Summary
Scientific evidence on coronary artery disease in sepsis is rather limited. We found that two-thirds of septic patients with elevated cardiac troponin I (cTnI) have obstructive coronary disease with complex features, including multivessel involvement in up to 50%, significant unprotected left main stenosis in up to 20%, and chronic total occlusion in up to 40%. Acute culprit lesions, which were apparent in up to 10%, were associated with markedly elevated cTnI levels. These patients may be candidates for coronary angiography, percutaneous coronary intervention, and antiplatelet treatment.
Introduction
Elevated cardiac troponin is a well-established phenomenon in sepsis, which is characterized by dysregulated host response to infection and organ dysfunction [1, 2]. An increased systemic inflammatory response may lead to coronary plaque destabilization and rupture, followed by obstructive thrombosis due to increased platelet activation and type 1 myocardial infarction [3, 4]. On the other hand, arterial hypotension and acute respiratory failure coupled with compensatory tachycardia may cause an imbalance between myocardial oxygen supply and demand, leading to type 2 myocardial infarction [5, 6]. Therefore, septic patients with coronary artery disease (CAD) are at increased risk for myocardial infarction [7–9].
Aim
In the present study, we investigated the incidence, extent, and characteristics of CAD in septic patients with elevated cardiac troponin and addressed possible mechanisms of myocardial infarction. We hypothesized that both type 1 and type 2 myocardial infarction may account for elevated cardiac troponin.
Material and methods
This was a single-center retrospective, observational study which enrolled consecutive septic patients with elevated high-sensitivity cardiac troponin I (cTnI) who were admitted to the Center for Intensive Internal Medicine at the University Medical Center Ljubljana (Slovenia) and underwent coronary angiography at the discretion of the treating physician during the index hospital stay.
Clinical and laboratory data and coronary angiography recordings were collected from the institutional database and department registry of critically ill patients. Peak cTnI during the hospital stay was used. Sepsis was defined according to Sepsis-3 criteria as life-threatening organ dysfunction caused by a dysregulated host response to infection [1]. Degree of organ dysfunction and circulatory/cellular metabolism abnormalities were quantified by Sequential Organ Failure Assessment (SOFA) and arterial lactate concentration. Coronary angiograms were reviewed by an experienced interventional cardiologist blinded to cTnI levels who evaluated the number of diseased vessels with obstructive stenosis (≥ 70% diameter stenosis), presence of unprotected left main stenosis (ULM) (≥ 50% diameter stenosis), chronic total occlusion (CTO), and an acute culprit lesion. An acute culprit lesion was defined as abrupt closure with Thrombolysis in Myocardial Infarction (TIMI) flow 0–1 or TIMI 2–3 with angiographic images suggesting thrombus or ulcerated plaque [10]. Complexity of CAD was quantitatively estimated using the SYNTAX (Synergy between Percutaneous Coronary Intervention with Taxus and Cardiac Surgery) score, which was calculated for each coronary lesion with more than 50% diameter stenosis in vessels of at least 1.5 mm by the proposed website algorithm (www.syntaxscore.com) [11]. The decision for percutaneous coronary intervention (PCI) was at the discretion of the interventional cardiologist and intensive care physician.
Statistical analysis
Continuous variables are reported as median with 25th and 75th interquartile range (IQR) and categorical variables as frequencies and percentages. A scatter plot was constructed to depict the relationship between SYNTAX score and peak cTnI. Whisker plots were used to graphically depict the distribution of peak cTnI according to the presence of an acute culprit lesion. Comparisons between continuous variables were performed with Student’s t-test or Mann-Whitney U test, and comparisons between categorical variables were made using the χ2 test. Pearson correlation was used to assess the association between SYNTAX score and peak cTnI. All probability values were two-sided, and p < 0.05 was considered statistically significant. Analyses were performed with R Software Environment for Statistical Computing and Graphics (R version 4.1.3; R Foundation for Statistical Computing, Vienna, Austria) [12]. The STROBE checklist (http://www.annals.org/) was used to define the content of the manuscript and guide the drafting.
Results
Between 1st January 2012 and 7th December 2022, among 1364 consecutive patients with sepsis/septic shock, 74 had positive cTnI and coronary angiography during the index hospital stay. The patients were predominantly men (73%) with an average age of 70 years, presence of ST-elevation in 6.8%, mean left ventricular ejection fraction 30%, mean peak cTnI 16,647 ng/l (normal < 40 ng/l), and arterial lactate of 5.1 mmol/l, with values above 2 mmol/l in 81% (Table I). Although the infection source was variable, the respiratory tract was predominant (31%), and hemocultures were positive in 39%. The systemic inflammatory response was characterized by increased white blood count (19.0 × 109/l), C-reactive protein (234 mg/l; normal < 5 mg/l), and procalcitonin (14 µg/l; normal < 0.20 µg/l).
Table I
Patient characteristics in patients who underwent coronary angiography during index hospital stay (n = 74)
Intensive care treatment included intubation/mechanical ventilation (92%), norepinephrine/vasopressin (95%), dobutamine/epinephrine (74%), hemodialysis (38%), PCI (24%) and coronary artery bypass grafting (8.1%). Intensive care unit survival was 39% and hospital survival was 31%. There was no significant difference in intensive care unit survival (56% vs. 34%; p = 0.100) or hospital survival (39% vs. 29%; p = 0.400) between patients with or without PCI.
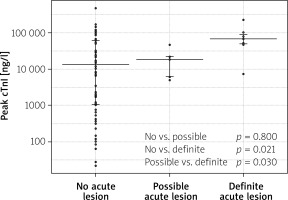
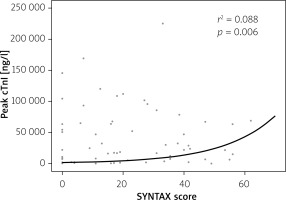
Obstructive coronary disease (> 70% diameter stenosis) was documented in 65%, with multivessel involvement in 51%. Chronic total occlusion (CTO) was present in 35% and > 50% unprotected left main (ULM) stenosis in 22% (Table II). Definite or possible acute culprit lesions were documented in 9.5% and 6.8%, respectively. There was only a weak, non-linear correlation between SYNTAX score and peak cTnI (r2 = 0.088; p = 0.006) (Figure 1).
Table II
Findings on coronary angiography
Figure 1
Relationship between SYNTAX score and peak concentration of cTnI (logarithmic scale) in patients with coronary angiography during index hospitalization
cTnI – cardiac troponin I, SYNTAX – Synergy between Percutaneous Coronary Intervention with Taxus and Cardiac Surgery.
Peak cTnI was significantly elevated in patients with a definite acute culprit lesion (67,659 ng/l; IQR 49,426–90,153 ng/l) compared to patients with a possible (18,422 ng/l; IQR 6060–21,965 ng/l; p = 0.030) or no acute culprit lesion (13,410 ng/l; IQR 1052–61041 ng/l; p = 0.021) (Figure 2).
Discussion
We found that two-thirds of septic patients with elevated cTnI have obstructive coronary disease with complex features, including multivessel involvement in around 50%, significant ULM stenosis in around 20%, and CTO in up to 40%. In a similar study, Tsai et al. [13] investigated 239 patients hospitalized for acute infection who developed acute myocardial infarction without ST-elevation and found an even higher percentage of obstructive CAD, with multivessel involvement in 95%, significant ULM in 27%, and CTO in 35%. Another study in 78 septic patients, which excluded patients with known CAD, showed obstructive CAD in only 41% [14]. In contrast to these studies, we also used the generally accepted SYNTAX score to quantitatively describe the severity of CAD [11]. Although statistically significant, the correlation between SYNTAX score and peak cTnI was very weak and clinically meaningless. This is in contrast with our hypothesis that increased complexity of CAD would be associated with more pronounced elevations in cTnI.
The added value of our study lies in addressing the incidence of acute culprit lesions, which were apparent in up to 10%. Despite the fact that assessment was performed by an experienced interventional cardiologist blinded to cTnI levels and was based on predefined angiographic criteria, such a classification is subjective, given the shortcomings of coronary angiography. This was also the reason for creating a subgroup of “possible acute culprit”. Intravascular imaging, especially optical coherence tomography (OCT), which would provide much more accurate plaque characterization and probably even further increase the percentage of acute culprit lesions, was not used.
While stable CAD is likely to be responsible for type 2 myocardial infarction, presence of an acute culprit lesion may result in type 1 myocardial infarction, which was characterized by much more pronounced elevation in cTnI. However, caution is warranted in making this distinction, which may be especially problematic in septic patients addressed in our study. The presence of significant stenosis and performance of PCI are not sufficient to clearly distinguish type 1 from type 2 myocardial infarction. Furthermore, septic patients often experience multifactorial myocardial injury related to cytokine-mediated myocardial damage, microvascular dysfunction, and septic cardiomyopathy, which may by itself cause or perpetuate myocardial necrosis.
In view of the rather limited scientific evidence on CAD in sepsis [13–16], we believe that our study may represent the initial step to better stratify septic patients by using cTnI. This is practically important in view of obvious difficulties in symptom reporting in sedated and mechanically ventilated patients and frequent lack of obvious ischemic changes in the 12-lead electrocardiogram. Patients with very high cTnI might be candidates for coronary angiography, PCI, and antiplatelet treatment. On the other hand, one must consider potential interventional risks in the setting of a critical condition, multiorgan failure, and threat of bleeding, which have to be counterbalanced with potential PCI benefits. Although not designed to address potential benefits, our study did not show a significant association between PCI and either intensive care unit or hospital survival. Identification of septic patients who may benefit from PCI therefore remains a major challenge, which should be addressed in future appropriately designed and powered clinical studies. Since septic patients exhibit a broad spectrum of organ dysfunction, the SOFA score could be used for further risk stratification and to assess whether angiographic findings are predictive of survival independently of SOFA score, or conversely, if PCI may improve prognosis in specific subgroups defined by SOFA. Moreover, ROC curves for SYNTAX score, cTnI, SOFA score, and their combination could uncover eventual synergistic prognostic value, strengthen clinical relevance of the angiographic findings, and eventually improve decision-making for the invasive coronary strategy in septic patients.
Conclusions
Our results, however, should be interpreted in view of several study limitations. This is a single-center retrospective observational study with a rather small number of patients, which precluded subgroup analyses. Because of the retrospective design of our study, we cannot provide more detailed information on ST depression suggestive of myocardial infarction. We reported only obvious ST elevation, which was rather rare. The decision for coronary angiography and PCI was not protocolized but individually decided by the treating physicians. We therefore do not know the number of septic patients with positive cTnI who did not undergo coronary angiography either because of the decision of the treating physician or because of the severe clinical condition or death before any intervention. This may represent a clinically very sick and important subgroup, introducing potential selection bias. Furthermore, data on the exact timing of coronary angiography were unavailable. Based on all these limitations, our findings need further validation in external and preferably larger cohorts of patients.