Summary
We aimed to assess the impact of the number of occluded native arteries on prognosis among post-coronary artery bypass grafting (CABG) patients undergoing percutaneous coronary intervention (PCI) for acute coronary syndrome (ACS) treatment. All-cause 1-year mortality was comparable among study groups, reaching 14% in single-, 12% in two- and 14% in multi-vessel disease groups. Our study suggests that in high-risk post-CABG ACS patients undergoing graft PCI, the extent of native-vessel disease does not further worsen short- or mid-term prognosis. While this aligns with guideline recommendations prioritizing native-vessel PCI when feasible, it indicates that graft PCI, when necessary, can still achieve acceptable outcomes even in patients with multivessel native disease.
Introduction
Over 7 million people worldwide each year are diagnosed with acute coronary syndrome (ACS) [1]. Due to the development of both invasive and non-invasive therapeutic strategies, ACS-related mortality has decreased significantly over the last 2 decades to under 5% in all patients, whether unstable angina (UA), ST-elevation (STEMI) or non-ST-elevation myocardial infarction (NSTEMI) ACS is present [2, 3]. In acute cases, such as STEMI-ACS, the gold standard of treatment is percutaneous coronary intervention (PCI). Mortality rates after PCI and CABG do not differ significantly in stable patients; they are however higher in the group in which, due to ACS, urgent coronary artery bypass grafting (CABG) was performed [4]. Post-CABG patients with ACS have a higher risk and poorer prognosis than patients without prior CABG, regardless of whether they are in the STEMI, NSTEMI, or unstable angina group.
Due to older age, higher number of comorbidities developing with age and progression of coronary artery disease or graft failure, PCI is usually the first-choice revascularization procedure for patients with prior CABG presenting with ACS (although it happens less frequently than PCI in native arteries) [5–8]. The patient mortality rate is higher when PCI is performed in a bypass graft compared to surgery in a native artery.
Aim
This study aimed to evaluate whether the number of occluded and stenosed native coronary arteries in post-CABG patients affects their prognosis following PCI ACS.
Material and methods
Study design and patient population
The PL-ACS is a national, multi-centre, ongoing, prospective observational registry that includes data on patients hospitalized with acute coronary syndromes (PL-ACS) in Poland. The registry was created by the Silesian Center for Heart Diseases in Zabrze and the Polish Ministry of Health. Data were collected from PL-ACS Registry questionnaires that include demographic factors (gender, age), clinical presentation on admission (Killip class, heart rate, systolic and diastolic blood pressure, TIMI score), risk factors (smoking, hypertension, hypercholesterolaemia, diabetes mellitus, obesity), prior coronary incidences and related diseases (MI, peripheral vascular disease, stroke, chronic kidney disease, chronic heart failure, COPD, pulmonary oedema, cardiogenic shock, bleeding, cardiac arrest), electrocardiographic abnormalities, left ventricular ejection fraction, related procedures (percutaneous coronary intervention, CABG), coronary intervention details, in-hospital and post-discharge treatment, as well as 1-year follow-up based on the records gathered by the National Health Fund [9, 10]. Missing data were handled using complete-case analysis. Due to the retrospective nature and anonymisation of the collected data in the registry, obtaining the consent of the Bioethics Committee was waived.
Patient characteristics and study variables
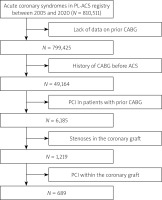
The study was based on a cohort of total of 689 patients who had a history of CABG of native coronary arteries (Figure 1). The patients were selected from the period January 2005 to December 2020. PCI of the bypass graft was performed in 689 patients, constituting our entire research group.
Participants of the study were stratified into three subgroups depending on the number of occluded or significantly stenosed native arteries. The numbers of patients with single-vessel disease (SVD), double-vessel disease (2VD) and triple/multi-vessel disease (MVD) were 181 (26%), 186 (27%) and 322 (47%), respectively. The first group (SVD, n = 181) included patients with a single artery narrowed by ≥ 80%, confirmed by physiological assessment (fractional flow reserve [FFR] < 0.8 and/or instantaneous wave-free ratio [iFR] < 0.89), including those with chronic total occlusions and verified contractility reserves. The second (2VD) and third (MVD) groups were defined by narrowing of two arteries and three or more arteries, respectively, each with ≥ 80% stenosis confirmed by the same physiological criteria (FFR < 0.8 and/or iFR < 0.89).
Of note, not all affected native arteries could be bypassed, and therefore some of them were left, while the others underwent prior, concomitant or subsequent PCI. Additionally, some bypasses may have closed since the index procedure, due to ACS, in a clinically silent manner, or without medical consultation, and consequently without an entry in the patient’s history. In this study, the number of affected native arteries is taken into account; it does not always coincide with the number of actual bypasses performed.
Statistical analysis
Quantitative variables were shown as medians with interquartile ranges (IQR) and compared among groups stratified by the number of affected coronary arteries using the Kruskal-Wallis rank sum test. Categorical variables were presented as absolute and relative frequencies and were evaluated using either the χ2 or Fisher’s test, as appropriate.
The cumulative incidence of all-cause mortality during the 1-year follow-up was illustrated using the Kaplan-Meier method and compared between groups using the log-rank test. The linearity of the association between the number of affected coronary arteries and all-cause mortality was rated applying a likelihood ratio test, comparing the Cox regression model with a linear term against the model including both a linear term and penalized spline.
A multivariable Cox regression model was constructed to adjust for the relationship between the number of affected coronary arteries and all-cause mortality, considering potential confounders, and was based on the results of univariable analysis. Variables with a p-value of < 0.2 in the univariate model or those of clinical significance were included in the multivariable model, which was further refined using a bidirectional elimination approach with entry and stay levels set at 0.05 and 0.15, respectively.
The relationship between the number of occluded or significantly stenosed coronary arteries and in-hospital mortality was analysed using a logistic regression model, adjusted for the same confounders as in the Cox regression analysis. A significance level of p < 0.05 (two-sided) was considered statistically significant. Statistical analyses were conducted using R (R Core Team, 2022) and Statistica, version 13.3 (TIBCO Software, CA, USA).
Results
Study population and general characteristics
Baseline characteristics at admission, stratified into three groups, are summarized in Table I. Males accounted for over 80% of the entire population. The overall median age was 70 years, with medians of 71 years (66; 77), 69 years (64; 75), and 71 years (66; 80) for SVD, 2VD, and MVD, respectively (p = 0.01). Patients were admitted to the hospital at a median time of 6 h from the pain onset. Their median ejection fraction at baseline was 45%. The majority of them (83%) presented non-ST-elevation ACS, although Killip-Kimball class 1 was present in almost four fifths of the whole group. Arterial hypertension (88%), hyperlipidaemia (71%), and diabetes (44%) were frequent in the study cohort. Cardiac arrest before admission was more prevalent in the SVD group (4.5%), as compared to 2VD (1.6%) and MVD groups (0.9%); p = 0.04. On the other hand, chronic kidney disease was more common in the MVD group, with 20% of the patients, compared to 12% and 13% in the SVD and 2VD groups; p = 0.023. Other concomitant diseases and variables, including smoking status and previous revascularization attempts, were comparable between study groups (Table I).
Table I
Baseline clinical characteristics at admission
[i] Data are presented as absolute numbers (percentages) or medians (first quartile, third quartile). Group 1 consisted of patients with single-vessel disease, group 2 of two-vessel disease, and 3 of multivessel disease. CA – cardiac arrest, COPD – chronic obstructive pulmonary disease, DBP – diastolic blood pressure, HR – heart rate, NSTE-ACS – non-ST-segment elevation acute coronary syndrome, PCI – percutaneous coronary intervention, SBP – systolic blood pressure.
Procedural characteristics
These data can be found in the Table II. Before the procedure, TIMI flow 3 (i.e. normal flow in the distal coronary bed) was present in three fourths of the SVD patients, but only in 53% and 39% of the 2VD and MVD patients, respectively. On the other hand, TIMI flow 0 was noted in 11%, 34%, and 53% for SVD, 2VD, and MVD groups, respectively. The distribution of TIMI flow measurements was statistically significant (p < 0.001, Table II). Access site preference was comparable between the groups, as radial and femoral approaches were chosen in roughly half of cases. Technical success of the procedure (TIMI flow 3 after the PCI) was similar among all the groupings, reaching 95% in the SVD group and slightly less for the other groups. No statistically significant difference was found in intra-aortic balloon pump (IABP) or glycoprotein IIb/IIIa inhibitor usage during the PCI.
Table II
Procedural characteristics
[i] Data are presented as absolute numbers (percentages) or medians (first quartile, third quartile). Group 1 consisted of patients with single-vessel disease, group 2 of two-vessel disease, and 3 of multivessel disease. Cx – circumflex, IABP – intra-aortic balloon pump, LAD – left anterior descending, LM – left main, PCI – percutaneous coronary intervention, RCA – right coronary artery, TIMI – Thrombolysis in Myocardial Infarction.
Periprocedural complications and mortality
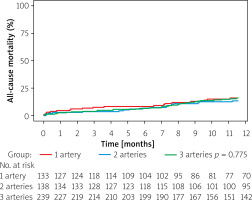
In-hospital and 1-year mortality occurred in 4.8% and 14% of cases in the overall group (Table III). No difference was found when comparing sequelae among the study groups. Similarly, the Kaplan-Meier survival curve (Figure 2) showed that all-cause 1-year mortality was comparable between the groups (p = 0.78), namely: 14% in single-, 12% in two- and 14% in multi-vessel disease.
Table III
Post-procedural complications
Risk factors of 1-year overall mortality
Univariate analysis (Table IV) determined the predictors of 1-year mortality, while the results of the multivariate data confirmed age, chronic heart failure, ejection fraction (%), major bleeding and Killip-Kimball score as the main 1-year mortality predictors (Table IV).
Table IV
Risk factors of mortality at one-year follow-up: uni- and multivariate analysis
[i] HR – hazard ratio, CI – confidence interval, CA – cardiac arrest, COPD – chronic obstructive pulmonary disease, Cx – circumflex, D – diagonal, DBP – diastolic blood pressure, HR – heart rhythm, IM – intermediate, LAD – left anterior descending, LM – left main, Non-STE ACS – non-ST segment elevation acute coronary syndrome, PCI – percutaneous coronary intervention, PTCA – percutaneous transluminal coronary angioplasty, RCA – right coronary artery, SBP – systolic blood pressure, TIMI – thrombolysis in myocardial infarction, TVR – target vessel revascularization.
Discussion
This study showed significant differences between groups in patients’ characteristics such as age, cardiac arrest at admission (before PCI) and chronic kidney disease, as well as differences in peri-procedural TIMI flow and Killip-Kimball grade scale. Despite these differences, data analyses revealed that the number of affected native arteries did not affect long-term survival after PCI in patients who previously underwent CABG. The 1-year mortality was 14%, similar for the whole cohort and in all three subgroups.
Patients with prior CABG account for 3–20% of ACS cases and up to 18.5% of the PCI volume [7, 11–17]. In this study they constituted 6%, with 49,164 out of 810,511 patients. They are considered a high-risk subgroup due to their older age, more comorbidities, and higher mortality rates compared to patients without prior CABG [18].
The study by Gouda et al. showed one-year mortality of 10.6% (15 patients) in STEMI patients and 10.1% (131 patients) in NSTEMI patients, while Stone et al. reported mortality among patients with prior CABG up to 9%. Those findings are similar to the 8% 1-year mortality of NSTE-ACS found in this study [13].
A study by Brilakis et al. showed that approximately 18.5% of all patients operated on both urgently and electively undergoing PCI had prior CABG [17]. Therefore, while occlusion of the native artery can occur after CABG, it is also possible for the bypass graft itself to develop stenosis, presenting as ACS and requiring PCI treatment. According to this study, a median of 36% (32.1–39.8%) of patients will at some point after CABG undergo PCI of the graft, the rate increasing with time after the procedure [17]. In the study population there were patients with heart failure (33% with native coronary and 35% with bypass graft), stable angina (36% vs. 24%), ACS UA (28% vs. 28%), NSTEMI (16% vs. 28%) and STEMI (3% vs. 5%). Out of those post-CABG patients undergoing PCI, a median of 54% presented with ACS (47% of patients with occluded native arteries, 61% of patients with graft failure) in 3.11 years. [17]. Similarly, Cader et al. conducted a study showing that 37.7% of patients with previous CABG presented again with ACS in 4-year follow-up [19].
Data from studies with follow-up up to 5 years show higher mortality rates of patients with PCI of the bypass graft compared to native arteries [6, 7]. Nevertheless, it remains a preferable treatment option, since repeat CABG carries a higher risk of mortality [20].
Since patients with previous cardiac procedures, such as CABG, have a much higher mortality risk compared to those who did not undergo the surgery in the past, the analysis of risk factors is very important [17]. Among them, the SYNTAX score is a well-established method for assessing the severity of changes in coronary arteries taking into account risk factors such as age, left ventricular ejection fraction, chronic obstructive pulmonary disease, kidney function parameters and peripheral vascular disease, as well as morphology of the atherosclerotic changes and the vessels themselves [21]. Although this inference is not supported by empirical data, it results indirectly from the complexity of evaluating the number of significantly narrowed or blocked arteries.
The number of significantly narrowed or fully occluded vessels prior to CABG, regardless of whether they have already been bypassed or not, is related to the SYNTAX score and mortality. According to the SYNTAX Score II nomogram, the higher the score, the greater the probability of mortality within 4 years [21]. As stated by the ESC guidelines, CABG is a recommended treatment method in accordance with scores higher than 32 [11, 22, 23].
Out of the cohort of 689 patients with ACS comparing the outcomes of PCI in single-vessel vs. double-vessel vs. multiple-vessel coronary disease (the number of changed vessels prior to CABG), no significant difference between the three groups was observed in all-cause one-year mortality.
Data considering patients’ baseline characteristics do not differ significantly between the groups in blood pressure, heart rate, history of smoking and other risk factors, which is in line with other studies [6, 24]. The mean age of patients was 70 years (66, 78) and was found to be an important predictor of 1-year mortality in both univariate and multivariate analysis [25–27].
Cardiac arrest was found to be more frequent in patients with SVD before admission. In the MVD group, chronic kidney disease before CABG was more frequent. This may be due to a better vascular adaptation to cardiac ischaemia in patients who have concomitant chronic diseases and in whom MI develops for a long time, causing clinical symptoms later.
Findings considering in-hospital mortality did not meet the statistical significance level of p < 0.05; however, its median rate of 4.8% is higher than 1.79% reported by Brilakis et al. [5, 17]. Regardless, MVD patients have higher in-hospital mortality prognosis than SVD and 2VD, a result corresponding with known literature [24, 28]. Risk factors, such as obesity, diabetes, hyperlipidaemia or hypertension, did not differ significantly between the three groups, which is similar to the results from the Shabbir et al. study [29].
Risk factors revealed by univariate analysis, such as chronic heart failure, heart rate, chronic obstructive pulmonary disease, pulmonary oedema, cardiogenic shock, use of inotropes, major bleeding after surgery and in-hospital CA, correlated with higher risk of 1-year mortality. Results of the multivariate analysis confirmed age, chronic heart failure, ejection fraction (%), major bleeding and Killip-Kimball score as statistically significant overall mortality predictors.
As mentioned, this study’s aim was to assess the impact of the number of native vessels on prognosis expressed as 1-year overall mortality in ACS patient with a prior history of CABG. The end point of all-cause one-year mortality was similar among the three groups, despite differences in selected data in peri-procedural characteristics. This may suggest that different factors may have influenced the risk of mortality in each group. It should also be noted that the use of all-cause mortality as an endpoint could have simply diluted the cardiac-specific associations.
The SVD group was characterized by a higher median age of 71 years and a greater number of cardiac arrests; however, the TIMI flow before surgery had a score of 3 (p < 0.001) in most cases. Patients with 2VD were younger, median 69 years old, and had fewer cases of CA than SVD patients, but there were more recorded previous MI and PCI cases (borderline statistical significance, p = 0.051 and p = 0.068, respectively). The average age of the MVD group was higher than 2VD, at 71 years, more patients suffered from chronic kidney disease (20%, p = 0.023, compared to 12% and 13% in SVD and 2VD groups, respectively) and TIMI flow was 0 before PCI in most patients (53%). However, this group had the fewest cases of CA before admission and fewer previous PCIs (with marginal statistical significance, p = 0.051 and p = 0.068, respectively). These risk factors could have counteracted each other and thus could explain similar results in terms of 1-year mortality in all three groups.
There are several limitations to our analysis. The primary limitation is its retrospective design. The data were collected from reports submitted by various centres, which may introduce heterogeneity. Secondly, the study included a relatively small cohort with a limited number of events, so the neutral findings may partly reflect insufficient power to detect moderate effect sizes. What is more, the study spans 15 years during which practice patterns evolved, which may also have impacted the results. Another limitation is the absence of systematic data on the specific infarct-related artery or graft, which precluded territory-based subgroup analyses. Furthermore, we had access only to aggregated data rather than individual patient-level data, which constrained the depth of statistical analyses possible. Finally, some key factors – including time since CABG, graft type and patency, completeness of revascularisation, use of the left internal mammary artery, and duration of dual antiplatelet therapy – were not captured in the dataset. These variables are known to significantly influence long-term outcomes and may interact with the burden of native-vessel disease.