Summary
Atherosclerosis, also known as hardening of the arteries, can affect multiple vascular beds simultaneously, but it is unclear whether plaque characteristics are similar across different regions. This study employed advanced computed tomography imaging (multidetector computed tomography angiography – MDCTA) to compare atherosclerotic plaques in the coronary arteries of the heart and the carotid arteries of the neck within the same patients. We found a strong correlation between the total plaque burden and the volume of calcified plaque in both critical arterial systems. Moreover, in patients with risk factors such as smoking or high cholesterol, the composition of these plaques, particularly their lipid content, exhibited significant similarities. These findings suggest that atherosclerosis is a systemic disease that displays parallel features in different arteries. Consequently, the evaluation of carotid plaques may offer valuable information regarding coronary artery status without the need for invasive procedures, potentially enhancing cardiovascular risk assessment.
Introduction
Atherosclerosis is a systemic and progressive inflammatory disease that is a leading cause of mortality and morbidity worldwide [1]. Although the pathological process can affect different vascular beds simultaneously, its involvement in the coronary and carotid arteries is of critical clinical importance due to its potential to cause life-threatening events such as myocardial infarction and ischemic stroke, respectively [2].
In the management of atherosclerotic disease, beyond the degree of luminal stenosis, the composition and morphological characteristics of the plaque play a fundamental role in risk stratification. Particularly, “vulnerable” plaques with high lipid content and thin fibrous caps carry a higher risk of rupture compared to stable and calcified plaques [3].
A key challenge is to non-invasively differentiate the contribution of systemic, cumulative drivers from local, active drivers of atherosclerosis [1]. In this study, we propose a quantitative, imaging-based model that uses plaque composition, as defined by computed tomography (CT) Hounsfield units (HU), to dissociate these components. We conceptualize calcified plaque as a surrogate for the systemic burden and soft plaque as a marker for local pathological activity. By applying this model, we aimed to investigate the differential impact of these drivers in the coronary and carotid arteries, hypothesizing that their unique hemodynamic and biological environments would lead to distinct plaque composition profiles.
In recent years, multidetector computed tomography angiography (MDCTA) has revolutionized the non-invasive assessment of atherosclerotic plaques [4]. This technology not only determines the degree of stenosis but also provides quantitative data on critical morphological features such as plaque volume, composition (soft, mixed, calcified), and vascular remodeling [4]. This has enabled a more accurate determination of individual patient risk profiles.
Although the systemic nature of atherosclerosis is well understood, studies in the literature have generally focused on a single vascular bed (either the coronary or carotid arteries) [5]. The number of studies that simultaneously and quantitatively compare plaque characteristics in both coronary and carotid arteries using MDCTA in the same patient group is quite limited. Understanding whether the plaque morphology and composition in these two critical arterial systems are similar could provide valuable insights into the systemic progression of the disease and reveal the potential of findings in one arterial bed to predict risk in the other [6].
Aim
In this context, this study aimed to quantitatively compare the morphological and compositional characteristics of atherosclerotic plaques in the coronary and carotid arteries within the same patient population using MDCTA, and to investigate the relationship between these characteristics and systemic cardiometabolic risk factors.
Material and methods
Study design and patient population
Of the 153 patients who underwent both coronary and carotid MDCTA at our institution between January 2017 and April 2024, 88 were included in the final analysis. Patients with a history of coronary, carotid stenting, or carotid endarterectomy, severe image artifacts, an acute ischemic event during imaging, or severe renal insufficiency (GFR < 30 ml/min/1.73 m2) were excluded. The study was approved by the Clinical Research Ethics Committee (Decision No: GOKAEK-2024/20/16) and conducted in accordance with the principles of the Declaration of Helsinki. Written informed consent was obtained from all patients.
MDCTA imaging protocol
All examinations were performed with a 640-slice MDCTA scanner (Aquilion ONE GENESIS; Canon Medical Systems). For coronary MDCTA, patients with a heart rate >65 beats/min were administered a β-blocker to achieve a target heart rate of < 60 beats/min. A non-ionic contrast agent (350 mgI/ml) was injected at a rate of 4–6 ml/s, and the scan was initiated with prospective or retrospective ECG gating when a threshold of 200–230 HU was reached in the aortic root. ECG gating was not used for carotid MDCTA. The scan area extended from the aortic arch to the intracranial internal carotid artery segments.
Image analysis and plaque measurement
All MDCTA images were independently analyzed by two experienced radiologists using Vitrea Advanced Imaging Platform software. Plaques were classified based on their mean attenuation values as MDCTA-defined soft (< 50 HU), MDCTA-defined mixed (50–149 HU), or MDCTA-defined calcified (≥ 150 HU). For the remainder of this manuscript, these will be referred to simply as soft, mixed, and calcified plaques for brevity, acknowledging that they are imaging-based classifications. These HU thresholds for plaque characterization are based on previously published validation studies that have demonstrated a high correlation between MDCTA plaque composition analysis and findings from intravascular ultrasound (IVUS) or histopathology, which are considered gold-standard methods [3]. For each plaque, the total plaque volume and plaque burden (calculated as [(vessel area – lumen area)/vessel area] × 100) were measured semi-automatically. Coronary artery disease severity was categorized based on the number of major epicardial vessels (LAD, LCX, RCA) with significant stenosis (> 50%) as one-, two-, or three-vessel disease. Carotid artery stenosis was determined according to the North American Symptomatic Carotid Endarterectomy Trial (NASCET) criteria, measuring the narrowest luminal diameter and the diameter of the distal normal internal carotid artery.
Statistical analysis
Statistical analyses were conducted using IBM SPSS Statistics 25.0 (IBM Corp., Armonk, NY). Continuous variables with a normal distribution were presented as means (standard deviations) and compared with the two-sample t-test. Pearson or Spearman tests were employed for correlation analyses. The Pearson correlation was used to examine the relationship between plaque morphology in coronary and carotid arteries and computed tomography. A two-tailed p < 0.05 was considered statistically significant.
Results
Patient demographics and baseline characteristics
A total of 88 participants were included in the study. The mean age of the study population was 72.9 ±9.7 years, and the mean body mass index (BMI) was 28.3 ±5.1 kg/m2. Of the participants, 47 (53.4%) were male. The baseline demographic, clinical, and key imaging characteristics of the study population are summarized in Table I. The prevalence of comorbid conditions was as follows: hypertension in 67 individuals (76.1%), diabetes mellitus in 41 (46.6%), dyslipidaemia in 33 (37.5%), and chronic kidney disease (CKD) in 12 (13.6%). Additionally, 35 participants (39.8%) reported a history of smoking, and 19 (21.6%) were receiving statin therapy.
Table I
Demographic and clinical characteristics of the patients (n = 88)
Analysis of coronary and carotid artery disease burden revealed a significant atherosclerotic load in the study group. The distribution of coronary artery disease was as follows: 22 (25.0%) patients had one-vessel disease, 40 (45.5%) patients had two-vessel disease, and 26 (29.5%) patients had three-vessel disease. The mean carotid artery stenosis for the entire cohort was 74.5 ±10.2%. When stratified by the extent of coronary disease, the mean carotid artery stenosis was 68.9 ±8.5% in patients with one-vessel disease, 75.1 ±9.8% in those with two-vessel disease, and 78.8 ±11.3% in those with three-vessel disease, suggesting a trend toward higher carotid stenosis in patients with more extensive coronary artery disease.
Plaque quantitative comparison and plaque composition analysis
The quantitative and compositional characteristics of atherosclerotic plaques in coronary and carotid arteries were compared and are summarized in Table II. A total of 71 of the 88 patients (80.7%) had at least one atherosclerotic plaque. One hundred fifty-two plaques in the coronary arteries and 138 plaques in the carotid arteries were analyzed. The plaque burden was significantly higher in the coronary arteries compared to the carotid arteries (63.7 ±12.9% vs. 36.9 ±17.7%, p < 0.001). Similarly, the percentage of soft plaques was markedly greater in the coronary arteries (23.5 ±8.4%) than in the carotid arteries (7.2 ±6.7%, p < 0.001).
Table II
Comparative analysis of atherosclerotic plaque burden and composition in coronary and carotid arteries
The proportion of calcified plaques was higher in the carotid arteries compared to the coronary arteries (52.2 ±17.4% vs. 42.6 ±15.7%, p < 0.001); mixed plaques were more prevalent in the carotid arteries (40.4 ±16.6%) than in the coronary arteries (33.7 ±10.0%), and this difference was statistically significant (p = 0.001). The total plaque volume was significantly greater in the carotid arteries (142.5 ±111.4 mm3) compared to the coronary arteries (75.9 ±37.0 mm3, p < 0.001).
Correlation analysis of plaque morphology in coronary and carotid arteries
Soft plaque volume in the coronary arteries showed a strong positive correlation with mixed plaque volume in the coronary arteries (r = 0.780, p < 0.001), and a moderate positive correlation with calcified plaque volume in the coronary arteries (r = 0.416, p < 0.001), as well as with total plaque volume in the coronary arteries (r = 0.782, p < 0.001).
The total carotid plaque volume was very strongly correlated with calcified plaque volume in the carotid arteries (r = 0.910, p < 0.001) and significantly associated with mixed plaque volume (r = 0.795, p < 0.001) and soft plaque volume (r = 0.540, p < 0.001).
The soft plaque volumes in coronary and carotid arteries were not correlated (r = –0.037, p = 0.735). Mixed plaque volume in the carotid arteries was strongly associated with soft plaque volume in the carotid arteries (r = 0.522, p < 0.001) and with calcified plaque volume in the carotid arteries (r = 0.538, p < 0.001), as well as with total carotid plaque volume (r = 0.795, p < 0.001). Mixed plaque volume in the coronary arteries was strongly associated with soft plaque volume in the coronary arteries (r = 0.780, p < 0.001), mixed plaque volume in the carotid arteries (r = 0.656, p < 0.043), calcified plaque volume in the coronary arteries (r = 0.404, p < 0.001), and total coronary plaque volume (r = 0.782, p < 0.001).
The calcified plaque volume in the coronary arteries showed a strong positive correlation with calcified plaque volume in the carotid arteries (r = 0.651, p < 0.001). A significant correlation was found between carotid and coronary total plaque volumes (r = 0.548, p = 0.033) (Table III, Figures 1, 2).
Table III
Correlation analysis of plaque morphology in coronary and carotid arteries with computed tomography
Figure 1
Three-dimensional (3D) volume-rendered and quantitative plaque analysis images of the patient’s coronary arteries obtained by multidetector computed tomography angiography (MDCTA). The yellow arrows indicate the lesion showing positive remodeling in the left anterior descending (LAD) artery and its narrowest cross-section. The blue arrows point to the cross-sectional (upper box) and volumetric (lower box) quantitative plaque analysis results (plaque burden, plaque composition, etc.) for this lesion. Plaque 1 – low-density, non-calcified (soft); plaque 2 – non-calcified (mixed); plaque 3 – calcified
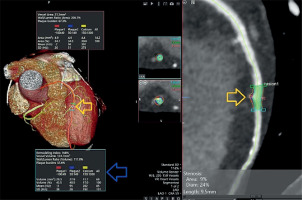
Figure 2
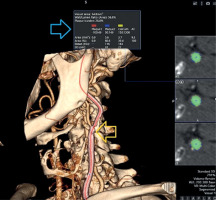
Three-dimensional (3D) volume-rendered and quantitative plaque analysis images of the same patient’s carotid arteries obtained by MDCTA. The yellow arrow indicates a non-obstructive plaque detected in the carotid artery. The blue arrow shows the analysis results for this plaque, including vessel cross-sectional area, plaque burden (26.8%), and plaque composition. Plaque 1 – low-density, soft; plaque 2 – mixed; plaque 3 – calcified
Discussion
This study quantitatively compared the atherosclerotic plaque burden, morphology, and composition in coronary and carotid arteries within the same patient group using MDCTA. The main findings of our study reveal that although atherosclerosis is a systemic disease, it exhibits significant regional differences in various vascular beds. Specifically, while the total plaque volume in the carotid arteries was significantly higher, the relative plaque burden (% stenosis) and the proportion of soft plaque were greater in the coronary arteries. These findings offer important implications for the pathophysiology and clinical risk assessment of atherosclerotic disease.
The finding in our study that the total plaque volume in the carotid arteries was significantly higher than in the coronary arteries may seem contradictory at first glance. However, this can be explained by the anatomical differences between the two vascular beds. Carotid arteries have a larger lumen compared to coronary arteries, which allows for the accumulation of larger volume plaques without causing hemodynamically significant stenosis. Similarly, previous studies have also reported that the atherosclerotic burden in carotid arteries is generally greater, but plaques in the coronary arteries can cause severe stenosis even at smaller volumes [7]. A recent study also showed that CTA-area stenosis measurement should not be used as a basis for carotid revascularization [8]. This indicates that the concepts of “plaque burden” and “plaque volume” should be considered separately in risk assessment.
One of the most remarkable findings of our study is the regional differences in plaque composition. The significantly higher proportion of soft plaque in the coronary arteries (23.5%) compared to the carotid arteries (7.2%) is of great clinical importance. Pathological studies have demonstrated that soft, lipid-rich plaques are the “vulnerable plaques” with the highest potential to rupture and lead to acute coronary syndromes [6, 9].
Our finding that the proportion of soft plaque is significantly higher in the coronary arteries and that this plaque volume does not correlate with carotid soft plaque volume is further strengthened by recent research that goes beyond simple plaque classification. Today, the concept of “soft plaque” is now understood as a fundamental component of “high-risk plaque features” (HRPF), which also includes elements such as positive remodeling, spotty calcification, and the “napkin-ring sign”. Landmark prospective trials, such as SCOT-HEART, have definitively proven that the detection of these features by coronary CT angiography (CCTA) provides powerful prognostic information beyond luminal stenosis alone, directly influencing treatment strategies and improving patient outcomes [10]. Furthermore, diagnostic capabilities have also advanced significantly; technologies such as radiomics and artificial intelligence (AI) can now extract tissue characteristics from the plaque that are imperceptible to the human eye, offering more robust signatures of plaque vulnerability [11]. This localized vulnerability is also directly visualized using molecular imaging techniques, such as 18F-FDG PET/CT, which identifies active inflammation – the primary driver of plaque instability [12]. Collectively, these modern advancements strongly support our conclusion: while calcified burden may be a systemic process, the formation of high-risk, non-calcified plaques is a local phenomenon driven by factors such as inflammation and adverse hemodynamics, which are increasingly identifiable with today’s advanced diagnostic tools.
This regional predisposition can be attributed to two main local factors. First, the coronary arteries are characterized by unique hemodynamic stresses, such as turbulent flow, torsion, and flexion, due to the cyclical motion of the heart. Regions with low and oscillatory wall shear stress (WSS) are particularly known to trigger endothelial dysfunction and pro-inflammatory processes, creating a fertile ground for the development of atherosclerotic plaques, especially vulnerable ones [13, 14]. Second, the intrinsic biological and molecular heterogeneity of vascular smooth muscle cells (VSMCs) in different arterial beds also influences the composition of plaques. VSMCs exhibit different phenotypic characteristics depending on their developmental origin and microenvironment. The responses of these cells to atherogenic stimuli (e.g., proliferation, migration, and transition to a synthetic phenotype) can vary from one artery to another [15]. Therefore, the different biological behaviors of VSMCs in the coronary and carotid arteries can lead to the formation of more fibrotic and stable plaques in one region and more inflammatory and lipid-laden plaques in another, even if systemic risk factors are the same.
Our findings on the differential plaque composition are further contextualized by research into the fundamental biology of atherosclerosis. While our study defines plaque characteristics through the lens of non-invasive imaging, it is crucial to acknowledge the complex molecular and genetic underpinnings that drive plaque development, as detailed in bioinformatics studies [16]. These studies highlight specific gene expression signatures (e.g., FGF2, CNN1, VAV3) associated with atherosclerotic progression. Our study, which focused on the clinical application and interpretation of MDCTA, complements such molecular investigations. The regional differences we observed in plaque composition likely stem from these underlying biological variations interacting with local hemodynamic forces. Future research integrating non-invasive imaging phenotypes with molecular and genetic markers could provide a more holistic understanding of plaque vulnerability, bridging the gap between macroscopic appearance and microscopic pathology.
Our correlation analyses further elucidate these regional differences. The lack of correlation between the soft plaque volumes in the coronary and carotid arteries (r = –0.037, p = 0.735) strengthens the hypothesis that soft plaque formation is driven more by the local vascular environment than by systemic factors. This is a critical finding, showing that the detection of a soft plaque in a patient’s carotid artery does not necessarily mean that a similar condition exists in the coronary arteries [17]. The strong correlations observed between mixed plaque volume and both soft and calcified plaque components suggest that mixed plaques may represent a transitional stage in the natural history of atherosclerosis. These lesions likely evolve from lipid-rich, non-calcified plaques toward more fibrotic or calcified phenotypes, reflecting the dynamic interplay between inflammation and healing processes within the vessel wall.
Conversely, we found a strong positive correlation between the calcified plaque volumes in the coronary and carotid arteries (r = 0.651, p < 0.001). Our analysis of disease extent further reinforces this evidence for a systemic atherosclerotic process. Our results showed a clear trend: patients with more widespread coronary artery disease (i.e., two- and three-vessel disease) also presented with a higher mean percentage of carotid artery stenosis. This dose-response relationship between the extent of disease in the coronary bed and the degree of stenosis in the carotid bed strongly supports the hypothesis that the chronic, calcified atherosclerotic burden is a reflection of a systemic pathology rather than purely local factors. This concept is consistent with large-scale studies demonstrating that vascular calcification is a more systemic process, influenced by general risk factors such as age, diabetes, and chronic kidney disease [18, 19]. For example, the MESA (Multi-Ethnic Study of Atherosclerosis) study demonstrated that the coronary artery calcium score is also an indicator of atherosclerosis in other vascular beds [20]. Therefore, the presence of significant calcification or extensive disease in one arterial bed increases the expectation of a similar calcified plaque burden in different regions.
Clinical implications and future perspectives
These findings reinforce the concept that systemic atherosclerosis requires a multimodal evaluation strategy. Carotid imaging, although valuable for stroke risk assessment, does not reliably reflect the dynamic and heterogeneous nature of coronary plaque morphology. Our observations offer a mechanistic rationale for employing advanced, direct coronary imaging modalities such as IVUS or optical coherence tomography (OCT) to visualize and quantify vulnerable, lipid-rich plaques that underlie acute coronary events. In line with this concept, recent findings by Ozderya et al. have highlighted that body composition metrics, particularly internal fat and metabolic age, are strongly correlated with coronary lesion complexity, emphasizing the systemic metabolic contribution to coronary plaque vulnerability and reinforcing the need for individualized imaging-based risk assessment [21]. Integrating these techniques into contemporary risk models may bridge the gap between anatomical imaging and functional risk prediction, facilitating personalized preventive therapy and more precise monitoring of disease progression.
Limitations. Our study has some limitations. The retrospective nature of the study design introduces a potential for selection bias. The patient cohort consists of individuals who underwent both coronary and carotid MDCTA for various clinical indications, which were not standardized for this research. These indications often included the evaluation of patients with multiple cardiovascular risk factors presenting with complex or overlapping symptoms (e.g., exertional chest pain in a patient with a history of transient ischemic attack), or as part of a comprehensive pre-operative assessment for major non-coronary vascular or cardiac surgery. Consequently, our study population might represent a cohort with a higher pre-test probability and burden of systemic atherosclerosis compared to the general population of patients with suspected CAD or carotid disease alone. This potential bias could influence the observed prevalence and characteristics of plaques, and the generalizability of our findings to a broader, lower-risk population may be limited. Another limitation was the absence of a formal power analysis for the soft plaque correlation.
The method of classifying plaque composition based on fixed HU threshold values obtained from MDCTA data has inherent limitations. Technical factors such as image noise, calcium “blooming” artifacts, which can overestimate calcified volume, and partial volume averaging effects, especially in smaller vessels, can affect attenuation measurements, potentially leading to the misclassification of plaque components. This creates some uncertainty, particularly in distinguishing soft and mixed plaques from calcified tissue, and may impact our quantitative analyses. As detailed reviews of this topic have pointed out, while MDCTA is a powerful non-invasive tool, it does not offer the same level of granular detail as invasive intravascular imaging methods, such as IVUS or optical coherence tomography (OCT) [22]. These gold-standard techniques provide superior resolution for characterizing specific plaque features, such as fibrous cap thickness or the precise morphology (e.g., thickness, arc, nodularity) of calcific deposits, which are also key determinants of plaque stability.
Conclusions
Our study highlights significant regional differences in plaque volume and composition between coronary and carotid arteries. While we found no anatomical correlation in soft plaque volumes between these vascular beds, this does not diminish the prognostic value of carotid imaging for predicting coronary events.
Existing literature indicates that specific features of carotid plaques, such as echo-lucent plaques, are predictive of major adverse cardiovascular events [23, 24]. This suggests that carotid plaque characteristics may reflect systemic atherosclerotic processes that increase the risk of coronary events, despite the lack of direct anatomical correlation.
A potential hypothesis for this paradox is that local inflammatory processes in carotid plaques may signal systemic vulnerabilities affecting the coronary arteries. Therefore, integrating carotid imaging findings with coronary assessments can enhance risk stratification and management for patients at high cardiovascular risk.







