Transcatheter pulmonary valve implantation (TPVI) has revolutionized the management of dysfunctional right ventricular outflow tract (RVOT). Techniques for complex anatomies include valve folding, multi-stent preparations, “doll” or “diabolo” configurations, and even branch pulmonary artery implantation [1, 2]. All rely on Melody (Medtronic) valve delivery using the Ensemble (Medtronic) system.
We report the first case of Melody valve implantation using a balloon-in-balloon (BIB; NuMed) catheter, bypassing the Ensemble system due to mechanical obstruction.
A 9-year-old boy (23 kg) with tetralogy of Fallot, partial Cantrell syndrome, coronary anomaly, horseshoe kidney, cerebral palsy, left hemiparesis, epilepsy, aphasia, mutism, and iodinated contrast allergy (Iomeron; Bracco) was referred for urgent TPVI.
Surgical history included neonatal abdominal repair (diaphragmatic defect, omphalocele, intestinal resection), a 14 mm Contegra (Medtronic) right ventricle to pulmonary artery (PA) conduit at age 1, replacement with a 21 mm Biointegral (Biointegral. Inc.) graft and a 10 × 17 mm Valeo stent (Bard) implanted in the left pulmonary artery (LPA) at age 4.
At age 8, due to RVOT narrowing and distal LPA stent protrusion, he underwent catheterisation with implantation of a 10 × 30 mm Formula (Cook) stent in the LPA and a 39 mm covered CP (NuMed) stent on a 20 × 45 mm BIB in the RVOT.
After syncope and frequent premature ventricular contractions during rehabilitation, urgent TPVI was indicated. Due to contrast allergy and neurological comorbidities, pre-procedural computed tomography (CT) was omitted. Planning relied on virtual reality modelling based on three-dimensional rotational angiography from the previous catheterisation (Figures 1 A, B).
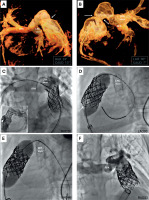
Figure 1
Virtual reality-guided planning and stepwise Melody valve implantation bypassing the Ensemble delivery system. A, B – Virtual reality (VR) reconstructions generated from three-dimensional rotational angiography (3DRA) performed at a prior catheterization. A – Right anterior oblique projection with mild caudal angulation (RAO 33°/CAUD 10°) demonstrating the spatial orientation of the pulmonary arteries (RPA – right pulmonary artery; LPA stent – previously implanted stent in the left pulmonary artery) and proximal narrowing of the right ventricle-to-pulmonary artery (RV-PA) graft (white arrow). VR was used to identify optimal working angles for subsequent catheterization, later confirmed during angiography (C, F). B – Left anterior oblique projection with mild caudal angulation (LAO 90°/CAUD 7°) highlighting the tight proximal narrowing at the RV-PA graft anastomosis (white arrows), which subsequently became the site of proximal infolding of the previously implanted CP stent (NuMed). C–F – Stepwise course of Melody (Medtronic) valve implantation directly on a balloon-in-balloon (BIB, NuMed) catheter, bypassing the Ensemble (Medtronic) delivery system. C – Initial angiography (main panel: RAO 26) demonstrating mild stent narrowing (white arrow) within the right ventricular outflow tract (RVOT). The LPA contains previously implanted stents of appropriate diameter, with mild protrusion into the RVOT but without flow obstruction. A guidewire is advanced deeply into the RPA. Inset (LAO 90) shows proximal RVOT stent infolding (white arrowhead). D – Disruption of the previously implanted RVOT stent (white dashed arrow), reinforced with a 39 mm CP stent on a 22 × 45 mm BIB balloon, creating a secure landing zone for Melody valve deployment. E – Melody valve mounted directly on a 20 × 45 mm BIB catheter positioned in the newly prepared RVOT. A sufficient distance is maintained from the LPA stent, avoiding interference and eliminating the need for deep advancement of the long, bulky proximal shaft (“carrot”) into the pulmonary artery branch. F – Final angiography in the RAO projection (corresponding to panel A) confirming optimal valve expansion, competent function, and unobstructed flow to both pulmonary arteries
The procedure was performed under general anaesthesia, heparinization, systemic corticosteroids, antihistamines, and iso-osmolar contrast (Visipaque, GE Healthcare).
Initial angiography showed proximal RVOT stent infolding and mild LPA stent protrusion into the RVOT (Figure 1 C). Due to contrast allergy and comorbidities, the LPA-stent angioplasty was deferred to minimize contrast exposure and procedure time. RVOT pre-dilatation with a 22 × 20 mm Atlas (Bard) balloon caused tearing of the RVOT stent, which was reinforced with an additional 39 mm CP stent deployed on a 22 × 45 mm BIB catheter (Figure 1 D). Despite correct guidewire position and previous successful stent delivery, the Melody valve could not be advanced on the Ensemble system due to mechanical obstruction from the protruding LPA stent.
The operator removed the entire system, manually crimped the valve onto a 20 × 45 mm BIB balloon, and successfully deployed it over the existing guidewire in the stented RVOT (Figure 1 E), followed by post-dilation with a 22 × 40 mm Atlas balloon. Final angiography confirmed an optimal result (Figure 1 F). The patient was discharged on day 3 on aspirin. Three-month follow-up confirmed competent valve function, no new arrhythmias, and satisfactory exercise tolerance.
The LPA stent, although not obstructing pulmonary flow, mechanically blocked deep pulmonary artery advancement of the Ensemble shaft (“carrot”).
Valve implantation on the shorter BIB balloon avoided this interference and enabled accurate deployment. Pre-emptive LPA stent optimization was considered [3], but deferred due to comorbidities, contrast allergy and already satisfactory LPA branch dimensions.
Fusion imaging techniques may further reduce contrast exposure during TPVI [4].
In conclusion, this experience confirms that direct, BIB-assisted Melody implantation is feasible and a safe bailout option when Ensemble delivery fails, ensuring proper valve positioning without centring or deployment symmetry issues.







