Introduction
Brain metastases from solid tumours are by far the most common intracranial neoplasms, surpassing the incidence of primary brain tumours [1]. Their overall incidence is increasing due to improvements in diagnostic imaging techniques, prolonged survival of cancer patients, and the general increase in life expectancy. Brain tumour deposits are diagnosed in more than one out of ten cancer patients, adversely affecting their quality of life and overall survival (OS). Lung cancer is thought to rank first among different neoplasms as the primary tumour most commonly associated with the onset of brain metastatic disease, followed by breast cancer and melanoma, the latter of which has the highest propensity for infiltrating the central nervous system (CNS) [2, 3].
Brain metastases from solid tumours share the same histological features as their primary source [2]. Dissemination of neoplastic cells into the brain usually occurs through the hematogenous route, resulting in tumour deposits that present with focal growth, in contrast to primary brain tumours, which may exhibit diffuse parenchymal infiltration [4]. Imaging techniques such as magnetic resonance imaging and computed tomography are used to diagnose brain metastases, with the most common locations being the cerebral hemispheres, cerebellum, and brainstem. Following imaging results, patients are treated with radiation therapy, systemic therapy, and, in some cases, neurosurgical excision of the metastatic lesions [3].
Over the last decade, the advent of immunotherapy and targeted therapy has led to significant improvements in patient outcomes. Tumour molecular profiling has become an integral part of cancer treatment, enabling the application of novel agents with remarkable results. Immune checkpoint inhibitors (ICI), particularly anti-programmed cell death 1 (anti-PD-1), anti- programmed death ligand 1 (anti-PD-L1), and anti-cytotoxic T-lymphocyte- associated protein 4, have been approved for the treatment of various tumours, including lung cancer, breast cancer, melanoma, gastrointestinal and genitourinary tumours, among others. However, most contemporary immunotherapy clinical trials have excluded patients with metastatic brain disease [5]. Therefore, the efficacy of immunotherapy in treating brain metastases from solid tumours remains to be fully elucidated [6].
Investigating the intracranial tumour microenvironment (TME) is inherently challenging. The unique cells of the CNS, their complex interactions, their relationship with the extracellular matrix, as well as the blood-brain barrier, the blood-cerebrospinal fluid barrier, and specific metabolic conditions, result in an immune-privileged environment [7]. The invasion of metastatic cancer cells into this area leads to the formation of a TME that has not been adequately explored, leaving many questions unanswered. It has not been widely studied whether brain metastases retain the same immunological characteristics as the primary lesion, which could make them susceptible to treatments based on the primary tumour biopsy [8]. Additionally, tumours evolve over the course of the disease, exhibiting spatial and temporal heterogeneity. Therefore, investigating the TME in brain metastatic lesions could provide valuable insights into this poorly studied area, which is difficult to explore due to the challenges in obtaining tissue samples [9, 10].
The aim of our study was to investigate the host’s immune response to primary tumours and concomitant brain metastases in the CNS from various malignancies. Specifically, we aimed to assess the presence or absence of an immune response in brain metastases. The evaluation of the immune response was conducted by analysing tumour-infiltrating lymphocytes (TIL) and the expression of PD-1 and PD-L1 in tissue samples from brain metastases and matched primary tumours. Additionally, we explored the differences and similarities in immune activity between primary tumours and brain metastases, as well as their prognostic significance for disease outcome, response to treatment, and OS.
Material and methods
This was a retrospective study using archival tissue samples from the Pathology Laboratory of the University Hospital of Ioannina. The study focused on patients with adequate tissue samples from predominant solid tumours and synchronous or metachronous brain metastases, who were diagnosed and treated in the Neurosurgery Clinic and Medical Oncology Clinic at the University Hospital of Ioannina between 2002 and 2018. The selected archival tissues were first evaluated for sample adequacy to proceed with immunohistochemistry (IHC) evaluation. When available, the medical records of the patients were also used to assess the prognostic significance of immune activity in terms of response to treatment, overall survival, and association with clinical parameters. The study was approved by the scientific board of our institution.
Patient characteristics
In total, we evaluated brain metastatic tissue samples from 72 patients treated at our institution. All 72 patients had undergone surgical excision of brain metastatic lesions in the Neurosurgery Clinic at the University Hospital of Ioannina. All were adults above 18 years old, and for 62 of them, medical records were available in the Medical Oncology Clinic of Ioannina. We identified nine cases with both brain metastases and paired tissue samples from the primary tumour.
Immunohistochemistry
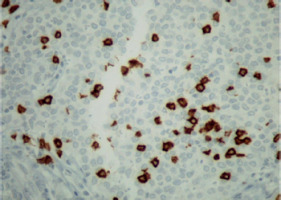
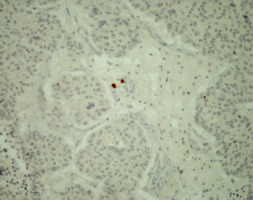
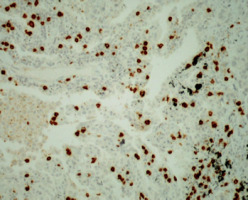
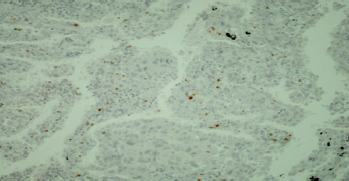
To evaluate TIL and the expression of PD-1 and PD-L1, archival tissue from brain metastases of all 72 patients was used. Nine patients also had matching tissue samples from both the primary tumour and brain metastases available for evaluation. After microdissection, representative formalin-fixed paraffin-embedded histological sections of 4 µm were used. The primary antibodies used in our study, including CD3, CD8, FOXP3, PD-1, and PD-L1, are listed in Table 1. Figures 1–12 show screenshots of immune biomarkers. Whole tissue sections were used instead of tissue microarrays (TMA), as the latter were not recommended for evaluating potential TIL heterogeneity according to published data.
Table 1
Primary antibodies, the source, the clone and the dilution
A binocular optical microscope was used to assess TIL immune infiltration in tissue sections, after excluding areas of tumour necrosis, crush artifacts, and normal tissue. Tumour-infiltrating lymphocytes were evaluated within the tumour borders, including both the central tumour and the invasive margin. Strong cytoplasmic staining for CD3 and CD8, and strong nuclear staining for FOXP3, were considered positive. Counts were performed individually in high-power fields (400×, 0.24 mm2 per field) for up to 10 fields per case. Tumour areas were divided into stromal and intraepithelial compartments, and intratumoral and stromal TIL were counted separately. Tumour-infiltrating lymphocytes density was compartment area, and the median density per case was used as a cut-off to classify samples as high or low for CD3, CD8, and FOXP3 [11].
The diagnostic companion test Ventana SP142 was used to assess PD-L1 expression, based on the proposed interpretation guide for the Ventana PD-L1 assay. Programmed cell death 1 expression was evaluated using the Roselin PD-1 MRQ-22 CellMarque companion test.
Statistical methods
For the statistical analysis of our results, we used descriptive statistics, including median values (with minimum and maximum) for patient characteristics. χ2 tests and Fisher’s exact tests were used to determine correlations between IHC results and patient clinicopathological parameters. Non-parametric methods, such as the Kruskal- Wallis test and the Wilcoxon rank-sum test, were used to evaluate continuous variables. Spearman’s rank correlation was applied to assess correlations between continuous variables. Median values of the evaluated biomarkers were used as cut-offs to categorize tumours as high or low expression for CD3, CD8, and FOXP3. For categorizing immune cells as PD-L1 positive or negative, a cut-off of 1% was used, while for tumour cells, a three-tier system for PD-L1 expression was applied: < 1%, 1–49%, and > 50%.
Log-rank tests and Kaplan-Meier curves were used to evaluate overall survival. Specifically, OS1 was estimated from the time of diagnosis of the primary tumour until death from any cause or last contact, while OS2 was estimated from the time of brain metastasis diagnosis until death from any cause or last contact. Cox regression models were applied to evaluate the effects of clinicopathological parameters and study biomarkers on OS1 and OS2. Variables with a univariate regression p-value < 0.1 were included in the multivariate regression analysis.
Results
Among the 72 patients included in our retrospective study, 26.4% were female and 73.6% were male. A total of 36 patients had primary lung cancer, followed by breast cancer, melanoma, gastric and gastroesophageal junction cancer, kidney cancer, prostate cancer, cancer of unknown primary, bladder cancer, and primary peritoneal cancer (Table 2). The median age at diagnosis was 64 years, and the majority of patients were diagnosed with high-grade tumours.
Table 2
Characteristics of the patients
Among the 62 patients with available medical records, half reported a history of arterial hypertension. Twenty- nine had undergone surgical excision of the primary tumour, and twenty-two had undergone chemotherapy. All 62 patients with available records had received whole brain radiation therapy for the treatment of brain metastases. More than half of them received first-line systemic therapy, and 42.6% received second or further lines of therapy. Among the total of 72 patients in our study, we were able to identify 9 patients with matched primary tumour and brain metastasis tissue samples. Of these, five had primary lung cancer, two had breast cancer, one had renal cell cancer, and one had urothelial carcinoma.
Immunohistochemistry evaluation for CD3, CD8, FOXP3, PD-1, and PD-L1 was performed on 62 out of the 72 available samples (86.1%). Data for CD3, CD8, and FOXP3 were available for 97.2%, 98.6%, and 93.1% of the cohort, respectively. Data for PD-1 and PD-L1 were available for 97.2% and 94.4% of the total cohort, respectively. Tumour-infiltrating lymphocyte density was calculated for 70 patients (97.2%) (Figure 13).
Furthermore, we examined possible correlations between clinicopathological patient characteristics and the study’s leukocyte immune infiltration biomarkers. No statistically significant correlations were documented, and TIL density also did not statistically correlate with any of the clinical parameters (p-values > 0.050).
Specifically, comorbidities did not show a statistically significant impact on overall survival. For OS1, patients without comorbidities had 24/29 events with a hazard ratio (HR) of 0.65 (95% CI: 0.37–1.15, p = 0.139) compared to patients with comorbidities (26/30, reference). For OS2, patients without comorbidities had 24/29 events with an HR of 1.14 (95% CI: 0.66–1.98, p = 0.642) vs. patients with comorbidities (27/31, reference). These findings indicate that comorbidities were not significantly associated with OS1 or OS2 in this cohort.
Regarding paired samples from primary tumour lesions and brain metastases, data were available for seven out of the nine matched samples. In four cases, there was an increase in TIL density in brain metastases compared to the primary lesion, two cases showed a decrease in TIL density in brain metastases, and in one pair, there was no difference in TIL density between the primary and metastatic lesions (Figure 14).
All metastatic tumour samples had reduced intraepithelial CD3 expression levels, and more than 50% of the samples also exhibited lower stromal CD3 and total CD3 levels. Four out of the seven paired samples had lower intraepithelial, stromal, and total CD8 levels in the metastatic tissue compared to the primary lesion. Of the nine paired samples, four had reduced intraepithelial FOXP3 levels in the metastatic lesions, while five exhibited higher stromal and total FOXP3 levels in the brain metastasis tissue. In the majority of patients, there was no difference in PD-L1 levels in tumour cells between primary and metastatic tissue, with only two cases showing an increase. Programmed death ligand 1 expression in immune cells was lower in the metastatic lesions compared to the primary tumours for most patients.
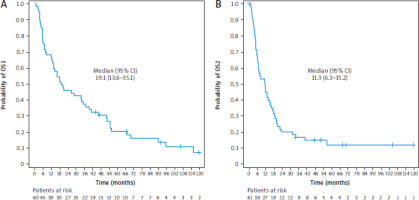
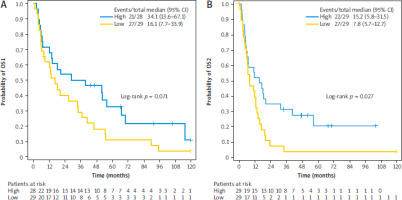
Follow-up data were available for 61 patients. Notably, for one patient, only the date of brain metastasis diagnosis was documented and not the primary tumour diagnosis, leading to the estimation of OS1 for 60 out of 61 patients. The median follow-up was 106.03 months, during which 52 deaths were reported. The median OS1 was 19.1 months (95% CI: 13.6–35.1), and the median OS2 was 11.35 months (95% CI: 6.3–15.2). The 5-year OS1 and OS2 were 20.4% and 11.9%, respectively (Figure 15). There was only a trend toward increased death risk in patients with low levels of FOXP3 (Figure 16).
Discussion
Despite recent advances in contemporary oncology therapeutics, brain metastases are still associated with clinical deterioration and a decrease in overall survival. Immunotherapy has had a significant positive impact on cancer patients’ life expectancy, while simultaneously highlighting the importance of immune response and its mechanisms of action. The presence of TIL and higher tumour PD-L1 expression have been associated with improved responses to ICI. It has been documented that tumours with a high mutational burden are usually enriched in TIL and, therefore, more responsive to immunotherapy [12]. However, the effects of immunotherapy appear to be attenuated in brain metastases. The tumour microenvironment and immune response in brain metastases have come into focus to elucidate the documented differences in immunotherapy efficacy between intracranial and extracranial tissues [13].
Berghoff et al. [14] have already reported that TIL density, specifically CD3 and CD8, was higher in primary lung tumours compared to matched brain metastases, while PD-L1 expression levels were higher in brain metastatic lesions compared to the primary tumour. In our study, we identified lower CD3 levels in intracranial tissues compared to extracranial primaries. All brain metastasis tissue samples had lower intraepithelial CD3 levels, and more than half exhibited lower levels of stromal CD3.
Our study also depicted an interception in the immune response, as total CD8 levels, both intraepithelial and stromal, were found to be lower in brain metastases compared to primary tissues. Galon et al. [15] reported that CD8 lymphocytes are usually present in higher levels at disease onset and are associated with better outcomes. However, during the course of the disease, CD8 levels, known for their cytotoxic and antineoplastic properties, decrease, which may result in worse outcomes.
Furthermore, FOXP3 lymphocytes, which have immunosuppressive properties, were found to be upregulated in brain metastases compared to primary tumour tissues. Indeed, higher levels of FOXP3 may correlate with worse outcomes observed in patients with brain tumour deposits [16].
Various attempts have been made to explain the differences observed in immunotherapy effects between primary tumours and brain metastases. Kim et al. [17] studied the TME in both lung cancer primaries and paired brain metastases. According to their results, higher levels of TIL and PD-L1 expression were noted in primary tumour tissues compared to cranial metastatic deposits, resulting in better responses of primary lesions. Similarly, Berghoff et al. [14] reported higher levels of PD-1 in primary lesion tissues compared to brain metastases. Our study reached the same conclusion, as the majority of paired samples showed lower levels of PD-1 expression in brain metastases. Conversely, PD-L1 expression levels in tumour cells did not differ between primaries and brain metastases in most samples, while lymphocyte PD-L1 expression levels were lower in metastatic lesions compared to primaries. A more recent study reported that PD-L1 expression in brain metastases is concordant with that of the primary tumour in 75% of cases, with no predictive discordance factors. CD8 expression levels also showed concordance, demonstrating a similar immunohistochemical profile between primaries and matched tumour samples [8].
In addition, higher levels of PD-L1 have been associated with poor prognosis in both early and advanced disease stages. However, in our study, we did not find any associations between PD-1/PD-L1 levels and OS [17]. This lack of association may be attributed to our study sample, which primarily comprised patients who had undergone surgery and additional systemic therapies in the pre-immunotherapy era. Furthermore, higher FOXP3 levels have been associated with a poor prognosis in patients with urothelial carcinoma, while their positive predictive value has been documented in colorectal cancer patients [18, 19]. In our study, there was a trend towards a higher risk of dying in patients with lower FOXP3 levels. Finally, our study identified advanced age and the number of lines of systemic therapy as factors that adversely affect survival.
Conclusions
The brain TME has been postulated to be immune- privileged, and recent scientific advances in oncology therapeutics have largely overlooked this complex system of brain parenchyma and its properties. Although the association between the levels of TIL and PD-L1 and the effects of immunotherapy has been documented in primary tumours, similar data regarding brain metastases are scarce, primarily due to smaller sample sizes and often inaccessible tissue. Nevertheless, emerging data suggest the need for further investigation into the brain metastases microenvironment to expand our knowledge and identify new and effective therapies for patients with intracranial metastases. Our study, despite its inherent limitations of being retrospective and having a small sample size, is among the few in the current literature that highlights immune activity in the brain as a host. The cerebral parenchyma was found to be a microenvironment with varying levels of TIL and immune checkpoint expression, warranting further investigation.