Introduction
Diabetes mellitus is a chronic metabolic disorder whose global prevalence has risen dramatically in recent decades. According to the latest report by the International Diabetes Federation, approximately 537 million people worldwide are currently living with diabetes, reflecting a substantial public health burden [1]. The alarming increase in prevalence, projected to rise further in the coming decades, underscores an urgent need for effective therapeutic interventions. Characterized primarily by chronic hyperglycemia, diabetes results from impaired insulin secretion, decreased insulin sensitivity, or a combination of both, eventually leading to numerous debilitating complications including cardiovascular diseases, neuropathy, nephropathy, and severe pancreatic damage [1]. Conventional therapeutic strategies, such as oral hypoglycemic drugs and insulin therapy, have been widely used to manage diabetes; however, these approaches frequently encounter significant limitations including adverse drug reactions, progressive loss of efficacy, and prohibitive treatment costs [2]. Due to these limitations, there has been a marked shift toward exploring complementary and alternative medicinal treatments, especially those derived from natural sources. Medicinal plants, owing to their wide range of bioactive compounds such as flavonoids, alkaloids, phenolics, and terpenoids, have gained considerable attention for their potential in managing diabetes through mechanisms such as enhanced insulin secretion, improved glucose tolerance, and reduced oxidative stress [3].
Caesalpinia bonduc (L.) Roxb or C. bonduc, locally known as Kebiul in Indonesia, has emerged as a promising candidate due to its longstanding use in traditional medicine for diabetes management across various tropical regions. Empirical evidence suggests that its bioactive compounds, notably flavonoids, may offer significant antidiabetic benefits, enhancing both glycemic control and pancreatic protection [4].
Nevertheless, despite promising preliminary results from studies using C. bonduc extracts, significant gaps persist in understanding its specific efficacy and optimal application, particularly regarding the plant’s different anatomical parts. Research has predominantly centered on seed extracts, demonstrating marked hypoglycemic effects in diabetic models induced by diabetogenic agents such as streptozotocin (STZ) and alloxan [4–6]. Previous research has shown that C. bonduc significantly reduced blood sugar levels at 3 hours (p < 0.01) and five hours (p < 0.001) when administered at a dosage of 250 mg/kg b.w., resulting in decreases of 9.99% and 14.90% in alloxan-induced models and 16.49% and 31.51% in STZ-induced models [7]. Although the phytochemical profiles of seeds and leaves share similarities, the leaves potentially hold greater pharmacological value due to their relatively higher flavonoid content. Despite this advantage, comprehensive evaluations of leaf extract efficacy, particularly in STZ-induced diabetic models, remain scarce, thereby necessitating focused scientific inquiries.
The flavonoid compounds present in C. bonduc are reported to enhance insulin secretion, improve glucose tolerance, decrease peripheral insulin resistance, and reduce intestinal glucose absorption [8]. Notably, phytochemical analyses indicate that flavonoids are most abundantly concentrated in the leaves of C. bonduc, followed by the seed core and stem [3, 9]. These findings suggest promising potential for C. bonduc leaf extract as an effective hypoglycemic agent with pancreatic tissue-protective properties.
Addressing the gap in knowledge concerning the effectiveness of C. bonduc leaf extract in STZ-induced diabetic models is essential, as STZ offers a robust method for mimicking human type 1 diabetes through selective destruction of pancreatic β-cells [10]. This selective toxicity facilitates precise analysis of therapeutic interventions aimed at β-cell regeneration and insulin production restoration, critical elements in comprehensive diabetes management. The STZ-induced model thus provides a clinically relevant platform for elucidating the potential regenerative and protective mechanisms offered by bioactive phytochemicals.
Flavonoids, prominent bioactive constituents within C. bonduc, have been scientifically established to enhance insulin secretion, improve peripheral glucose uptake, and exhibit potent antioxidant effects that protect pancreatic β-cells from oxidative and inflammatory damage [8, 11]. Several studies underscore the beneficial effects of flavonoids in diabetes management through modulation of key metabolic pathways, including the suppression of enzymes such as α-amylase and α-glucosidase, consequently limiting glucose absorption from the gut [12, 13]. These multifaceted pharmacological activities suggest that flavonoid-rich C. bonduc leaf extracts could represent a promising natural therapeutic strategy for diabetes control, highlighting their potential as alternatives or adjuncts to conventional therapies.
Previous literature underscores the importance of exploring different plant parts in the development of phytotherapeutics. Flavonoids, a group of polyphenolic compounds, play critical roles in modulating glucose metabolism by enhancing insulin sensitivity and mitigating oxidative stress [8]. Previous studies provided evidence for the antidiabetic effects of C. bonduc seeds in alloxan-induced diabetes; further investigation into C. bonduc leaves is warranted given their higher flavonoid content [14, 15]. Moreover, the streptozotocin-induced diabetic model offers a more clinically relevant context for examining pancreatic β-cell damage and recovery, contrasting with the mechanism of alloxan induction [10].
Previous studies specifically focusing on C. bonduc have validated the hypoglycemic efficacy of seed extracts across diverse experimental diabetic models. For instance, research has demonstrated significant blood glucose reductions following the administration of C. bonduc seed extract in both STZ-induced and alloxan-induced diabetic rats, indicating robust antidiabetic potential attributed largely to flavonoids and other phytochemical constituents [4, 7]. Despite these promising findings, the therapeutic potential of leaf extracts remains under-examined. Initial evidence suggests that leaf-derived flavonoids could surpass seed extracts in potency due to their higher flavonoid concentrations, underscoring the necessity for targeted research to explore their therapeutic benefits fully [14].
Additionally, flavonoids have been shown to exert protective effects on pancreatic β-cells by reducing inflammation and oxidative damage, key factors implicated in the pathogenesis of diabetes [8, 11]. While considerable evidence has documented the broader antidiabetic effects of flavonoids and their pancreatic protective roles, specific research investigating C. bonduc leaf extract’s ability to mitigate diabetes-induced pancreatic tissue damage is notably limited. Few studies have systematically explored the histopathological implications of C. bonduc extracts, despite indications that flavonoids effectively attenuate pancreatic inflammation, promote β-cell regeneration, and improve overall tissue morphology in diabetic models [15]. Such histopathological assessments are crucial for comprehensively understanding therapeutic efficacy and guiding the clinical application of botanical interventions in diabetes care.
Given these existing gaps in literature, the present study aimed to elucidate the antidiabetic efficacy of C. bonduc leaf extract specifically within an STZ-induced diabetic rat model, focusing explicitly on blood glucose regulation and pancreatic tissue integrity. The underlying hypothesis proposes that C. bonduc leaf extract effectively reduces hyperglycemia and promotes pancreatic recovery, attributing these outcomes primarily to the flavonoid content, renowned for its insulinotropic and antioxidant capabilities. By evaluating both biochemical parameters and histological changes in pancreatic tissue over a defined intervention period, this study sought to bridge critical knowledge gaps and substantiate the therapeutic utility of C. bonduc leaf extract, thereby laying foundational evidence for future translational and clinical research.
Material and methods
Subjects and experimental framework
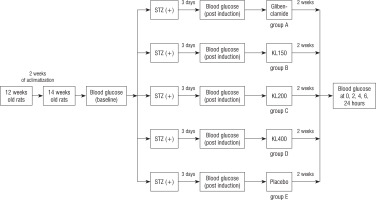
In this study, we used 25 male Wistar rats (Rattus norvegicus), aged 12–14 weeks and weighing 200–250 g. The Federer formula, (n-1)(t-1) ≥ 15, where n = sample size per group and t = number of treatments, was used to justify the sample size. All rats were healthy and free of disabilities. They were housed in suitably sized cages, maintained at 28 °C and under 40–60% humidity. Rats had ad libitum access to food and water. Rats were acclimatized for 2 weeks. During adaptation, rats were nourished with pellets of corn, coconut oil, soybeans, rice flour, fish meal, wheat pollard, tapioca, fish oil, and water ad libitum.
During acclimatization, rats were fed a high-fat diet. For the first 3 days, they were fed a basic mixed high-fat diet at a 1 : 1 ratio to aid digestion. On the fourth day, the proportion of high-fat diet was steadily increased to induce type 2 diabetes. Food and water were provided ad libitum twice a day.
After acclimatization, a diabetic rat model was established. Rats were fasted overnight to induce non-insulin-dependent diabetes mellitus. Streptozotocin was administered at a dose of 45 mg/kg BW as a single intraperitoneal injection.
The recommended dosage of streptozotocin for a typical adult weighing 70 kg is 45 mg. To induce diabetes mellitus, STZ, a well-established diabetogenic compound known to selectively destroy pancreatic β-cells, was administered intraperitoneally at a dosage of 45 mg/kg body weight, as previously described by Furman (2021) and Bajwa et al. (2022) [16, 17]. The conversion factor from humans to mice is 0.018. Therefore, for a rat weighing 300 g, the appropriate dose of streptozotocin would be 0.81 mg [18]. After 72 hours (3 days), hyperglycemia was confirmed by higher glucose levels in the plasma of the rats. Hyperglycemia was confirmed 3 days after STZ administration by measuring fasting blood glucose levels. Rats with blood glucose concentrations exceeding 200 mg/dl were selected for further experimentation [19].
The diabetic rats were subsequently randomized to five groups, each comprising five rats, using an online randomization tool to ensure unbiased distribution. Group A received glibenclamide (5 mg/kg body weight) and served as a positive control. Groups B, C, and D were administered C. bonduc leaf extract at doses of 150, 200, and 400 mg/kg body weight, respectively. Group E was administered sodium carboxymethyl cellulose (Na-CMC) as a placebo, functioning as a negative control. Treatments were administered once daily via a nasogastric sonde for two consecutive weeks. Na-CMC was specifically chosen as a suspension medium to enhance the bioavailability and intestinal absorption of the extract due to its colloidal properties, as documented by a previous study [20]. A schematic diagram of the in vivo experimental protocol is shown in Figure 1.
Caesalpinia bonduc (L.) Roxb extract preparation
Leaves of Caesalpinia bonduc (L.) Roxb were collected from the Bombana region in Southeast Sulawesi Province, Indonesia. Initial processing involved wet-sorting to remove contaminants, followed by thorough washing under running water. After washing, leaves were drained and air-dried, avoiding direct sunlight exposure to preserve phytochemical integrity. After drying, leaves were inspected to ensure the removal of all impurities, chopped, and accurately weighed.
The prepared plant material (simplicia) underwent extraction using a standard maceration technique as outlined by Xu et al. [21]. Approximately 2.5 liters of 96% ethanol was added to the simplicia in a sealed container. This mixture was stirred every 24 hours to enhance phytochemical extraction, continuing for three days. The resultant liquid was filtered to separate residues, yielding a concentrated extract after solvent evaporation using a rotary evaporator under reduced pressure. The final thick leaf extract was preserved in cool, dark conditions until further use.
Blood glucose measurement
Blood glucose levels were systematically monitored using an Easy Touch glucometer (Bioptik EasyTouch-GCU ET-301) from Bioptik Technology Inc, Taiwan, employing reagent strips that function based on glucose oxidase reactions. Blood samples (0.5–1.5 µl) were collected via tail vein puncture. Measurements were taken at several predetermined intervals: at baseline (pre-intervention), after induction with STZ (to confirm diabetes), and subsequently at 0, 2, 4, 6, and 24 hours following treatment administration, ensuring rigorous monitoring of glucose fluctuations.
Phytochemical screening
Qualitative phytochemical analyses were conducted to identify the presence of flavonoids, saponins, and tannins in C. bonduc leaf extracts. For flavonoid determination, 0.1 g of extract was dissolved in 10 ml of ethanol, followed by the addition of concentrated hydrochloric acid and magnesium powder. The emergence of red, orange, or yellow coloration within three minutes confirmed flavonoids. Detection of saponins involved boiling 1 g of extract for 2–3 minutes and vigorously shaking for 15 minutes to observe stable foam formation, indicating saponin presence. Tannins were detected by mixing 1 ml of the extract with 10% ferric chloride (FeCl3) solution; a dark blue or greenish-black color indicated the presence of tannins.
Histopathology preparation
After the 2-week treatment regimen, rats were euthanized, and pancreatic tissues were carefully excised for histopathological analyses. Extracted tissues were fixed using a 10% neutral-buffered formalin solution to preserve cellular morphology, dehydrated through graded ethanol series, cleared with xylene, and embedded in paraffin wax. Sections (approximately 3–5 µm thick) were cut using a microtome, mounted on slides, and stained with hematoxylin and eosin (H&E). The histological examination focused on identifying cellular alterations, including necrosis, hemorrhage, fibrosis, and inflammation.
Data analysis
All collected data were expressed as mean ±standard error of mean (SEM). Statistical analysis involved one-way analysis of variance (ANOVA) and repeated measures ANOVA to detect differences among experimental groups. Post-hoc analyses were performed using Dunn-Bonferroni correction. The significance level was set at p < 0.05. Statistical computations were conducted using SPSS v.26 software (IBM Corporation, New York, USA).
Bioethical standards
This study strictly adhered to ethical guidelines for animal research and conformed to the ARRIVE guidelines. Ethical clearance was obtained from the Health Research Ethics Committee of the Faculty of Medicine and Health Sciences at Universitas Muhammadiyah Makassar (Registration No: 512/UM.PKE/I/45/2024, approved January 2024). Ethical considerations were implemented throughout the experiment to ensure minimal discomfort to animals, aligning with international principles for animal biomedical research.
Results
Baseline characteristics of animals
The characteristics of animals used in this study were statistically evaluated at the beginning of the experimental phase. The initial characteristics, including age, body weight, and baseline blood glucose levels, demonstrated homogeneity across all treatment groups. Specifically, the age of the rats ranged from 12.6 ±0.89 to 13.4 ±1.14 weeks, body weight varied between 242.6 ±36.58 to 248.6 ±48.62 g, and baseline glucose levels were closely aligned, from 86.4 ±6.35 mg/dl to 89.6 ±7.40 mg/dl, with no statistically significant differences (p-values: 0.765, 0.999, and 0.936 respectively; Table I).
Table I
Characteristics of rats used in experiment
Overall glycemic changes across time
The repeated measures ANOVA revealed significant differences in blood glucose levels among all groups at various time points (p < 0.05), with the exception of Group E. Furthermore, the one-way ANOVA conducted to assess the mean differences among the groups at 0, 2, 4, 6, and 24 hours of observation indicated a statistically significant difference (p < 0.05).
Time-course by treatment groups
Following interventions, significant decreases in blood glucose levels were observed in Groups A (positive control with glibenclamide), B (KL150), C (KL200), and D (KL400). Specifically, Group B showed a notable reduction from 237.40 ±7.81 mg/dl at baseline to 140.80 ±6.09 mg/dl at 2 hours, further decreasing to 112.40 ±6.01 mg/dl and 103.00 ±4.60 mg/dl at 4 and 6 hours, respectively. Group C experienced a decline from 324.00 ±42.26 mg/dl at baseline to 222.80 ±44.00 mg/dl at 2 hours and continued reduction to 183.60 ±41.87 mg/dl and 199.40 ±67.73 mg/dl at subsequent measurements. Similarly, Group D showed substantial decreases, recording 214.40 ±23.72 mg/dl, 106.40 ±4.88 mg/dl, and 116.20 ±8.65 mg/dl at 2, 4, and 6 hours respectively, from an initial 336.00 ±23.34 mg/dl (Table II).
Table II
Mean level of blood glucose measurement after intervention at 0, 2, 4, 6, and 24 hours
Post-hoc pairwise comparisons
Post-hoc Dunn-Bonferroni tests further clarified these reductions, showing statistically significant differences in blood glucose levels when comparing intervention groups (B, C, D) to the placebo group (E). At 2 hours after treatment, Group B had a mean difference of 285.60 mg/dl (95% CI: from –402.64 to –168.56, p = 0.000), Group C a mean difference of 203.60 mg/dl (95% CI: from –108.64 to –125.44, p = 0.000), and Group D a mean difference of 212.00 mg/dl (95% CI: from –329.04 to –94.96, p = 0.000). These significant reductions were consistent at 4 and 6 hours. Notably, at 24 hours, although blood glucose levels rose slightly across all treated groups, they remained significantly lower compared to the placebo, confirming the sustained hypoglycemic effect of C. bonduc leaf extract (Table II).
Time-course visualization
The repeated measures ANOVA revealed significant differences in blood glucose levels among groups across various observation points (0, 2, 4, 6, and 24 hours), except for the placebo group (Group E). At the initial measurement (0 hours after treatment), elevated blood glucose levels were observed across all groups, indicative of the diabetic state induced by STZ (Figure 2).
Figure 2
Distribution of blood glucose levels across different observation times (0, 2, 4, 6, and 24 hours) for each treatment group
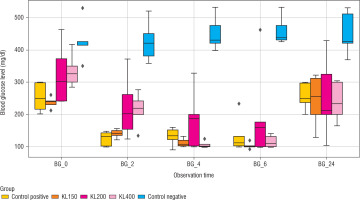
At 0 hours, elevated blood glucose levels were observed in all groups. Following the intervention, the positive control group (Group A) exhibited a significant reduction in blood glucose over time, with notable decreases evident as early as 2 hours after treatment and continuing through 24 hours. The C. bonduc leaf extract groups (KL 150, KL 200, KL 400) also experienced reductions in blood glucose levels, generally with a dose-dependent effect. The 200 mg/kg and 400 mg/kg doses (Groups C and D) were associated with larger declines compared to the 150 mg/kg dose, especially at 2 and 4 hours after treatment, suggesting potential hypoglycemic activity of the extract. In contrast, the negative control group (group E) maintained elevated blood glucose levels throughout the observation period, indicating no hypoglycemic effect in this group.
Phytochemical profile of the extract
Phytochemical screening confirmed the presence of bioactive compounds within the C. bonduc leaf extract. The qualitative analysis indicated positive results for flavonoids, saponins, and tannins, indicated respectively by the appearance of an orange color, formation of a stable emulsion, and a blackish-green coloration (Table III). These phytochemicals have been reported previously to exert significant hypoglycemic and pancreatic protective effects through mechanisms involving antioxidant activity, enhancement of insulin secretion, and modulation of glucose metabolism.
Pancreatic histopathology (hematoxylin and eosin)
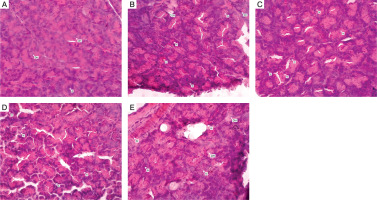
Figure 3 illustrates representative sections stained with hematoxylin and eosin. In the positive control group (glibenclamide), slight hydropic degeneration was observed, but the structure of acini appeared largely preserved (Figure 3A). Treatment with KL150 exhibited areas of hemorrhage, necrosis, and mild inflammation within pancreatic tissue (Figure 3B). However, higher doses of C. bonduc leaf extract (KL200 and KL400) demonstrated improved tissue integrity, reduced necrotic zones, and decreased hemosiderin deposits, indicating enhanced protective effects at elevated dosages (Figures 3C and 3D). In contrast, pancreatic sections from the placebo group displayed extensive pathological changes, including marked necrosis, hydropic degeneration, and pronounced inflammation, clearly illustrating the absence of therapeutic intervention effects (Figure 3E).
Figure 3
Histological presentation of pancreatic tissue stained with hematoxylin and eosin dye. A. Positive control: pancreatic section from rats treated with glibenclamide (400×). The pancreatic area exhibits slight hydropic degeneration (DH), while the acini formation appears normal. HE 400×. B. KL150: pancreatic section from rats treated with C. bonduc leaf extract at a dose of 150 mg/kg body weight after diabetic induction (400×). Throughout the tissue, hemosiderin is present, indicating the occurrence of bleeding, areas of necrosis (N), hydropic degeneration (DH), and a few inflammatory cells (R) within the acini. HE 400×. C. KL200: pancreatic section from rats treated with C. bonduc leaf extract at a dose of 200 mg/kg body weight after diabetic induction (400×). Some areas show necrosis (N) and the presence of hemosiderin (H). HE 400×. D. KL400: pancreatic section from rats treated with C. bonduc leaf extract at a dose of 400 mg/kg body weight after diabetic induction (400×). There are regions with necrosis (N) surrounding the acini cells (A) and the presence of hemosiderin (H). HE 400×. E. Negative control: pancreatic section from rats treated with placebo (400×). Several areas are experiencing necrosis (N) along with the loss of some tissue components, hydropic degeneration (DH), and the appearance of hemosiderin (H) around the acinar cells (A). HE 400×
Summary of key findings
Collectively, these results underline the potent hypoglycemic effects of C. bonduc leaf extract and its significant protective potential for pancreatic tissue integrity. The observed biochemical and histological improvements support its potential therapeutic application in diabetes management, aligning closely with documented activities of phytochemicals such as flavonoids, saponins, and tannins previously described in the literature.
Discussion
The administration of C. bonduc leaf extract demonstrated a significant hypoglycemic effect in streptozotocin (STZ)-induced diabetic rat models, indicating its potential as an effective agent for glycemic control. Consistent with previous studies, the extract notably lowered blood glucose levels at various measured intervals (0, 2, 4, 6, and 24 hours after treatment), reflecting substantial efficacy across doses of 150, 200, and 400 mg/kg body weight [22]. These reductions in glucose levels were particularly pronounced at intermediate observation times, reinforcing the possibility that the bioactive phytochemicals present, especially flavonoids, exert robust short-term hypoglycemic activity [14].
The phytochemical screening of the extract confirmed the presence of flavonoids, tannins, and saponins, reinforcing prior literature indicating the significant pharmacological roles of these phytochemicals in managing diabetes [23]. Flavonoids, identified by the characteristic orange coloration in qualitative tests, are particularly critical due to their documented abilities to modulate glucose metabolism by inhibiting α-glucosidase and enhancing insulin secretion [24]. Similarly, saponins and tannins contribute to glycemic regulation by modulating carbohydrate metabolism and exerting antioxidant effects, collectively enhancing overall metabolic control [25, 26].
Comparatively, previous studies focusing predominantly on seed extracts of C. bonduc have reported significant antidiabetic properties, highlighting differences in flavonoid profiles compared to leaf extracts [14, 27]. The leaf extract in our study showed comparable, if not superior, hypoglycemic efficacy, possibly attributable to different concentrations or combinations of bioactive compounds, underscoring the importance of plant part-specific phytochemical analyses in evaluating therapeutic potential [27].
The molecular mechanisms underlying the observed effects involve multiple biochemical pathways. Flavonoids, notably quercetin-like compounds abundant in C. bonduc, have been shown to promote insulin secretion via calcium influx in pancreatic β-cells and improve insulin sensitivity through activation of pathways such as AMP-activated protein kinase (AMPK) [28, 29]. Furthermore, flavonoids’ inhibition of digestive enzymes such as α-glucosidase substantially contributes to lowered postprandial glucose absorption, reflecting their multifaceted antidiabetic potential [30].
Histopathological examination of the pancreatic tissue revealed improved morphology, including a reduction in cellular necrosis, inflammatory infiltration, and structural derangements in the islets of Langerhans [31]. These observations suggest that the extract’s antioxidant and insulinotropic properties contribute to both improved blood glucose regulation and the preservation of pancreatic tissue integrity. Additionally, biochemical markers of oxidative stress, such as lipid peroxidation, were significantly reduced in the treated rats, further supporting the extract’s role in mitigating oxidative damage to pancreatic β-cells [32]. The extract’s flavonoid content, known for its antioxidant properties, likely plays a crucial role in protecting pancreatic tissue from oxidative damage, contributing to the preservation and regeneration of β-cell function [15].
Furthermore, histopathological analyses provided additional supportive evidence for the therapeutic benefits of the extract, revealing considerable improvements in pancreatic tissue integrity. Notably, there was observable amelioration of cellular necrosis, inflammation, and structural damage to the islets of Langerhans, correlating closely with biochemical indicators of improved glycemic control. The capacity of C. bonduc to confer both hypoglycemic effects and pancreatic tissue protection suggests a dual mechanism of action involving antioxidant and insulinotropic properties. This aligns closely with previous research emphasizing flavonoids’ capacity to mitigate oxidative damage and enhance insulin secretion [33]. The findings from this study align with existing research that highlights the dual action of C. bonduc leaf extract in reducing oxidative stress and enhancing insulin secretion [31, 34]. Previous studies have shown that flavonoid-rich plant extracts exert potent antioxidant effects, thereby protecting β-cells from oxidative damage and improving insulin secretion [33]. Another study reported that C. bonduc extract increases the expression of genes involved in insulin synthesis and secretion (Pdx-1, Ins-1, ngn-3), as well as glucose uptake (GLUT-4, IRS-1), and decreases the expression of stress-related genes (Mapk-8, Traf-4, Traf-6) [15]. Similar results have been observed with other plant-based therapies, such as Eugenia jambos, which also exhibited significant antihyperglycemic effects through improved glucose tolerance and reduced oxidative stress [31]. However, the unique combination of antioxidative and insulinotropic effects provided by C. bonduc offers a more comprehensive approach, addressing both hyperglycemia and pancreatic tissue integrity, which sets this extract apart from other botanical remedies.
The observed improvements in both biochemical and histological parameters following treatment with C. bonduc leaf extract suggest its potential as a viable complementary therapy in diabetes management [31, 32, 35]. A previous study found that administering C. bonduc extract enhances the structure of pancreatic tissue and increases β-cell mass in diabetic rats. Additionally, the extract promotes the regeneration of pancreatic β-cells and raises fasting insulin levels, thereby improving pancreatic function [35]. The extract’s multifaceted action, targeting both insulin secretion and cellular protection, offers a promising strategy for managing diabetes and its associated complications. Practically, the use of plant-based extracts such as C. bonduc could serve as an adjunct to conventional antidiabetic therapies, potentially reducing side effects and enhancing overall treatment efficacy [34]. From a scientific perspective, the molecular mechanisms underlying the extract’s actions, such as the regulation of antioxidant enzymes and enhancement of insulin secretion pathways, provide essential insights into how flavonoid-rich compounds can be harnessed to improve metabolic health in diabetic conditions [32]. The antioxidant activity of the extract protects against oxidative damage in pancreatic tissue, as evidenced by increased activities of catalase and superoxide dismutase, and reduced lipid peroxidation [4].
The protective roles of antioxidants in preserving pancreatic β-cell integrity emerged strongly from this study. By significantly reducing lipid peroxidation and enhancing activity of endogenous antioxidant enzymes such as superoxide dismutase (SOD) and catalase, antioxidants in C. bonduc extracts provide critical defense against oxidative stress, a major contributor to β-cell apoptosis and dysfunction [36]. Such antioxidative protection is essential for sustaining insulin production and secretion, ultimately aiding in glycemic control and pancreatic tissue preservation.
In comparison with other plant-based antidiabetic therapies, C. bonduc demonstrates robust dual efficacy in controlling blood glucose and protecting pancreatic tissues, akin to established treatments derived from Anacardium occidentale and Eugenia jambos [37, 38]. Notably, C. bonduc’s dual action addresses a broader therapeutic target, offering substantial promise for integrated diabetes management strategies. The unique blend of flavonoids, saponins, and tannins could provide a comprehensive approach, potentially superior to therapies targeting singular aspects of diabetes pathophysiology.
The broader implications of integrating herbal medicine, such as C. bonduc, into conventional diabetic therapy regimens are considerable. Herbal therapies offer significant advantages, including reduced side effects, improved patient adherence, and synergistic enhancement of conventional medications [39]. However, careful consideration of the safety profile, standardized dosages, and potential interactions with existing treatments remains crucial. Rigorous standardization and clinical validation are essential to facilitate broader clinical acceptance and effective integration into diabetic management protocols.
Several research gaps and future directions emerge from this study. Foremost, clinical trials are critically needed to verify the efficacy and safety of C. bonduc extracts in human diabetic populations. While animal models provide foundational insights, clinical validation through randomized controlled trials remains imperative for robust therapeutic endorsement [40]. Furthermore, long-term safety studies are essential to ensure sustainable use without unforeseen adverse effects, reinforcing patient safety across diverse demographics and clinical scenarios.
Additionally, deeper mechanistic investigations are necessary to unravel the intricate biochemical interactions underpinning C. bonduc’s effects. While current evidence identifies flavonoids as key active components, detailed exploration of their molecular interactions, receptor pathways, and cellular targets in diabetic contexts is required for comprehensive understanding and therapeutic optimization [41]. Comparative efficacy studies, evaluating Caesalpinia bonduc alongside other recognized herbal antidiabetic agents such as Gymnema sylvestre and Momordica charantia, could offer valuable insights into relative strengths, synergistic potentials, and optimized combination therapies [42].
In conclusion, the extensive biochemical and histopathological evidence presented strongly supports the therapeutic potential of Caesalpinia bonduc leaf extracts for diabetes management. Its demonstrated hypoglycemic efficacy and pancreatic protective effects align well with broader scientific literature on plant-derived antidiabetic agents, positioning it as a promising candidate for future therapeutic applications. Rigorous clinical evaluation, standardization, and comprehensive mechanistic studies will further solidify its role in diabetes care, contributing significantly to the evolving landscape of integrated herbal-conventional treatment strategies.
Conclusions
This study demonstrated significant hypoglycemic and pancreatic protective effects of C. bonduc leaf extract in streptozotocin-induced diabetic rats. Administration of the extract notably reduced blood glucose levels at doses of 150, 200, and 400 mg/kg, with marked improvements at intervals of 2, 4, and 6 hours after treatment. Histological analysis corroborated biochemical findings, revealing substantial reductions in pancreatic cellular necrosis and inflammation, and improved islet morphology. The presence of bioactive flavonoids, tannins, and saponins identified in phytochemical screening is likely responsible for these therapeutic outcomes. Flavonoids particularly exhibited potent insulinotropic and antioxidant activities, contributing significantly to enhanced insulin secretion, reduced oxidative stress, and protection of pancreatic β-cells. This dual-action profile distinguishes C. bonduc leaf extracts as a promising adjunct in diabetes management strategies. The study adds considerable value to existing knowledge by delineating leaf extract efficacy relative to the commonly investigated seed extracts, suggesting specific phytochemical advantages and broader therapeutic applicability. Future research directions include comprehensive clinical trials to establish safety and efficacy in humans, mechanistic studies to clarify the molecular pathways involved, and comparative analyses with other herbal therapies to evaluate synergistic potentials. Such investigations will further validate and potentially expand the therapeutic utility of C. bonduc, contributing substantially to integrated diabetes management.