Summary
Severely calcified left main (LM) coronary lesions represent a high-risk subset of patients for whom percutaneous coronary intervention (PCI) is technically challenging, and current evidence on intravascular lithotripsy (IVL) in this setting is limited. In this study, IVL use in LM PCI was associated with high procedural success and long-term outcomes, including mortality and major adverse cardiovascular events rates, comparable to non-LM interventions, despite greater anatomical complexity and higher procedural demands. These findings suggest that IVL provides a safe and effective option for LM PCI, expanding treatment opportunities.
Introduction
Coronary artery bypass grafting (CABG) has traditionally been considered the standard revascularization strategy for significant left main (LM) coronary artery disease. Current European guidelines, however, support percutaneous coronary intervention (PCI) in LM lesions with low or intermediate SYNTAX scores [1]. Given that the LM trunk supplies an extensive myocardial territory, urgent PCI is frequently performed in high-risk patients.
Revascularization of the LM poses unique technical challenges. Ostial LM disease is complicated by marked vessel wall rigidity and elastic recoil, which may contribute to less favorable acute and long-term results compared with non-ostial lesions [2]. Distal LM involvement typically requires bifurcation PCI, adding procedural complexity and elevating the risk of peri-procedural complications [3]. Furthermore, calcification – present in approximately 20–25% of LM lesions [4] – can impede device crossing and stent expansion.
Adequate plaque modification is therefore essential. Contemporary approaches can be broadly classified into balloon-based systems, such as non-compliant, high-pressure, and cutting or scoring balloons, which exert high radial forces, and atherectomy techniques, including rotational, orbital, and laser atherectomy, which achieve plaque debulking [5]. Although angiographic success rates exceed 90%, each technique has inherent limitations and safety concerns [6], sustaining interest in alternative approaches.
Intravascular lithotripsy (IVL) represents one such alternative. The Shockwave C2 and C2+ IVL systems (Shockwave Medical, Santa Clara, CA, USA) convert electrical energy into acoustic pulses that fracture both superficial and deep calcium, improving vessel compliance and facilitating optimal stent deployment. Most safety and efficacy data for IVL originate from peripheral artery studies and small real-world coronary registries [7]. Evidence specific to the LM remains scarce and is drawn largely from retrospective series or under-powered sub-analyses [8, 9].
Aim
The present investigation was therefore designed to assess procedural success, efficacy, safety, and 2-year clinical outcomes of IVL in calcified LM stenosis.
Material and methods
The analysis included 53 patients who underwent LM PCI using the Shockwave C2 and C2+ Intravascular Lithotripsy systems (Shockwave Medical Inc, Santa Clara, CA, USA) in our department beginning in April 2020. Outcomes were compared with those of 148 patients who underwent PCI for non-LM lesions during the same period.
Significant LM disease was defined as ≥ 50% diameter stenosis of an unprotected LM, with or without concomitant involvement of the left anterior descending (LAD) ostium, the left circumflex (LCx) ostium, or both. For intermediate lesions, intravascular ultrasound (IVUS) was performed, and a minimal lumen area (MLA) < 6.0 mm2 was considered significant.
All procedures were carried out at a high-volume tertiary referral center with on-site cardiac surgery, following multidisciplinary Heart Team evaluation. IVL was used for severely calcified lesions unresponsive to conventional plaque-modification strategies, namely non-compliant (NC) balloon inflation at ≥ 16 atm or rotational atherectomy. Procedural decisions – including timing, balloon sizing, maximum inflation pressure, and the use of additional debulking devices – were left to the discretion of the operator. In the LM group, IVL balloons with a diameter of 3.5 mm were used in 58.5% of patients, while 4.0 mm balloons were applied in 15.1%. Lesions were classified as resistant if ≥ 20% under-expansion persisted after high-pressure NC balloon dilation [10]. Severe calcification was diagnosed angiographically as fixed radiopaque densities ≥ 15 mm in length involving both vessel walls, or by IVUS/optical coherence tomography (OCT) demonstrating a calcium arc ≥ 270° [11].
Procedural success was defined as optimal deployment of a new stent or effective post-dilatation of an under-expanded stent, with < 20% residual in-stent stenosis and final TIMI grade 3 flow [12]. The primary short-term endpoint was a composite of in-hospital death or myocardial infarction. Periprocedural safety endpoints included coronary perforation, slow- or no-reflow, de novo intracoronary thrombus, ventricular tachyarrhythmias, acute vessel closure, unsuccessful stent delivery, inadequate stent expansion, or device-related failure (inability to cross the target lesion or mechanical malfunction).
The clinical and angiographic characteristics of the patients were assessed alongside both short- and long-term outcomes. Baseline clinical data were collected at the time of the initial procedure, while procedural details, including periprocedural and in-hospital complications, were systematically documented and analyzed.
Baseline demographic, clinical, and angiographic characteristics were recorded at the index procedure. Procedural details, as well as periprocedural and in-hospital complications, were documented. Follow-up data (mean: 557 days; median: 445 days, interquartile range: 491 days) were obtained by telephone interview or through National Health Fund records.
The study was conducted in accordance with the Declaration of Helsinki (1975) and received approval from the Institutional Review Board and Bioethics Committee of Poznan University of Medical Sciences.
Results
A total of 201 patients were analyzed: 53 in the LM-IVL cohort and 148 in the non-LM cohort (Table I). The two groups were similar in age (69.6 ±8.1 vs. 69.9 ±8.1 years; p = 0.91) and male sex (75.5% [40/53 patients] vs. 73.0% [108/148 patients]; p = 0.72). Clinical presentation differed, with non-ST elevation myocardial infarction (NSTEMI) more frequent in the LM group (43.4% vs. 23.6%; p = 0.006), whereas rates of stable angina, unstable angina and ST elevation myocardial infarction (STEMI) were comparable. Body mass index was lower among LM patients (26.9 ±4.0 vs. 29.2 ±6.0 kg/m2; p = 0.005). Heart failure history was more common in the LM cohort (47.2% vs. 29.1%; p = 0.017). Other comorbidities – including hypertension, dyslipidemia, diabetes (overall or insulin-treated), chronic kidney disease, chronic obstructive pulmonary disease (COPD) and prior stroke/transient ischemic attack (TIA) – showed no significant differences. The LM group had substantially higher anatomical complexity, with a median SYNTAX score of 26.7 ±9.1 vs. 15.0 ±9.6 (p < 0.001). Left-ventricular dimensions and ejection fraction were similar between cohorts.
Table I
Baseline characteristics
[i] MI – myocardial infarction, PCI – percutaneous coronary intervention, LAD – left anterior descending coronary artery, LCX – left circumflex coronary artery, RCA – right coronary artery, CABG – coronary artery bypass graft, CKD – chronic kidney disease, HF – heart failure, COPD – chronic obstructive pulmonary disease, TIA – transient ischemic attack, LVEDD – left ventricle end-diastolic diameter, LVEF – left ventricular ejection fraction.
By definition, all LM patients underwent intervention to the left main artery, but they also more often required adjunctive treatment of the LAD (73.6% vs. 47.3%; p < 0.001) and LCx (54.7% vs. 12.8%; p < 0.001) (Table II). Bifurcation disease was markedly more prevalent (83.0% vs. 17.6%; p < 0.001). Stent-under-expansion and in-stent restenosis rates were comparable. Chronic total occlusion, lesion length > 20 mm and prior rotational atherectomy were similarly distributed.
Table II
Procedure characteristics
[i] LM – left main coronary artery, LAD – left anterior descending artery, LCX – left circumflex coronary artery, RCA – right coronary artery, CTO – chronic total occlusion, IVL – intravascular lithotripsy, DES – drug-eluting stent, IVUS – intravascular ultrasound, OCT – optical coherence tomography, MLA – minimal lumen area, MSA – minimal stent area, MI – myocardial infarction.
Larger IVL balloon diameters were chosen in LM lesions, with less use of 3.0 mm balloons (24.5% vs. 44.6%; p = 0.016) and more use of 4.0 mm balloons (15.1% vs. 2.0%; p < 0.001). Postdilatation was performed in all cases. Accordingly, more drug-eluting stents were implanted (1.7 ±1.1 vs. 1.1 ±1.0; p < 0.001) with a greater cumulative stent length (38.5 ±26.8 vs. 28.1 ±26.5 mm; p < 0.001). Intravascular imaging (IVUS/OCT) was performed in nearly every LM case (94.3% vs. 40.5%; p < 0.001). Minimal lumen area in LM PCI increased from mean 4.2 (1.2) mm2 to 10.5 (3.6) mm2. Radial access rates did not differ, but LM PCI more often required ≥ 7 F guides (86.8% vs. 33.8%; p < 0.001) and generated a higher radiation dose (1731 ±1133 vs. 1365 ±1000 mGy; p = 0.025) and greater contrast use (267 ±111 vs. 213 ±86 ml; p = 0.004). Mechanical circulatory support was needed in 13.2% of LM cases and in none of the non-LM cases.
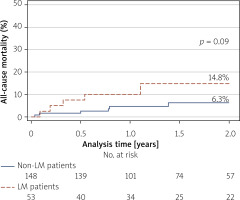
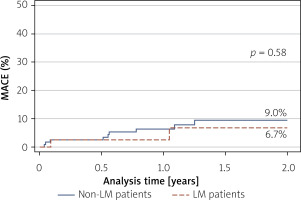
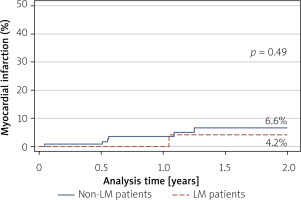
Despite these differences, IVL success rates were high in both groups (100% vs. 96.6%; p = 0.40), as were overall procedural success rates (100% vs. 99.3%; p = 0.59). No cases of coronary artery perforation or slow-flow/no-reflow phenomena were recorded. During long-term follow-up, there were no significant differences between the LM and non-LM cohorts in all-cause mortality (14.8% vs. 6.3%; p = 0.09) (Figure 1), major adverse cardiovascular events (MACE) (cardiac death, myocardial infarction, stroke incidence) (6.7% vs. 9.0%; p = 0.58) (Figure 2), or myocardial infarction rates (4.2% vs. 6.6%; p = 0.49) (Figure 3).
Discussion
To our knowledge, this pilot analysis offers one of the longest follow-up observations reported to date for IVL in LM disease. Patients undergoing PCI for LM lesions constitute a high-risk cohort. The LM supplies more than 75% of the left ventricular (LV) myocardial blood flow, even in individuals with a dominant right coronary artery (RCA) [13]. The extensive ischemic territory, combined with the technical challenges inherent to LM intervention, is reflected in current guidelines, which recommend CABG as the preferred mode of revascularization [1, 14]. The presence of severe calcification further increases procedural complexity, contributing to higher rates of periprocedural complications, difficulties in stent delivery, and suboptimal stent expansion – factors that adversely affect both short- and long-term clinical outcomes [15].
Despite that fact, PCI often remains the last therapeutic option for patients who are not suitable for surgery. Therefore, the real risk is imprecise due to the lack of these patients in large randomized clinical trials. The personalized patient-to-patient Heart Team decision with detailed analysis of the individual clinical status and cardiac and non-cardiac comorbidities is crucial here [16]. Ostial disease is particularly problematic because the rigid aortic wall resists expansion and readily recoils, so aggressive plaque preparation with specialty balloons and high-pressure post-dilatation is often indispensable [17]. When the distal left main artery bifurcates into the LAD and LCx, the operator must deal with complex branching anatomy, which usually prolongs the procedure and necessitates the use of large-bore guiding catheters, microcatheters and hydrophilic guidewires. Approximately one-quarter of left-main procedures encounter heavy calcification, a substrate that impairs stent expansion and increases the risk of intraprocedural complications [4]; therefore, meticulous calcium modification is required.
In a large multicenter study by Riley et al. focusing on LM IVL PCI, acute safety events were infrequent. In-hospital MACE occurred in 4.4%, primarily driven by all-cause mortality (3.9%) and a low incidence of periprocedural myocardial infarction (2.2%); no cases of target-vessel revascularization (TVR) were reported. Angiographic complications – including perforation, abrupt vessel closure, and slow- or no-reflow – were each observed in ≤ 1% of cases. By 30 days, the cumulative MACE rate increased modestly to 8.8%, again largely attributable to death (7.2%) and non-fatal myocardial infarction (3.3%), with no early TVR. These outcomes were accompanied by a high procedural success rate of 90.6% [8]. Cosgrove et al. reported on 31 patients who underwent PCI with IVL for left main artery disease [9]. Procedural success was achieved in 97% [9]. No angiographic complications or in-hospital MACE (death, myocardial infarction, or target vessel revascularization) were observed. At 30 days, one (3.2%) non–ST-elevation MI occurred [9]. Rola et al. investigated IVL for heavily calcified, unprotected left-main stenoses in a cohort of 15 patients (mean SYNTAX score 23.3 ±13.0). Major adverse cardiac events remained low in the S-IVL arm: 6.7% during the index hospitalization and 13.3% at 6 months [18, 19]. Oliveri et al. analyzed 59 left-main cases alongside a non-LM comparator. Technical success did not differ, achieving 93.2% in the LM cohort and 89.8% in non-LM cases (p = 0.41). At 12 months, all-cause mortality was 11.9% [20]. Hesse et al. presented data on 44 LM patients treated with IVL, reporting a 1-year MACE rate of 22.7% [21]. Mechanical circulatory support was not used in this population [21]. IVUS was performed in 77.3% of cases, and OCT in 11.4% [21]. A recent meta-analysis by Lingamsetty et al., including 10 studies with 435 patients with calcified LM disease, demonstrated very high procedural success rates with IVL (98.8%) and low complication rates, including perforation (0.02%), major dissection (0.87%), and vessel closure (0.01%) [22]. Short-term outcomes were also favorable, with low rates of in-hospital and 30-day adverse cardiovascular events (MACE 4.79%, mortality 3.09%) [22]. In the multicenter registry reported by Riley et al., major adverse cardiac events occurred in 4.4% of patients before discharge and in 8.8% at 30 days [8]. IVL produced a marked luminal expansion, with the minimal lumen area rising from 4.1 ±1.3 mm2 to a post-PCI minimal stent area of 9.3 ±2.5 mm2 (p < 0.001). Oliveri et al. reported that, in the left-main cohort, the post-procedural minimal stent area (MSA) measured 10.90 ±0.46 mm2 [20]. In our study, pre-procedural MLA increased from 4.2 mm2 in the LM group to post-procedural MSA of 10.5 mm2. Although this more than two-fold increase is encouraging, the final MSA remained below the 11.8 mm2 benchmark (by Kim et al.) generally linked to favorable long-term outcomes [23]. In the study by Kim et al., which focused on optimal minimal stent area and the impact of stent underexpansion in the left main, the distribution of MSA within the distal LM (10.9 ±2.2 mm2, median 10.8 mm2) was comparable to that reported in previous studies conducted in Western populations and was not predictive of cardiac events, likely because it was already sufficiently large not to influence the prognosis [23–25]. Moreover, in the NOBLE study the mean MSA was 12.5 ±3.0 mm2, and in the EXCEL study 9.9 ±2.3 mm2; however, these studies did not specifically focus on patients with extensive calcification in the LM [24, 25].
Riley et al. reported that mechanical circulatory support with the Impella device was employed in roughly one quarter of left-main PCI cases (24.7%), typically as part of a planned protected-PCI approach [8]. In the study by Oliveri et al., the temporary mechanical circulatory support, mainly within a protected-PCI strategy, was employed significantly more often for left-main interventions (6.8% vs. 2.1%; p = 0.04) [20]. In our series, Impella or iVAC devices were used in 13.2% of LM + IVL interventions.
Our long-term outcome data are consistent with previously published IVL reports. The relatively high 2-year all-cause mortality in the LM group (14.8%) may be attributable to the presence of severe calcification, a factor known to adversely affect both short- and long-term clinical outcomes [15, 26]. Ongoing documentation and evaluation of results in these small, high-risk patient populations are essential to further refine the role of IVL in LM disease.
This study has several limitations. First, the study’s observational design inevitably exposes it to selection and confounding biases. Second, intracoronary imaging was not performed in every case; that said, the imaging rate is comparable to other contemporary registries, especially high in the LM group. Although our cohort is among the largest yet reported, it remains relatively small. The sample size was not designed to assess long-term clinical endpoints from the outset, as this study is a prospective analysis of patients who underwent PCI with Shockwave C2 and C2+ IVL in our department. Finally, heterogeneity between the groups may have influenced our findings. The non-left-main cohort was included to provide a broader clinical context.