Summary
Future research should focus on evaluating the long-term clinical impact and economic benefits of such systems across diverse healthcare settings, including resource-limited regions. Expanding interoperability with electronic health records and incorporating artificial intelligence for dynamic, guideline-based updates could further enhance precision.
Introduction
Cardiovascular diseases (CVD) represent one of the most significant global health challenges, responsible for approximately 20% of deaths in the United States and generating immense economic burdens [1]. Secondary prevention strategies, including antithrombotic agents, statins, and antihypertensive therapies, have been shown to reduce recurrent vascular events by 20–30% [2]. However, despite the widespread availability of evidence-based treatments, a substantial proportion of patients with dyslipidemia fail to achieve optimal low-density lipoprotein cholesterol targets, highlighting persistent gaps in treatment adherence and prescription accuracy [3]. This underscores the urgent need for innovative strategies to enhance medication safety and efficacy in CVD management. One promising approach involves the integration of intelligent digital platforms into clinical workflows. Electronic pre-prescription review systems, such as the Prescription Pre-Audit Intelligent Decision System, have demonstrated significant potential in reducing prescription errors by automatically screening for inappropriate dosages and drug interactions before dispensing [4]. These systems operate by cross-referencing prescriptions with up-to-date drug guidelines, clinical research, and safety protocols, ensuring alignment with evidence-based standards [4]. Studies have shown that such platforms not only improve medication appropriateness but also minimize the need for post-prescription corrections, thereby streamlining clinical workflows [5]. Furthermore, restricted prescriptive authority and pre-approval mechanisms – key components of antimicrobial stewardship – have been successfully adapted to CVD management, reinforcing the value of structured oversight in preventing medication errors [6].
The foundation of these intelligent systems lies in evidence-based medicine (EBM), which synthesizes high-quality research to guide clinical decision-making. Systematic reviews and meta-analyses have played a pivotal role in consolidating best practices for CVD prevention, such as the Mediterranean Diet, which has been associated with reduced risks of coronary artery disease, stroke, and cardiovascular mortality [7]. Additionally, large-scale trials such as REDUCE-IT have reinforced the importance of targeted lipid-lowering therapies in high-risk populations [1]. Beyond pharmacological interventions, lifestyle modifications – including physical activity, smoking cessation, and avoidance of nonsteroidal anti-inflammatory drugs – have been emphasized as critical components of secondary prevention [8]. Artificial intelligence and digital therapeutics are further transforming CVD care by enabling real-time, data-driven interventions. Machine learning algorithms, for instance, can analyze electrocardiograms to detect subtle cardiovascular abnormalities that may otherwise require specialized imaging [9]. Similarly, natural language processing tools are being used to enhance clinical supervision by extracting diagnostic insights from electronic health records [10]. These advancements align with the growing emphasis on digital health strategies to overcome traditional barriers in healthcare delivery [11].
Aim
This study evaluates the application of an EBM intelligent supervision platform for pre-prescription review in the secondary prevention of CVD. By leveraging automated decision-support tools, the platform aims to optimize medication appropriateness, reduce adverse cardiovascular events, and improve patient satisfaction – building upon prior innovations in digital health, clinical decision support systems, and evidence-based preventive cardiology. The findings may offer a scalable model for enhancing the quality and safety of CVD management in clinical practice.
Material and methods
Study subjects
We selected 140 patients requiring secondary prevention medication for CVD admitted to our hospital between May 2023 and June 2024 as the study subjects.
Inclusion criteria: (1) diagnosed with CVD (meeting at least one of the following conditions), including ST-segment elevation or non-ST-segment elevation myocardial infarction, stable or unstable angina, post-percutaneous coronary intervention or coronary artery bypass grafting, ischemic stroke or transient ischemic attack, intermittent claudication, carotid artery stenosis ≥ 50%, or other peripheral artery diseases; (2) high-risk populations (manifested as left ventricular ejection fraction ≤ 40%, diabetes with target organ damage, or chronic kidney disease stages 3-5); (3) populations recommended for guideline-directed medications, including those requiring long-term antiplatelet therapy (e.g., aspirin, P2Y12 inhibitors), statin therapy (for those with unachieved LDL-C targets), ACEI/ARB (e.g., in cases of hypertension, heart failure, or diabetic nephropathy), or β-blockers (e.g., post-myocardial infarction or heart failure); (4) age > 18 years.
Exclusion criteria: (1) absolute contraindications such as drug allergies, active bleeding or high bleeding risk, severe hepatic or renal dysfunction, pregnancy, or lactation; (2) life expectancy < 1 year; (3) coagulation disorders or immune system diseases; (4) cognitive impairments hindering effective communication; (5) missing clinical data (e.g., postoperative recovery indicators or follow-up records).
The patients were randomly allocated to the control group and the observation group using a random number table (n = 70 cases). No significant differences were observed in the baseline characteristics between the two groups (p > 0.05), indicating good comparability (Table I).
Table I
General data between the control and observation groups
Methods
The control group underwent traditional prescription review for secondary prevention of CVD. Clinical pharmacists manually reviewed the secondary prevention medications for CVD in the control group, assessing the appropriateness of medication use, rational medication rate, prescription modification rate, incidence of adverse cardiovascular events after medication, and satisfaction indicators for medication guidance.
In the observation group, an EBM approach was employed, utilizing an intelligent supervision platform for pre-prescription review of flagged data. (1) Construction of an EBM knowledge base: the CVD secondary prevention medication knowledge base was established based on the UK National Institute for Health and Clinical Excellence (NICE) guidelines regarding the consensus on CVD secondary prevention [12]. (2) Intelligent platform review: The intelligent supervision platform collected medication data from patients in the observation group, analyzed the data, and identified and addressed flagged items according to predefined rules, with the entire process guided by EBM principles. Following platform review, we assessed medication appropriateness, rational medication use rate, prescription modification rate, incidence of post-medication adverse CVD events, and satisfaction with medication guidance.
Observation indicators
Medication appropriateness score: The medication appropriateness scores before and after review were compared between the two groups. The assessment was conducted using the Chinese version of the Medication Appropriateness Index (MAI) [13]. The MAI is a widely used implicit measurement tool consisting of 10 questions, each with three options (A, B, and C). Option A indicates “appropriate,” B indicates “marginally appropriate”, and C indicates “inappropriate”, with assigned weights of 0, 0.5, and 1.0, respectively. Each question carries a different weight, ranging from 1 to 3 points. The score for each item is calculated by multiplying the option value by its weight, and the sum of the scores for all 10 items yields the medication rationality score for a single drug. The total medication rationality score is obtained by summing the scores of all drugs, with higher scores indicating greater irrationality. An MAI value of 0 indicates completely appropriate medication use.
Rational drug use rate: The rational drug use rate consists of the guideline compliance rate for drug types, the guideline compliance rate for dosage ranges, and the guideline compliance rate for combination therapy. We compared the rational drug use rates before and after review between the two groups.
Prescription modification rate: This includes both manual modification rate and platform modification rate. We reviewed and modified patient prescriptions through manual and platform-based audits, comparing the prescription modification rates between the control and observation groups before and after the audits.
Safety assessment after medication: The occurrence of cardiovascular adverse events, including myocardial infarction, unstable angina, heart failure, and arrhythmia, was monitored and compared between the two groups after 6 months of medication. Cardiovascular adverse events were diagnosed through electrocardiography and myocardial enzyme testing.
Patient satisfaction with medication: The patient satisfaction scores for medication were compared between the two groups after 6 months. The Treatment Satisfaction Questionnaire for Medication-II (TSQM-II) was used. This scale consists of four dimensions: medication effectiveness, adverse effects, convenience, and global satisfaction, totaling 11 items. Item 3 evaluates whether patients experienced adverse drug reactions, scored as “yes” or “no”. The adverse effects dimension employs a Likert 5-point scale, where scores from 1 to 5 represent “extremely dissatisfied” to “not dissatisfied,” respectively. The effectiveness, convenience, and global satisfaction dimensions use a Likert 7-point scale, with scores from 1 to 7 indicating “extremely dissatisfied” to “extremely satisfied”. Higher scores reflect greater patient satisfaction.
Statistical analysis
Data were processed using SPSS 26.0 statistical software. Measurement data were expressed as (mean ± SD), with intergroup comparisons performed by independent samples t-test and intragroup comparisons by paired t-test. Enumeration data were presented as frequency/percentage (%) and analyzed using the χ2 test. A p-value < 0.05 was considered statistically significant.
Results
Medication appropriateness scores
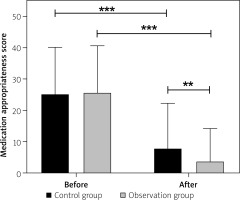
Prior to pre-prescription evaluation, the appropriateness of medication use was comparable across groups (p > 0.05). After review, both cohorts exhibited enhanced prescribing accuracy, with the observation group demonstrating a more pronounced improvement relative to controls (p < 0.05) (Table II, Figure 1). The MAI inappropriateness rate showed no significant difference between the control and observation groups before the review (p > 0.05). After review, the MAI inappropriateness rates in both groups were lower than those before the review, with the observation group exhibiting a significantly lower rate (15.7%) compared to the control group (25.7%) (p < 0.05) (Table II).
Table II
Comparison of appropriateness of medication use scores between the control and observation groups before and after review
Rational drug use rate
The rational drug use rate comprises guideline compliance rates for drug categories, dosage ranges, and combination therapies. No significant difference was observed between the two groups before the audit (p > 0.05). Compared with the pre-audit period, both groups showed significant post-audit improvements in guideline compliance rates for drug categories, dosage ranges, and combination therapies (p < 0.05). The improvement was significantly greater in the observation group than in the control group (p < 0.05) (Table III).
Table III
Comparison of rational drug use rates between the control and observation groups before and after review
Prescription modification rate
The prescription modification rate consists of the intelligent platform modification rate and the manual review modification rate. Before the review, there were no significant differences in the intelligent platform modification rate or the manual review modification rate between the two groups (p > 0.05). After the review, both the intelligent platform modification rate and the manual review modification rate were lower than those before the review in both groups (p < 0.05). Notably, after the review, the intelligent platform modification rate and the manual review modification rate in the observation group were significantly lower than those in the control group (p < 0.05). The prescription modification rate was calculated as: intelligent platform modification rate + (100% – intelligent platform modification rate) × manual review modification rate. We found that after the review, the prescription modification rate in the observation group decreased by 47.98% compared to that in the control group (Table IV).
Table IV
Comparison of prescription modification rates between the control and observation groups before and after review
Safety evaluation after medication
After medication, the incidence of cardiovascular adverse events in the observation group was 7.14%, significantly lower than that in the control group (21.43%, p < 0.05) (Table V).
Table V
Comparison of the incidence of cardiovascular adverse events between the control and observation groups after medication
Patient medication satisfaction score
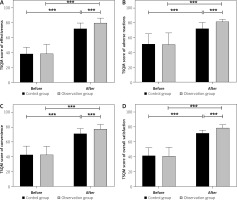
Before the review, there were no significant differences in the four dimensions of medication satisfaction scores between the two groups (p > 0.05). Following the review, overall satisfaction scores improved significantly in both groups compared to baseline (p < 0.05). Specifically, the observation group demonstrated higher scores in perceived treatment effectiveness, adverse reaction awareness, convenience, and overall satisfaction than the control group, with all differences reaching statistical significance (p < 0.05) (Table VI, Figure 2).
Table VI
TSQM-II scores for patient satisfaction between the control and observation groups (x– ± s, points)
Discussion
The integration of EBM into intelligent supervision platforms for pre-prescription review represents a significant advancement in optimizing secondary prevention strategies for CVD. As demonstrated in this study, the implementation of an EBM-based intelligent platform significantly improved medication appropriateness, reduced prescription errors, and enhanced patient outcomes compared to traditional review methods. These findings align with previous research indicating that automated prescription monitoring systems can effectively identify and rectify inappropriate prescriptions, thereby improving medication safety [14]. The observed reduction in adverse cardiovascular events further underscores the potential of such platforms in supporting evidence-based clinical decision-making for secondary prevention.
The current study demonstrates significant improvements in medication appropriateness, rationality, and patient outcomes through the use of an EBM-based intelligent supervision platform for pre-prescription review in CVD secondary prevention, which aligns with several findings from prior research. For instance, the observed reduction in inappropriate prescriptions and adverse events is consistent with the outcomes of the Antimicrobial Stewardship Program, which reported a decrease in multidrug-resistant infections and improved prescription compliance through systematic interventions [6]. This consistency underscores the broader applicability of structured, data-driven approaches in optimizing medication use. However, the current study innovates by specifically targeting cardiovascular secondary prevention, integrating real-time intelligent monitoring, and demonstrating a more pronounced improvement in rational medication rates compared to general antimicrobial stewardship programs.
Furthermore, the study’s emphasis on patient satisfaction and prescription rationality resonates with findings on telehealth and decision-support systems, which improved diagnostic accuracy and workflow efficiency [15]. However, the current research advances this by combining intelligent monitoring with evidence-based pre-review, achieving higher accuracy and applicability in a specialized clinical context. In contrast, some studies note that decision-support systems rarely reach “expert-level” performance [10], but the current platform’s success in reducing MAI inappropriateness rates and adverse events suggests a more robust integration of evidence-based protocols.
Overall, this study demonstrates that implementing an intelligent supervision platform for CVD secondary prevention can significantly improve prescription accuracy, reduce adverse cardiovascular events, and enhance patient satisfaction. By integrating real-time pre-prescription review with clinical decision support, the platform addresses key gaps in traditional prescription review systems, ensuring safer and more effective medication use. The innovation lies in its targeted application to cardiovascular medications, enabling disease-specific interventions that may yield more measurable benefits compared with broader, non-specific approaches. These findings align with broader trends in intervention efficacy while offering a scalable solution to optimize secondary prevention in CVD.
However, several limitations should be acknowledged. Due to the relatively small sample size, with only 70 patients in each group, subgroup analysis by gender was not conducted. In future studies, expanding the sample size would allow for exploration of potential differences in intervention effects between male and female patients. Additionally, the relatively short observation period limits our ability to assess long-term outcomes, particularly hard cardiovascular endpoints such as mortality or recurrent myocardial infarction.
Despite these limitations, this study highlights the critical role of evidence-based intelligent platforms in bridging the gap between guideline recommendations and real-world practice. The integration of pre-prescription review has been shown to reduce prescription modification rates and minimize adverse drug reactions, particularly in resource-rich settings where technological maturity supports system adoption. Nevertheless, AI-based approaches should be applied in a stratified manner, considering patient characteristics, healthcare resource availability, and cost-effectiveness; they are best positioned as tools to assist rather than replace clinicians.
Future research should focus on evaluating the long-term clinical impact and economic benefits of such systems across diverse healthcare settings, including resource-limited regions. Expanding interoperability with electronic health records and incorporating artificial intelligence for dynamic, guideline-based updates could further enhance precision. Multi-center prospective trials and patient-centered adaptations may improve both engagement and adherence, ultimately positioning this model as a blueprint for evidence-based interventions in other chronic disease management contexts.