.9-100%), and CSS 89% (range, 78.9-94%). There was no evidence of severe toxicity. In a large series of 404 stage IA patients (UPSC – 266, UCCC – 26, mixed – 112) better outcomes (5-year DFS 85%, LC 100%, OS 90%) were associated with chemotherapy plus radiotherapy and VIRt [11]. On the contrary, 2 studies showed similar results when chemotherapy plus VIRt was compared to chemotherapy plus radiotherapy and VIRt. The authors concluded that VIRt alone seemed to be as effective as radiotherapy in early-stage UPSC treated with surgery and adjuvant chemotherapy, because no significant clinical benefit of adding pelvic radiotherapy was recorded [10, 18].
Four studies reported a comparison between VIRt plus chemotherapy and exclusive VIRt, and all of them showed very low vaginal recurrence rate with 2-5-year LC ranged between 93% to 100% and 2-5-year OS from 83% to 100%, with not statistical impact of chemotherapy in surgically staged patients [12, 13, 17, 22]. Another study confirmed a local control in 96% of cases with exclusive adjuvant VIRt, and a risk of recurrence (13%) increased according to the stage [14]. Finally, three studies showed a median rate of vaginal cuff, pelvic, and distance relapse of 0%, 4%, and 9% in 117 UPSC and 12 mixed early-stage patients treated with VIRt and chemotherapy [19-21].
In these studies, when multivariate analysis was performed, myometrial invasion, lymphovascular invasion, and stage IB-IC/II were risk factors for poor DFS and/or OS, while chemotherapy had no impact on outcomes; however, in another large series of patients, both adjuvant VIRt and chemotherapy were associated with better local control and DFS [11-14, 17, 22]. Lymphadenectomy was associated with longer progression-free survival (PFS) [22].
Discussion
Radical surgery with total hysterectomy and bilateral salpingo-oophorectomy is the current standard treatment for non-metastatic UCS, UCCC, and UPSC [5, 23, 24]. The role of complete staging with systemic lymphadenectomy and omentectomy should be considered in early-stage diseases due to its improvement in survival and a decrease of overall recurrence rate, especially in UPSC [25-28].
Owing to the rarity of these tumors, there is no established consensus regarding adjuvant therapy. All suggestions for post-operative treatment are based on retrospective studies, limited by small simple size and/or heterogeneity of patients’ characteristics, especially due to the infrequent subset of these histologies.
Uterine carcinosarcoma (UCS)
Chemotherapy seems to improve OS and DFS in patients affected by advanced-stage UCS, but its role in early-stage is unclear [29-32]. A recent study evaluated the role of chemotherapy, radiotherapy, and a combination of both after surgery in 3,447 women with stage I UCS. The 5-year OS for no treatment was 51.9% compared to 67.6% for chemotherapy, 57.9% for radiotherapy, and 73.9% for chemotherapy plus radiotherapy. Adjuvant chemotherapy (95% CI: 0.52-0.79%, p < 0.001) and chemotherapy plus radiotherapy (95% CI: 0.42-0.64%, p < 0.001) were predictors for improved survival [33]. The efficacy of adjuvant radiotherapy in terms of LC and OS in patients with UCS has been reported in several retrospective studies [34-36]. However, the same results were not found in two prospective clinical trials [37, 38]. The phase III trial conducted by Gynecologic Oncology Group (GOG-150) enrolled 232 patients with stages I-IV primary UCS of the uterus or cervix. They were randomized to receive whole abdominal irradiation or chemotherapy (cisplatin/ifosfamide/mesna). The authors did not find a significant difference in relapse rate or OS between the two regimens, maybe due to relatively small sample size. This study has some limitations: whole abdominal radiation therapy is now rarely used, and the technique and dose prescribed are now considered obsolete. Moreover, carboplatin/paclitaxel is now the most commonly chosen first-line adjuvant chemotherapy regimen for UCS [37].
The European Organization for Research and Treatment of Cancer (EORTC) conducted a phase III trial with patients affected by stage I-II uterine sarcoma (including leiomyosarcoma, carcinosarcoma, and endometrial stromal sarcoma). Women were randomized to receive no therapy or pelvic radiotherapy as adjuvant treatment. No statistically significant differences were found in median PFS and OS between the two arms. However, patients affected by UCS and treated with radiotherapy had a better LC compared to those who did not receive adjuvant treatment. It should be noted that these histological sub-types, which used to be grouped as uterine sarcomas, are no longer grouped together [38].
In a series of 118 UCS early-stage patients, an advantage in LC has been demonstrated in patients treated with combined radiation therapy (external beam plus VIRt) and chemotherapy [9]. In contrast, in a study by Gunther et al., external beam radiotherapy was associated with better loco-regional control and time to distant metastatic spread without significant impact to OS compared to VIRt alone [8].
In a retrospective study of patients affected by stage I-II UCS, external beam radiotherapy and VIRt were compared in terms of effectiveness. VIRt alone did not have worse OS and DFS compared to external beam radiotherapy. The authors suggested VIRt as an alternative to pelvic radiotherapy in patients adequately surgically staged and undergone adjuvant chemotherapy [39]. Moreover, in SEER retrospective analysis, the use of VIRt alone showed a substantial increase over time, from 4.5% during 1988-1999 to 12.5% during 2005-2010. In other studies, the “cuff and chemo” approach had the largest survival benefit in women with node-negative UCS [6, 40].
In conclusion, currently, there is a lack of consensus on the optimal therapeutic strategy in early-stage UCS [6]. Moreover, there is no clinical trial that suggests a survival benefit with adjuvant radiotherapy (external beam and/or VIRt) with or without chemotherapy. Results of several recent studies provided evidence regarding the importance of adequate surgical staging and comparative effectiveness of external beam radiotherapy versus VIRt. Despite a worse acute toxicity profile, VIRt and chemotherapy seemed to be an effective alternative strategy to external beam radiotherapy in UCS early-stage patients [39-41].
Uterine clear cell (UCCC) and uterine papillary serous carcinomas (UPSC)
A multi-institutional study published in 2009 demonstrated the role of systemic therapy for early-stage UPSC. PFS and CSS for chemotherapy-treated patients were more favorable than for those who did not receive chemotherapy. Five-year PFS and CSS rates were 81.5% and 87.6% in chemo-radiotherapy group, 64.1% and 59.5% in radiotherapy alone group, and 64.7% and 70.2% in observational group, respectively [42]. Platinum-based chemotherapy improved the DFS and OS rates in patients with stage I UPSC, and vaginal cuff radiation provided LC [43]. PORTEC-3 trial investigated the role of chemotherapy plus radiotherapy versus radiotherapy alone in patients affected by high-risk endometrial cancer. The combined treatment significantly improved the 5-year failure-free survival (FFS) compared to radiotherapy alone (95% CI: 75.5% vs. 68.6%, p = 0.067). No significant difference in OS was observed (95% CI: 81.8% vs. 76.7%, p = 0.213). Patients with UPSC had much benefit from combined treatment in terms of FFS compared to the other histologies (HR = 0.44, 95% CI: 0.31-0.60%). However, this benefit was not statistically significant because of the small number of UPSC cases [44].
Contrary to previous reported data, international guidelines and studies suggested exclusive radiotherapy as adjuvant treatment for patients with non-endometrioid uterine carcinoma, and VIRt may be considered an option in early-stage UPSC and UCCC [12, 13, 16, 17, 45, 46]. A national large-scale study of cancer-specific outcomes demonstrated that VIRt was the only treatment to significantly improve cancer-specific outcomes in stages I-II cohort, and that both VIRt and chemotherapy appeared beneficial in early-stage patients with papillary-serous histology [47].
Qu et al. observed a significant advantage from VIRt as adjuvant treatment associated with chemotherapy in improving local control, even if failed in detection of a dose-response relationship between radiation dose and local control [11]. The first study available in literature comparing external beam radiotherapy and VIRt in 87 women with stage I UPSC or UCCC, described the same effectiveness of VIRt alone as external beam radiotherapy and VIRt when associated with surgery and adjuvant chemotherapy [10].
In their multi-institutional retrospective review, Madhi et al. confirmed a lower risk of vaginal recurrence in patients with early non-invasive UPSC, who underwent VIRt ± chemotherapy compared to patients who received chemotherapy alone or no treatment (2.6% vs. 10.9%). In contrast, they did not detect an improvement in PFS or OS [22]. These results were confirmed in other retrospective studies [17, 20, 21].
A retrospective analysis of adjuvant therapy for patients affected by stage I-II UPSC showed an increasing use of VIRt and chemotherapy, and a decrease of external beam radiotherapy from 1998 to 2012. In these patients, VIRt and chemotherapy were associated with a reduction of mortality not seen with the use of external beam radiotherapy [48]. Donovan et al. advocates the use of chemotherapy and VIRt for patients with stage IA UPSC, given the low-rate of pelvic recurrence [19].
Finally, a recent phase III trial showed no different outcomes in terms of recurrence-free survival (RFS) and OS, in patients affected by high-risk endometrial cancer randomized to receive external beam radiotherapy vs. chemotherapy plus VIRt. The 60-month RFS was 0.76 (95% CI: 0.70-0.81%) for radiotherapy and 0.76 (95% CI: 0.70-0.81%) for chemotherapy plus VIRt (HR = 0.92, 90% CI: 0.69-1.23%). The 60-month OS was 0.87 (95% CI: 0.83-0.91%) for radiotherapy and 0.85 (95% CI: 0.81-0.90%) for chemotherapy plus VIRt (HR = 1.04, 90% CI: 0.71-1.52%) [49].
There is no consensus regarding the optimal dose-fractionation schedule for the delivery of VIRt. The 2014 American Brachytherapy Society (ABS) update survey reported that 7 Gy for three fractions is the most common schedule for post-operative VIRt alone. Nevertheless, a wide variation in dose schedules was noted [50]. In this review, the median total surface dose was 21 Gy in 3 fractions (range, 15-37.5 Gy in 3-6 fractions).
Several studies confirmed a lower if any acute gastrointestinal and genitourinary toxicity in the VIRt treatment with or without chemotherapy [7, 10, 13, 15, 20, 21], resulting in a good quality of life as well as PORTEC-2 trial [48-52]. To note, PORTEC-2 was a randomized trial for women with intermediate-risk endometrioid uterine cancer, comparing pelvic external beam radiotherapy with VIRt in terms of LC and toxicity [53].
These data are consistent with a recent review, in which authors established that acute endovaginal toxicity may occur in less than 20.6% of cases, while late vaginal toxicity G1-G2 may appear in less than 27.7% and G3-G4 in less than 2% [54]. Furthermore, limited available evidence suggested that there is no difference in terms of acute and late toxicity between low-dose-rate and high-dose-rate VIRt [55], or using high-dose-rate VIRt short schedule [56].
Overall, these results are informative, but not completely generalizable. The major limitation of this review was the retrospective nature of the included studies. Based on the assumption that retrospective studies start with a “low quality” rating [57], the usefulness of adjuvant VIRt was difficult to interpret. Certainly, the surgical approach remains the gold standard in this setting of patients.
Conclusions
This systematic review suggests the potential role of VIRt in the management of non-endometrioid carcinoma of corpus uteri. Overall, adjuvant VIRt seems to be helpful to improve clinical outcomes, especially DFS and LC. No definitive conclusions can be drawn, mainly due to the lack of adequately powered randomized studies. At present, adjuvant VIRt may be justified, based on its positive effect on local control, while minimizing severe toxicity. Undeniably, the proper treatment strategy in patients with non-endometrioid carcinoma of corpus uteri should be described in a prospective trial.