Introduction
Short stature affects approximately 3% of the population. It remains one of the main reasons for endocrinological consultations in outpatient clinics for children and one of the leading causes of hospitalization in pediatric endocrinology departments. According to Linsday et al., up to 80% of children referred to endocrinology clinics and endocrinology departments with short stature will finally be diagnosed as having idiopathic short stature (ISS) [1].
Idiopathic short stature is defined as a height more than two standard deviations (SD) below the mean for age and sex for the population group without evidence of systemic, endocrine, nutritional, or genetically determined disorders [2]. Children with ISS have normal birth weight and growth hormone (GH) secretion. Children with ISS were subdivided into two subgroups: familial short stature (FSS) and non-familial short stature (NFSS). When the patient is shorter than the population reference but remains within the target height (TH) calculated based on the parents’ height, it is described as FSS. When the patient is shorter compared to both the national reference and TH (defined as equal to or greater than a target height SDS of –1.28), the patient is diagnosed as NFSS. The NFSS group includes children with constitutional delay of growth and puberty (CDGP) [3]. Even though an endocrinologist has to exclude many different disorders to diagnose ISS, ISS remains a highly heterogeneous group with multiple potential pathophysiological mechanisms [4]. In the diagnostic workup for short stature, dysmorphic features must be excluded, but it is not clarified how an extended genetic diagnostic workup should be performed before the diagnosis of ISS. Endocrinologists agree that all girls should be tested for Turner syndrome, but there is no consensus about testing for SHOX gene mutations, which are responsible for 2.5% of ISS [5].
Treatment with recombinant human growth hormone (rhGH) for ISS patients was approved by the Food and Drug Administration (FDA) in the United States in 2003 for children shorter than –2.25 SD. However, it is still unavailable in many countries, such as Poland, where rhGH therapy is not registered for children with ISS. A potentially beneficial combination of rhGH and a gonadotropin-releasing hormone analog (GnRHa) and a combination of rhGH and aromatase inhibitors (AIs) are considered [6].
Attempts to estimate the final height (FH) in patients with ISS remain inaccurate. Patients and parents worry about the potential impact of short stature on patients’ quality of life (QoL) [7]. Findings from a systematic literature review suggest that adults and children with short stature may experience poorer QoL than those with normal stature [8–12]. Still, it should be highlighted that in some cases, findings were inconsistent, even among studies of patients with the same condition [7–12]. Several studies suggest that parents of children with ISS, in comparison to parents of children with growth hormone deficiency, had a more significant burden [8, 9].
Although several studies indicated that more significant height gain is associated with improved QoL, the current literature does not provide enough evidence to mark a potential height gain threshold beyond which QoL benefit is no longer gained [7].
The main aim of this study was to identify parameters useful in predicting final adult height in patients with ISS and to make appropriate therapeutic decisions for these patients.
Material and methods
We analyzed the spontaneous FH, correlated FH with calculated predicted adult height based on bone age (PAH) assessed in the prepubertal period, and in available cases with further bone age analysis, and correlated FH with the height standard deviation score (HSDS) at ages 4, 6, and 9 years, based on the available retrospective data.
Study group
The study group consisted of 16 patients (13 girls, 3 boys) with ISS (HSDS < –2) diagnosed in the pediatric endocrine department, who were regularly seen for follow-up. Patients were enrolled according to the following recruitment criteria:
Height below < –2 SD or the difference between height and target height more than –1 SD.
Growth hormone secretion above 10 ng/ml in at least one of the two performed stimulation tests – after clonidine, glucagon, insulin, arginine, or after sleeping.
Absence of chronic diseases that could be the cause of short stature, such as celiac disease, inflammatory bowel diseases, cystic fibrosis, chronic renal diseases, decompensated hypothyroidism, asthma, hematooncological diseases, heart defects, autoimmune diseases of the musculoskeletal system, and exclusion of chronic glucocorticoid therapy.
Turner syndrome was excluded, and there were no signs suggesting other genetically determined disorders.
Previously, the PAPP-A2 gene analysis, free IGF-1, IGFBP-5, and acid-labile subunit (ALS) analysis were performed in these patients, and data have already been published [13]. In each patient we evaluated free IGF-1, IGFBP-5, and ALS concentrations, and genetic analysis of the PAPP-A2 gene (Gene ID: 60676, 1q25.2) at the sites of the mutations described to date, i.e. c.3098C>T, p.Ala1033Val in exon 8, and p.D643fs25* in exon 3 and analysis of exons 3, 4, and 5, which encode the fragment of the catalytic domain with the active site of PAPP-A2 protein, in the absence of the known mutations were performed [13]. The genetic analysis was carried out after ISS diagnosis. The patient numbers remain the same as in the previous article; patients 10, 11, 13, and 14 have not yet completed the growth process, hence the lack of data on their final height [13].
Auxological analysis
Birth weight (BW) and birth length (BL) were expressed as SDS using reference data by Fenton et al. [14]. Small for gestational age (SGA) was described as birth weight or birth length < –2 SD. The attending physician assessed the patient’s postnatal height and weight. Height measurements were taken in the same room using a Harpenden-type measuring device, with an accuracy of 1 mm; three measurements were taken, and the mean was calculated. Some auxological parameters were obtained from the patient’s medical history. The attending physician assessed the patients’ parents’ height using a Harpenden-type measuring device, with an accuracy of 1 mm; three measurements were taken, and the mean was calculated. The auxological analysis of height and weight referred to the Polish population growth chart developed by Palczewska and Niedzwiecka, and was expressed as centile and SDS [15]. Height standard deviation score (HSDS) was calculated based on the formula:
HSDS = patient’s height – 50th centile height / ½ × (50th centile height – 3rd centile height)
where 50th centile height and 3rd centile height correspond to the 50th centile and 3rd percentile positions for a given age and gender, respectively.
The target height (TH) based on parents’ height was estimated according to the Tanner formula [16]:
TH = (father’s height + mother’s height /2) ±6.5 cm.
For boys, 6.5 cm was added, and for girls, 6.5 cm was subtracted. The target height was expressed as height, and target height SDS (TH SDS). Target height SDS was calculated based on the formula:
TH SDS = TH – 50th centile height / ½ × (50th centile height – 3rd centile height)
Where TH corresponds to the parental prediction calculated based on the Tanner formula, and 50th centile height and 3rd centile height correspond to the 50th centile and 3rd centile positions at 18 years of age for a given gender, respectively.
The corrected height standard deviation score (corrHSDS) was defined as the difference between FH SDS and TH SDS.
The puberty stage was evaluated according to the Tanner scale [18, 19].
Bone age assessment
Bone age (BA) was assessed according to Greulich and Pyle and predicted adult height (PAH) based on bone age was estimated using the Bayley and Pinneau formula [17, 20].
Predicted adult height in some cases was estimated three times (PAH I, II, and III), based on available data. Data were not available for all patients for each analysis. In all patients, the first bone age analysis was performed in the prepubertal age. In the group with the second and third bone age assessments, in most cases, the second analysis was performed 2 years after the first, and the third two years after the second. The second bone age assessment concerned 9 of 16 patients for whom results were available; conclusions are formulated based on this subgroup for the entire group.
In the analysis, PAH I, II, and III are not equal to HSDS at the ages of 4, 6, and 9. We chose to analyze HSDS at the ages of 4, 6, and 9 years because during those periods of life, most patients had auxological analysis according to the national health program.
Statistical analysis
To assess the normality of the distributions, given the small sample size, skewness and kurtosis were evaluated. To examine the linearity of the relationships, scatterplots were generated and visually inspected. The linear relationship between the variables was assessed using Pearson’s correlation coefficient (r). A significance level of α = 0.05 was adopted for all tests. Analyses were conducted using IBM SPSS Statistics.
Bioethical standards
The local Ethics Committee approved the study (approval number 134/18).
The study is based on the routine medical documentation and routine medical care on the patients and did not need any additional procedures, so the formal agreement taken from patients/ parent/caregiver was not provided.
Results
None of the patients achieved the parental prediction calculated from the parents’ height, regardless of the parents’ final height. In three cases, the difference between FH and TH was 1 cm or less (Table I). The parental prognosis of seven patients was –1.8 SD or less, which corresponds to the 3rd percentile on the centile chart developed by Palczewska and Niedzwiecka. Also, this group did not achieve TH. The mean FH SDS was –2.85, and the mean corrFH SDS was –1.51. The average difference between the patients’ FH and TH was 9.2 cm, for boys 16.6 cm, and girls 7.5 cm. In almost all patients, FH was similar to PAH; in Patient 1, the difference between FH and PAH was larger (Table I).
Table I
Final height (FH), corrected final height standard deviation score (Corr FH SDS), predicted adult height (PAH), standard deviation score of height (HSDS) at the age 4, 6, and 9 years, and FH – TH
[i] F – girl; M – boy; FH – final height; FH SDS – standard deviation score of final height; TH – target height; TH SDS – standard deviation score of target height; PAH I, II, III – respectively, subsequent values of estimated height based on the bone age, HSDS at age 4, 6, and 9 – standard deviation score of height at the age 4, 6, and 9 years
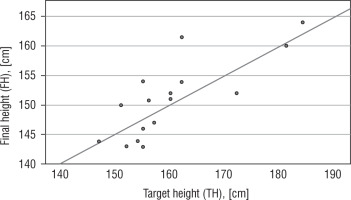
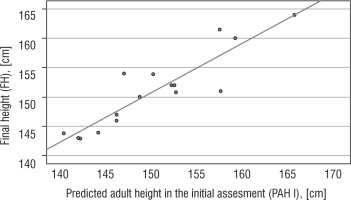
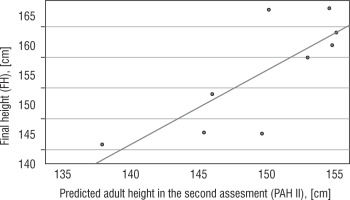
Statistical analysis showed a positive correlation between FH and TH (r = 0.790, p < 0.001; Fig. 1) and between FH SDS and TH SDS (r = 0.534, p = 0.033). There was also a positive correlation between FH and PAH I (r = 0.905, p < 0.001), PAH II (r = 0.780, p = 0.13), and PAH III (r = 0.923, p = 0.009). It has to be highlighted that the subgroup in which we estimated PAH III consists of 5 out of 16 patients. Moreover, a positive correlation was found between FH SDS and HSDS at the age of 4 (r = 0.749, p = 0.033) and 6 years of age (r = 0.946, p ≤ 0.001; Figures 2 and 3). A significant correlation was not found between FH SDS and HSDS at the age of 9 (r = 0.583, p = 0.1).
Discussion
The natural history of patients with ISS indicates varying degrees of difficulty in achieving the growth potential resulting from the height of their parents. Over the years, numerous studies have been published summarizing the FH of children with ISS [21–24]. In the group of patients with FSS, the average FH was lower than TH, which was 2.1 cm in the case of boys and 0.6 cm in the case of girls. In this group, FH SDS was similar to the SDS of prepubertal height. In our study group, patients 2, 12, 15, 16, 17, 19, and 20 can be described as FSS due to FH – TH. In that subgroup, average FH was lower than TH by 3.8 cm, which is greater than what has been reported in the literature. However, not all patients in this subgroup had FH SDS similar to the prepubertal HSDS (Table I).
In the NFSS group, the mean FH was less than TH by 8.3 cm for boys and 6.8 cm for girls, but the mean FH SDS was more significant than the initial HSDS. It was observed that the average FH in the group of boys was several centimeters (2–5 cm) less than the PAH of the patients. In contrast, in the group of girls, the average FH was similar to PAH based on the Bayley and Pinneau method. In our study group, patients 1, 3, 4, 5, 6, 7, 8, 9, and 18 can be described as NFSS based on FH – TH. In that subgroup, the average FH was lower than TH by 13.35 cm, in girls by 11.7 cm, and in boys by 16.6 cm, which is much greater than previously reported values for FH in the group of patients with NFSS. In the girls’ subgroup, similar to the previous results, the average FH was similar to PAH. In the boys’ subgroup, the average FH was several centimeters lower than the PAH (Table I). A significant delay in bone age beyond two years results in a substantial overestimation of FH, and a minor delay in bone age leads, in turn, to an underestimation of FH, which may result in differences between the average FH and estimated PAH [4, 25, 26].
The mean FH SDS in the study group was lower than the mean initial HSDS. Similarly to previous reports, in our study group, FH did not differ significantly from PAH I in the group of girls. Among boys with completed growth in our group, unlike previous reports, FH did not differ substantially from the PAH I. This fact may be related to the small size of the study group and the fact that patients with significant height deficiency were included in the study group.
Undoubtedly, further observation of patients with ISS in a larger group is necessary to improve the prediction of FH in ISS and further ease decision-making for initiating potential and available treatment – especially in the light of poorer QoL in groups of patients and patients’ parents with ISS. Wang et al. in a retrospective study compared the results of rhGH therapy, the combination of rhGH and GnRHa therapy, and the combination of rhGH and AIs therapy [6]. They found that compared with PAH, the adult height in all three groups improved significantly [6]. In the group treated with rhGH, patients did not exceed TH, and in the group treated with rhGH and AIs, patients could reach or even exceed TH [6]. In the group treated with GnRHa and rhGH, more prolonged therapy was necessary to obtain a clinically significant increase in height [6]. Finding a parameter that could precisely estimate FH would be helpful during a decision-making process, especially when a clinician considers available growth-promoting therapies.
Conclusions
Predicted adult height based on prepubertal bone age and height SDS at the ages of 4 years and 6 years can help to estimate the final adult height. It may be helpful in further facilitating decision-making about initiating potential and available treatment in patients with idiopathic short stature.