Summary
This study is a real-world registry of high bleeding risk patients with an acute coronary syndrome treated with percutaneous coronary intervention and stent implantation. In-hospital all-cause mortality, cardiovascular mortality, and major bleeding complications are significantly higher in these patients compared to non-high bleeding risk. Transfemoral access during revascularization and baseline impaired kidney function are independent predictors of major bleeding in high bleeding risk patients. An estimated glomerular filtration rate value ≤ 50 ml/min was calculated to predict in-hospital major bleeding events with 87.5% sensitivity and 64.37% specificity.
Introduction
The prognosis of patients with an acute coronary syndrome (ACS) has improved in recent years. Percutaneous coronary intervention (PCI) with implantation of new-generation drug-eluting stents (DES) has emerged as the optimal treatment. However, high bleeding risk (HBR) patients who have undergone PCI have a worse prognosis and suffer more major bleeding events compared to non-HBR patients [1, 2]. Therefore, they receive either dual antiplatelet therapy (DAPT) for a shorter period of time [3], or a less potent P2Y12 inhibitor (clopidogrel instead of ticagrelor or prasugrel) [4], in order to reduce the bleeding complications. High bleeding risk patients are defined as those who undergo PCI and have a Bleeding Academic Research Consortium (BARC) 3 or 5 bleeding risk ≥ 4% or a risk of intracranial hemorrhage (ICH) of ≥ 1% at 1 year [5]. In addition, many contemporary bleeding risk scores have been created to identify HBR patients, but the most prevalent are the Academic Research Consortium for High Bleeding Risk (ARC-HBR) criteria [5].
Aim
The aim of this study was to quantify, in a real-world population with ACS treated with PCI, the prevalence of HBR patients, identify their prognosis, and record their bleeding and ischemic complications as well as their antithrombotic regimen compared with non-HBR. In addition, possible predictors of short-term major bleeding were also examined.
Material and methods
Study population
In this prospective, observational, single-center, real-world registry, 314 consecutive patients with an unplanned admission to the hospital were enrolled between January 2023 and July 2024. All the participants had suffered an ACS which included ST-elevation myocardial infarction (STEMI), non-ST-elevation myocardial infarction (NSTEMI) or unstable angina according to the Fourth Universal Definition of Myocardial Infarction of the European Society of Cardiology (ESC) guidelines [6]. Patients with a STEMI or with a very high risk NSTEMI diagnosis underwent an urgent coronary angiogram immediately after their admission. Following the latest European guidelines, very high risk NSTEMI patients were identified if they had one of the following criteria: hemodynamic instability or cardiogenic shock, chest pain refractory to medical treatment, acute heart failure as a result of ongoing ischemia, life-threatening arrhythmias or cardiac arrest after presentation, mechanical complications, and recurrent electrocardiographic changes due to ischemia (intermittent ST-segment elevation) [7]. All the other patients with NSTEMI or unstable angina who did not fulfill one of these criteria had a coronary angiogram within the first 24 h after their admission. Patients were considered eligible for the study if they had successful revascularization and at least one stent implantation during the PCI. The PCI was considered successful if the patients survived the operation, the stents were well expanded with Thrombolysis in Myocardial Infarction (TIMI) grade III flow in the target vessels at the end of the procedure, and the target lesions were fully covered with no distal edge dissections. Five interventional cardiologists were the primary operators of the procedures. Patients who were treated conservatively or were scheduled for coronary artery bypass graft (CABG) surgery as well as those who underwent a revascularization procedure but without a stent implantation, for example those treated only with thrombus aspiration, were excluded. All the patients were classified into HBR and non-HBR based on the ARC-HBR criteria [8].
Data collection and study design
A pre-specified form was used to record patients’ demographics (age, sex), their comorbidities (previous MI, heart failure, family history of coronary artery disease, diabetes mellitus, dyslipidemia, arterial hypertension, smoking, previous stroke and atrial fibrillation), echocardiographic findings, and blood examinations. Estimated glomerular filtration rate (eGFR) was calculated with Cockcroft-Gault formula. All the antithrombotic agents which were used during and after the PCI were also recorded. The patients were monitored during their hospitalization until their discharge, and all their thrombotic and bleeding complications during this period were also recorded (Figure 1). Prior to PCI, all patients received a loading dose of aspirin (325 mg) and during the procedure unfractionated heparin (100 IU/kg). The choice of the P2Y12 inhibitor – ticagrelor (180 mg), prasugrel (60 mg), or clopidogrel (600 mg) – was at the attending physician’s discretion during PCI. After the procedure, anticoagulation was discontinued except in patients who had an absolute indication such as those with atrial fibrillation or a thrombus in the apex of the left ventricle. The study was approved by the local ethics committee and was conducted in compliance with the principles outlined in the Declaration of Helsinki. The ARC-HBR criteria were used to classify the participants into HBR and non-HBR, and bleeding complications were classified using the BARC bleeding definition [9]. A comprehensive cardiac echocardiogram was obtained before discharge with a GE Vivid 7 ultrasound machine, and left ventricular ejection fraction (LVEF) was evaluated with Simpson’s biplane method.
Figure 1
Study design diagram. A total of 314 consecutive patients with an ACS (STEMI, NSTEMI, unstable angina) were enrolled after successful revascularization with PCI and at least one stent implantation. They were classified into HBR and non-HBR based on the ARC-HBR criteria and were monitored during their hospitalization. Endpoints included all-cause mortality, cardiovascular mortality, major bleeding events and thrombotic complications
ACS – acute coronary syndrome, ARC-HBR – Academic Research Consortium for High Bleeding Risk, BARC – Bleeding Academic Research Consortium, eGFR – estimated glomerular filtration rate, HBR – high bleeding risk, NSTEMI – non-ST-elevation myocardial infarction, PCI – percutaneous coronary intervention, STEMI – ST-segment elevation myocardial infarction.

Endpoints
The primary endpoint was in-hospital all-cause mortality. Secondary endpoints included in-hospital cardiovascular mortality, major bleeding complications (BARC ≥ 3) [9], and the cumulative endpoint of thrombotic complications (stent thrombosis or ischemic stroke). Possible factors to predict in-hospital severe bleeding events in HBR patients were also examined.
Statistical analysis
The Kolmogorov-Smirnov test was used to examine the normality of the data. Continuous variables with normal distribution are presented as the mean and standard deviation (SD) and were compared with the independent-samples t-test. Continuous variables lacking normal distribution are expressed as the median and interquartile range (IQR) and were compared with the Mann-Whitney U test. Categorical variables are presented as frequencies and percentages and were compared with the chi-squared (χ2) test. Odds ratios (ORs) with corresponding 95% confidence intervals (CIs) were measured with univariate and multivariate binary logistic regression models. Univariate logistic regression analyses were performed to detect possible factors predicting adverse bleeding events in HBR patients. All variables that were statistically significant in the univariate analysis were included in the multivariate analysis. Statistical significance was set at p < 0.05 (two-tailed), and all statistical analyses were performed with SPSS Statistics v. 22 (IBM Corp, Armonk, New York).
Results
A total of 314 consecutive patients, 103 HBR (32.8%) and 211 (67.2%) non-HBR, with an ACS were enrolled between January 2023 and July 2024 (Table I). The majority (60.8%) presented with STEMI, 35.1% with NSTEMI, and 4.14% with unstable angina. The distribution of these ACS subtypes did not differ significantly between the two groups (p = 0.06, p = 0.08, p = 0.657, respectively). HBR patients were older (median [IQR] age: 75 [64–82] years old), had lower hemoglobin levels (median [IQR] Hgb: 13 [11.1–14.7] g/dl), worse kidney function (mean [SD] eGFR: 63.2 [35] ml/min) and lower left ventricular ejection fraction (median [IQR] LVEF: 45 [35.00–50.00] %) compared to the non-HBR group. HBR patients had suffered more often a previous MI (p = 0.001), and had a history of heart failure (p < 0.001). In addition, 5 HBR patients (4.85%) were on dialysis at the time of the procedure, and in total 16 (15.53%) patients in the HBR group had an estimated eGFR < 30 ml/min. Baseline characteristics and clinical data of both groups are presented in detail in Table I. Regarding procedural data, radial access was the default approach for the PCI in the majority of the patients. In cases of inability to carry out the intervention through the radial artery, an access switch was performed. The procedure was completed through the radial access more frequently in the non-HBR group (p < 0.001), while the completion of the PCI through the femoral artery was more frequently performed in the HBR group (p < 0.001) (Table II). In addition, HBR patients had more complex coronary artery disease compared to non-HBR, which is proved by the significantly higher left main (LM) revascularization procedures rates (p < 0.001).
Table I
Baseline characteristics of patients with an ACS who underwent successful revascularization with PCI (n = 314)
[i] ACS – acute coronary syndrome, CAD – coronary artery disease, eGFR – estimated glomerular filtration rate, HBR – high bleeding risk, Hgb – hemoglobin, IQR – interquartile range, LVEF – left ventricular ejection fraction, MI – myocardial infarction, NSTEMI – non-ST-elevation myocardial infarction, SD – standard deviation, STEMI – ST-segment elevation myocardial infarction, UA – unstable angina.
Table II
Procedural characteristics of PCI interventions in patients with an ACS who underwent successful revascularization (n = 314)
[i] ASA – acetylsalicylic acid, GP – glycoprotein, HBR – high bleeding risk, IQR – interquartile range, LAD – left anterior descending, LCx – left circumflex, LM – left main, OAC – oral anticoagulant, PCI – percutaneous coronary intervention, RCA – right coronary artery, UFH – unfractionated heparin.
In-hospital mortality (OR = 7.235, 95% CI: 2.776–18.857, p < 0.001) and cardiovascular mortality (OR = 4.935, 95% CI: 1.818–13.396, p < 0.001) were significantly higher in the HBR group (Table III). Although death from refractory cardiogenic shock was the leading cause of death in both groups, its incidence was higher in HBR patients compared with non-HBR (11.65% vs. 1.42%, p < 0.001). Moreover, HBR patients suffered more bleeding BARC ≥ 3 events (OR = 9.517, 95% CI: 3.093–29.283, p < 0.001), without any difference in the cumulative thrombotic endpoint of stent thrombosis or stroke (Table III). More specifically, patients with at least one major ARC-HBR criterion (OR = 7.243, 95% CI: 2.769–18.947, p < 0.001) or two minor HBR criteria (OR = 3.636, 95% CI: 1.436–9.208, p = 0.006) suffered more severe bleeding complications. All patients received the DES Resolute Integrity (Medtronic) or the DES Promus Premier (Boston Scientific) except 6 (5.82%) patients in the HBR group, who received stents with ultra-thin struts (Coroflex ISAR NEO, Braun).
Table III
Comparison of in-hospital mortality, cardiovascular mortality, and bleeding BARC ≥ 3 between HBR and non-HBR patients
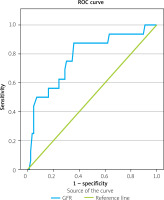
Univariate binary logistic regression analysis was performed to detect possible predictors of in-hospital bleeding (BARC ≥ 3) in HBR patients. Only trans-femoral access in PCI and eGFR were statistically significant (Table IV), and these predictors were included in a multivariable model. Both the trans-femoral access (OR = 3.524, 95% CI: 1.035–11.991, p = 0.044) and eGFR (OR = 0.966, 95% CI: 0.938–0.996, p = 0.027) maintained their significance (Table IV). A ROC curve was plotted to identify the optimal cut-off value of eGFR. The curve displayed a statistically significant AUC value of 0.781 (95% CI: 0.658–0.91, p < 0.001) (Figure 2). The optimal cut-off point for eGFR predictive for bleeding was calculated. An eGFR value ≤ 50 ml/min was found to predict in-hospital major bleeding events in HBR patients with an ACS and successful revascularization with 87.5% sensitivity and 64.37% specificity.
Table IV
Predictors of in-hospital bleeding (BARC ≥ 3) in HBR patients with an ACS who underwent successful revascularization with PCI
[i] ACS – acute coronary syndrome, ASA – acetylsalicylic acid, BARC – Bleeding Academic Research Consortium, CAD – coronary artery disease, eGFR – estimated glomerular filtration rate, GP – glycoprotein, HBR – high bleeding risk, Hgb – hemoglobin, IQR – interquartile range, LAD – left anterior descending, LCx – left circumflex, LM – left main, LVEF – left ventricular ejection fraction, MI – myocardial infarction, NSTEMI – non-ST-elevation myocardial infarction, OAC – oral anticoagulant, PCI – percutaneous coronary intervention, RCA – right coronary artery, STEMI – ST-segment elevation myocardial infarction, UA – unstable angina, UFH – unfractionated heparin.
All patients were treated either with DAPT (aspirin and a P2Y12 inhibitor) or, if anticoagulation was indicated, with triple antithrombotic therapy: aspirin, clopidogrel, and an oral anticoagulant (OAC). Non-HBR patients received potent P2Y12 inhibitors (ticagrelor and prasugrel) more frequently than HBR (p < 0.001 and p = 0.004 respectively) (Table II). The two groups did not differ in clopidogrel use (p = 0.173). One non-HBR patient received during his hospitalization intravenous cangrelor and died from a mechanical complication (ventricular septal rupture) even though the revascularization was successful. In addition, although the antithrombotic regimen used in the HBR group was not associated with bleeding complications (Table IV), clopidogrel was more frequently used in patients with severely impaired kidney function (eGFR < 40 ml/min) (clopidogrel OR = 3.45, 95% CI: 1.175–10.129, p = 0.02, ticagrelor OR = 1.24, 95% CI: 0.495–3.119, p = 0.64, prasugrel p = 0.998), as well as in patients with ≥ 2 major or ≥ 3 minor ARC-HBR criteria (clopidogrel OR = 5.107, 95% CI: 1.748–14.919, p = 0.003, ticagrelor OR = 0.457, 95% CI: 0.185–1.128, p = 0.089, prasugrel p = 0.998).
Discussion
The main findings of our study can be summarized as follows: 1) in this real-world registry, 32.8% of the patients with an ACS who undergo PCI are at high bleeding risk; 2) in-hospital major bleeding events, all-cause mortality and cardiovascular mortality are significantly higher in HBR patients with ACS and successful PCI compared to non-HBR; 3) the only factors that can predict in-hospital bleeding BARC ≥ 3 in HBR patients with ACS who undergo PCI are the use of femoral access during the intervention as well as the baseline impaired kidney function; 4) an e-GFR value ≤ 50 ml/min was calculated to predict in-hospital major bleeding events in HBR patients with an ACS and successful revascularization with 87.5% sensitivity and 64.37% specificity; 5) although the choice of the antithrombotic agents was not associated with bleeding events, clopidogrel was more frequently used in HBR patients who were deemed at higher bleeding risk and in HBR patients with severely impaired kidney function (eGFR < 40 ml/min).
Patients at high bleeding risk have a significantly worse prognosis compared with non-HBR patients. Recent studies have identified that HBR patients experience higher all-cause mortality (4.7% vs. 0.6%, p < 0.001) [10] and more serious bleeding events both in hospital during the periprocedural period (4.3% vs. 0.5%, p < 0.001) [11] and in 1-year follow-up (9.1% vs. 3.2%, p < 0.001) [10]. In our study, both in-hospital mortality (17.4% vs. 2.84%, p < 0.001) and bleeding during the periprocedural period (15.5% vs. 1.89%, p < 0.001) were also significantly higher in the HBR group compared with non-HBR. The higher death rates can be explained by the fact that the HBR patients were older (median [IQR] age: 75 [64–82] years) and had severe comorbidities. More specifically, HBR patients had suffered more frequently a previous MI (p = 0.001) as well as a previous stroke (p < 0.001) and more often had a history of heart failure (p < 0.001). In addition, although the radial access was always the default access site for revascularization, PCI was completed through the femoral access more frequently in the HBR group (p < 0.001). While this resulted in more access site bleeding, it also proved that PCI procedures in HBR patients were also more complicated and challenging, with significantly higher left main (LM) revascularization procedure rates (p < 0.001).
New-generation DES with ultra-thin struts have been identified as the best choice for HBR patients undergoing PCI in order to shorten the DAPT duration without increasing the ischemic complications. In the MASTER DAPT (Dual Antiplatelet Therapy after PCI in Patients at High Bleeding Risk) trial [12], all high bleeding risk patients received a biodegradable polymer sirolimus-eluting stent (Ultimaster, Terumo). In this study, almost half of the participants (48.3%) had undergone PCI for an ASC. One month of DAPT (abbreviated group) resulted in a lower incidence of major or clinically relevant nonmajor bleeding in comparison with the standard therapy of at least 2 more months. The two groups did not differ in the thrombotic complications (composite endpoint of all-cause death, MI or stroke, p = 0.001 for noninferiority). Other contemporary DESs have also been approved for HBR patients, such as the cobalt-chromium everolimus-eluting stent (XIENCE, Abbott) [13], the Synergy bioresorbable-polymer everolimus-eluting stent (Boston Scientific Corporation, Marlborough, MA) [14], the zotarolimus-eluting Resolute Onyx stent (Medtronic) [15], and the biodegradable polymer sirolimus-eluting stent Orsiro (Biotronik AG, Bülach, Switzerland) [16]. In our registry, only 5.82% of the HBR group received stents with ultra-thin struts (Coroflex ISAR NEO, Braun). This can probably be explained by the fact that HBR patients who undergo PCI are underdiagnosed in everyday clinical practice.
Chronic kidney disease (CKD) is one of the most well-established factors for the identification of HBR patients who undergo PCI both in ARC-HBR criteria as well as in the PRECISE-DAPT (predicting bleeding complications in patients undergoing stent implantation and subsequent dual antiplatelet therapy) score [17]. In a sub-study of the AQUITY (Acute Catheterization and Urgent Intervention Triage strategY) trial, ACS patients with CKD (creatinine clearance < 60 ml/min) had worse 30-day and 1-year clinical outcomes than those with normal renal function, with significantly more major bleeding events [18]. Although in this trial patients with end-stage renal function were excluded, in our registry, 16 (15.53%) patients in the HBR group were on dialysis or had an eGFR < 30 ml/min. In addition, in the HORIZONS-AMI (Harmonizing Outcomes With Revascularization and Stents in Acute Myocardial Infarction) Trial, STEMI patients with CKD also had a significantly worse prognosis as well as more major bleeding events compared with those without CKD [19]. Our study confirmed the existing findings of the former trials and identified that CKD with an eGFR value ≤ 50 ml/min could be an independent predictor of short-term major bleeding in HBR patients with ACS and successful PCI. In addition, in the CRUSADE (Can Rapid risk stratification of Unstable angina patients Suppress ADverse outcomes with Early implementation of the ACC/AHA guidelines) bleeding score, which is used to predict in-hospital major bleeding complications in patients with NSTEMI, a median creatinine clearance of 48.2 (30.1, 73.4) ml/min is associated with major bleeding [20]. These results are in line with the findings of our study in HBR patients, but due to its low specificity, the eGFR cut-off value of our study should be interpreted cautiously. Moreover, it is well established that femoral access should be avoided in order to reduce bleeding complications and mortality after PCI [21].
Although many recent studies have supported abbreviated one-month DAPT as a safe solution for HBR patients with ACS and coronary stenting [12, 22–25], it is not yet clear which P2Y12 inhibitor should be administered in addition to aspirin in HBR patients after PCI. The investigation of the optimal P2Y12 inhibitor has only been made in elderly patients with an ACS, not in HBR patients, with ambiguous results. In the open-label, randomized, controlled POPular AGE trial, clopidogrel, in patients aged 70 years or older with NSTEMI, resulted in fewer major bleeding events, without any differences in all-cause death, cardiovascular death, MI, or ischemic stroke compared with ticagrelor or prasugrel [26]. In addition, Szummer et al. noted in the SWEDEHEART registry that ticagrelor use was associated with an increased risk of death and bleeding at one year compared with clopidogrel in elderly patients ≥ 80 years old who suffered a myocardial infarction [27]. These results contrast with a substudy of the PLATO trial in which ticagrelor proved to be equally safe as clopidogrel in elderly patients (≥ 75 years old) with an ACS, with no statistically significant increase in major bleeding [28].
In our study, neither use of potent P2Y12 inhibitors (ticagrelor, prasugrel) nor triple antithrombotic therapy consisting of aspirin, clopidogrel, and an OAC in HBR patients resulted in augmentation of the major bleeding BARC ≥ 3 events during hospitalization. This can be explained by the fact that the follow-up period was short and the antithrombotic regimen was provided at the discretion of the interventional cardiologist during PCI. Therefore, in HBR who were considered more likely to suffer a major bleeding event, clopidogrel was used. More specifically, HBR patients with ≥ 2 major or ≥ 3 minor ARC-HBR criteria were treated with clopidogrel (OR = 5.107, 95% CI: 1.748–14.919, p = 0.003). In addition, in HBR patients with severely impaired renal function (eGFR < 40 ml/min) who in accordance with the multivariable analysis were also more likely to suffer a severe bleeding complication, clopidogrel was again the favorable antiplatelet agent (clopidogrel OR = 3.45, 95% CI: 1.175–10.129, p = 0.02). Despite the conflicting results of the randomized control trials, clopidogrel remains a safe choice in the HBR population. Even in the substudy of the PLATO trial, the absolute increase of major bleeding events with ticagrelor versus clopidogrel was numerically greater in elderly (1.2%) versus younger patients (0.7%) [28].
The study has several limitations. The observational design of the study is one major limitation. Moreover, the choice of the DAPT was at the attending physician’s discretion during PCI, without a standardized assessment of the bleeding risk of all the participants. One major limitation is the low number of patients enrolled (n = 314), which affects the detection of additional predictors of in-hospital bleeding events. Bleeding complications were classified using the BARC bleeding definition, and data collection was affected by the choice of the bleeding classification system used. Also, the short-term follow-up of the patients (only during their hospitalization) minimizes the number of adverse events, and a longer observation period might have been more clinically meaningful. In addition, the multivariate model for predicting in-hospital severe bleeding events in the HBR group was based on 16 major bleeding events. Therefore, the results of the multivariate analysis and the eGFR cut-off point should be interpreted as exploratory. The study was conducted in a single center, and potential differences in other centers’ practices, such as the threshold to proceed with PCI in all HBR patients, might also have affected the results. The patients were classified into HBR and non-HBR based on the ARC-HBR criteria, and the results should not be generalized to other populations.
Conclusions
In-hospital all-cause mortality, cardiovascular mortality, and bleeding BARC ≥ 3 are significantly higher in HBR patients with an ACS (STEMI, NSTEMI, and unstable angina) compared to non-HBR. Baseline impaired renal function and transfemoral access are independent predictors of severe bleeding. An e-GFR value ≤ 50 ml/min was calculated to predict in-hospital major bleeding events in HBR patients with an ACS and successful PCI with 87.5% sensitivity and 64.37% specificity. More studies and randomized controlled clinical trials are needed to determine the optimal duration of DAPT after PCI in these patients and the optimal DAPT combination to reduce bleeding complications.