A 69-year-old male patient with persistent atrial fibrillation, hypertension, and asthma was initially admitted to a neurology ward due to a second episode of ischemic stroke, presenting with left hemiparesis. This occurred despite acenocoumarol therapy, which was monitored every 2 weeks and remained for 75% of the time within the therapeutic range. On admission, besides acenocoumarol, the patient was taking a β-blocker, ACE inhibitor, statin, and anti asthmatic drugs. However, according to the patient, an oral anticoagulant was not used for a few days before the current episode (international normalized ratio (INR) on admission was 1.6). The stroke was treated with intravenous thrombolysis, followed by percutaneous mechanical thrombectomy of the right middle cerebral artery. In-hospital follow-up imaging revealed several foci of intracranial hemorrhage (ICH) and post-hemorrhagic foci within the left cerebral hemisphere, suggesting probable cerebral amyloid angiopathy (CAA) based on modified Boston criteria (Figure 1 A) [1]. The neurological deficit resolved during hospitalization. Chest X-ray, abdominal ultrasonography, blood tests, and tumor markers excluded coagulation disorders and overt malignancy. The patient was deemed suitable for left atrial appendage closure (LAAC) due to the increased risk of recurrent intracranial bleeding and a history of ischemic strokes on anticoagulation.
Figure 1
A – A non-contrast computed tomography scan showing multiple hemorrhages in the left hemisphere (white arrows). B – Transesophageal echocardiography image depicting thrombus presence in the left atrial appendage (red arrow). C – Introduction of a Sentinel System in the brachiocephalic artery and left common carotid artery for cerebral protection. D – Angiographic and echocardiographic (orange arrow) views showing acceptable device position without leak
Ao – aorta, LA – left atrium, RA – right atrium, IAS – interatrial septum
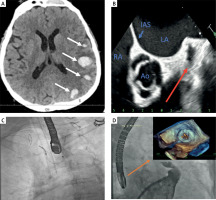
The LAAC procedure was scheduled for several weeks later; however, it was postponed because of a thrombus observed in the left atrial appendage during transesophageal echocardiography (TEE) (Figure 1 B). The patient was discharged on dabigatran 150 mg twice a day. Six weeks later, upon readmission, the initial transesophageal echocardiography (TEE) showed no signs of thrombus; however, during the procedure, a sudden appearance and growth of thrombus in the left atrial appendage (LAA) were noted before device deployment. The patient was again discharged on dabigatran 150 mg twice daily with a combination of aspirin 150 mg/day. Four weeks later, the LAAC procedure, with concomitant application of the Sentinel Cerebral Protection System (Claret Medical, USA), was successfully performed despite the unexpected reappearance of the thrombus during the procedure. In brief, a Sentinel System was introduced via the right radial artery accessed with a 6F sheath over a 0.014″ coronary guidewire in the brachiocephalic artery and left common carotid artery (Figure 1 C). The Watchman FLX LAA 24 mm occluder was subsequently implanted, resulting in complete occlusion of the appendage (Figure 1 D).
The “no-touch technique” with no contrast injection and restriction on the guidewire or catheter manipulation within the appendage was also applied. The procedure and periprocedural stay were uneventful, and the patient was discharged on the third day of hospitalization on a shortened double antiplatelet therapy (DAPT) of aspirin 75 mg once daily and clopidogrel 75 mg once daily for 1 month and later aspirin for 6 months to prevent thrombus formation, keeping in mind the patient’s susceptibility to ICH. Three- and six-month follow-up TEE excluded a leak or thrombus. In the 6-month follow-up period, the patient did not experience heart failure, bleeding, or stroke, and remains in good condition.
CAA is a type of cerebral small vessel disease that increases the risk of spontaneous and recurrent ICH and contributes to age-related cognitive decline. It results from the accumulation or ineffective clearance of amyloid proteins, primarily Aβ, in the brain’s vascular walls [1, 2]. The LAAC procedure allows the patient to stop taking oral anticoagulation therapy (OAT), thereby reducing the risk of ICH and persistent disabilities, but it carries the risk of complications such as tamponade, heart failure, thrombosis, and late leaks, which are uncommon and often manageable [3, 4]. In the present case, the LAAC had to be postponed several times as the patient displayed a unique and rapid formation of thrombus in the LAA, possibly due to atriopathy. Although literature data indicated the minimal risk of migration for thrombi located far from the LAA ostium (e.g., body or apex), in this case, the Cerebral Protection System was used to minimize the risk of an intra-procedural ischemic event. This case highlights CAA as a new potential indication for LAAC in the prevention of disabling cerebral episodes.







