Introduction
In most cases, dissection of the internal carotid artery (ICA) is asymptomatic [1, 2]. Still, about 10% of these patients can present with neurological symptoms, comprising head and neck pain, Horner’s syndrome, and symptoms of cerebral ischemia, including stroke [3, 4]. Cerebral ischemia can either result from compromised blood flow through the narrowed, dissected part of the ICA, or can be due to distal embolization by thrombi developing in the pseudoaneurysm [2, 5–7]. While a majority of ICA dissection can be managed conservatively, in patients manifesting signs of brain ischemia, especially if they are recurrent, or the pseudoaneurysm is expanding, a decision to perform endovascular repair should be considered. In these patients, particularly those with coexisting pseudoaneurysms, implantation of the covered stent is nowadays the preferred option. However, implantation of such a stent is not always feasible. A tortuous course of the target artery and stiffness of the delivery system of the covered stents both limit the use of these endovascular devices. Alternatively, flow-diverter stents can be used in these patients. However, these stents have their own limitations: a relatively low radial force, which is associated with unacceptably high restenosis rates (up to 20%), and a high cost. Moreover, the implantation technique for flow-diverter stents is challenging, and it is quite often necessary to implant an additional stent in order to address the entire lesion.
Recently, a new carotid stent with novel and promising properties became available. The CARESTO heal stent (Acandis GmbH, Pforzheim, Germany), which is made of coated nitinol composite wires with a platinum core, is primarily intended for standard carotid artery stenting, particularly in patients with vulnerable plaques. However, since this stent is characterized by a dense, single-layer mesh design, which equips it with a flow-diverter characteristic, and is highly flexible, and thus easily navigable through tortuous arteries, it could be used to address ICA dissections coexisting with pseudoaneurysms.
Material and methods
This is a retrospective analysis of the results of treatment for symptomatic dissection of the internal carotid artery, managed with CARESTO heal stents. We used these particular stents in 4 patients. These patients were aged 48 ±8.4 years. Initially, all these patients were managed conservatively. An expanding pseudoaneurysm or signs of cerebral embolism were the indication for an endovascular repair. Clinical and demographic details of these patients are provided in Table I. All patients were informed about potential risks and benefits associated with such treatment, and all of them gave their written consent.
Table I
Demographic, clinical, and diagnostic data, and endovascular interventions
Endovascular procedures
The femoral artery was the preferred access site, and it was used in all patients. First, we performed catheter angiography of the carotid and cerebral arteries. Then, over the Hi-Torque Flex or Hi-Torque Flex-T (Abbott Vascular, Abbott Park, IL, USA) we navigated a 5F or 6F, 90 cm long introducer sheath to the distal part of the common carotid artery. Then, with the support of a 0.014″ guidewire, we introduced the stent delivery system consisting of the NeuroSlider microcatheter (Acandis GmbH, Pforzheim, Germany) and the dilatator catheter (Acandis GmbH, Pforzheim, Germany), which facilitated the navigation. The dilatator was navigated across the lesion, and then the NeuroSlider microcatheter was introduced over the dilatator to the distal part of the target artery, above the lesion. After removal of the dilatator and flushing the system, the stent was ready to deploy. We implanted stents that were tailored to the length of the dissection (Table I). In 1 patient, post-implantation balloon angioplasty of the stenosis, which was still present after stent deployment, was needed. Periprocedurally, patients received unfractionated heparin at a dose of 100 U/kg. In addition, considering the risk of vascular spasm of tortuous carotid arteries, patients received intravenous infusion of a calcium channel blocker (Nimotop, Bayer AG, Germany). In each case, in order to exclude an in-stent thrombosis, 5 and 15 min after stent implantation we performed repeat catheter angiography. In 1 case, we performed intravascular ultrasonography to confirm correct apposition of the stent to the arterial wall.
Follow-ups were performed on the day of the procedure, and then after 1, 3, and 6 months after hospital discharge. Repeat CT angiographies were performed about 1 month after endovascular repair. In all patients, dual antiplatelet therapy was initiated before the procedure. Patients were discharged home with the recommendation of dual antiplatelet therapy (aspirin + clopidogrel or ticagrelor) for at least 3 months, optimally for 3–6 months.
Results
Stents were successfully implanted in all 4 patients. Stents completely covered the dissections and excluded pseudoaneurysms from the circulation (Figures 1 A–G). There were no new neurological events, in-hospital deaths, or other serious adverse event associated with the treatment. There were no new neurological events during the follow-up period. Repeat computed tomography (CT) angiographies in all 4 patients revealed complete coverage of pseudoaneurysms by the stents. There were no new lesions in the area of the previous dissection.
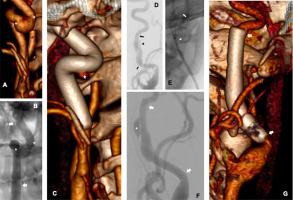
Figure 1
A – Computed tomography (CT) angiography of the right internal carotid artery – dissection is present in the segment between the bends, with a large pseudoaneurysm (asterisks indicate pseudoaneurysm); B – angiography after stent implantation showing persistent inflow to the pseudoaneurysm (arrows indicate the stent, asterisks indicate the pseudoaneurysm); C – repeat CT angiography – preserved artery shape, full coverage of the dissection, small inflow to the pseudoaneurysm (asterisk indicates residual, small flow to the pseudoaneurysm); D – angiography of the left internal carotid artery (arrows indicate the beginning and end of the dissection, arrowhead indicates the true lumen, next to it the pseudoaneurysm is visible); E – initial step of stent deployment (arrow indicates the beginning of the stent, arrowhead the end of the NeuroSlider catheter); F – angiography after stent implantation (arrows indicate the beginning and end of the stent, asterisk – the pseudoaneurysm); G – repeat CT angiography – complete coverage of the lesion, without inflow to the pseudoaneurysm (arrows indicate the beginning and end of the stent)
Discussion
Although pharmacological treatment remains the preferred therapeutic option in most cases of carotid dissections, such a dissection coexisting with pseudoaneurysm should instead be managed invasively. Clinical observations show that during the 2-year follow-up, these pseudoaneurysm do not occlude, and quite often increase. Currently, it is recommended to manage endovascularly those patients with carotid dissection who present with signs of cerebral ischemia, a high-grade stenosis within the pseudoaneurysm, an expanding pseudoaneurysm, or high-grade stenosis of the contralateral carotid artery. Importantly, the rate of adverse events associated with endovascular treatment of the carotid dissections is higher in comparison with carotid stenting for atherosclerotic stenoses. The incidence of such complications following invasive treatment of these dissections can be as high as 16% [8, 9].
Another problem associated with endovascular management of carotid dissections is the fact that often these dissections are found in very tortuous carotid arteries. In such a setting, implantation of the majority of currently available stents is quite challenging. This problem of a tortuous target artery can be overcome by using novel stents. One such promising device is the CARESTO heal stent, which is a coated stent, with nitinol composite wires and a platinum core forming a dense, single-layer mesh. Since the stent and its delivery system are flexible, they can be easily navigated through tortuous arteries. Moreover, the struts of this stent are covered by an anti-thrombogenic HEAL coating, composed of a fibrin-heparin network, which provides another potential clinical benefit. All these features make this stent potentially advantageous in comparison with other devices.
Currently, treatment of such lesions with a covered stent is preferred. Covered stents act as a mechanical barrier, immediately excluding a dissection or pseudoaneurysm. Since they form an impermeable membrane, they prevent blood flow into the false lumen. However, as has already been mentioned, they are less flexible in comparison with flow-diverting stents, and the risk of branch occlusion if the dissection is located in the intracranial part of the ICA cannot be ignored.
Flow-diverting stents, which provide an alternative to covered stents, are better suited for tortuous anatomy, yet the effect of pseudoaneurysm exclusion is delayed, and several weeks or even months are usually needed for complete healing of the lesion and a low radial force, which may lead to restenosis.
All the above-mentioned problems can potentially be overcome by using novel stents. One such promising device is the CARESTO heal stent, which is a coated stent, with nitinol composite wires and a platinum core forming a dense, single-layer mesh. Because of this micro-mesh, this stent provides immediate vascular sealing, similarly to the covered stents. Since the stent and its delivery system are flexible, they can be easily navigated through tortuous arteries, similarly to traditional flow-diverting stents, but unlike them, they possess a high radial force, resulting in full dilatation of the lesion. Additionally, the struts of this stent are covered by an anti-thrombogenic HEAL coating, composed of a fibrin-heparin network, which provides another potential clinical benefit, particularly enhanced endothelialization. All these features make this stent potentially advantageous in comparison with other devices.
It should be noted that the use of this stent is associated with certain limitations. First, the stent is deployed similarly to intracranial stents, but differently from typical carotid stents, where a guidewire is positioned above the lesion. Therefore, the NeuroSlider microcatheter should be positioned quite distally from the lesion, and after the initial step of the stent deployment, its position should be corrected. Our initial observations show that this stent with the delivery system can be easily navigated to the target location of the dissected internal carotid artery. Moreover, it is easy to correct its suboptimal position, since this stent is repositionable up to 96% of its length. On the other hand, the interventionalist should be familiar with the above-discussed mode of stent deployment; thus, experience is required.
This method of stent implantation limits the use of distal protection systems. These systems can be considered until the NeuroSlider catheter is inserted, but the system must be removed before implantation, which is due to the different stent implantation technique. If it is necessary to use the system throughout the procedure, a proximal protection system remains the only available option, which could limit the use of this stent in some patients (lack of vascular access, angiography shows no collateral circulation from the arteries of the other hemisphere, after closing the protection system, a back pressure drop of > 50 mm Hg, contralateral occlusion or critical stenosis).
In this small patient series, the endovascular treatment of carotid dissections proceeded uneventfully. This is in line with our results of endovascular management in similar patients but using different stents [10, 11]. Our initial results suggest that the CARESTO heal stent can be used in patients presenting with carotid dissection requiring endovascular repair, especially if the target artery is tortuous or difficult to navigate for another reason. It should be emphasized, however, that our observations are preliminary and should not be simply extrapolated to similar cases. A larger cohort of patients managed with this particular stent would be necessary, and such a study (probably a multicenter one, considering the rarity of carotid dissections requiring invasive treatment) is warranted. Of note, a recent report from ten European neurovascular centers stated that this stent can be used safely and effectively for endovascular management of carotid stenoses, and the above-mentioned advantages of the CARESTO heal stent were emphasized [12]. The results from our small patient series suggest that carotid dissection could be another indication for the use of this new endovascular device.







